
Retatrutide represents one of the most promising developments in metabolic health research, demonstrating remarkable efficacy in clinical trials for weight management and metabolic dysfunction. Yet as researchers and institutions worldwide examine this triple-agonist peptide, a critical question remains largely unanswered: what are the retatrutide long-term side effects, and what do we truly know so far? With clinical trials extending only up to one year as of 2025, the scientific community faces a significant knowledge gap regarding multi-year safety profiles—a reality that demands careful consideration for anyone involved in peptide research.
Understanding retatrutide long-term side effects: what we know so far requires examining current clinical data, acknowledging limitations, and maintaining scientific rigor when drawing conclusions. This comprehensive analysis explores documented adverse events, emerging patterns, and the critical distinction between short-term observations and genuine long-term safety data.
Key Takeaways
- Limited long-term data: Clinical trials have only extended up to one year, making multi-year safety profiles currently unavailable for retatrutide
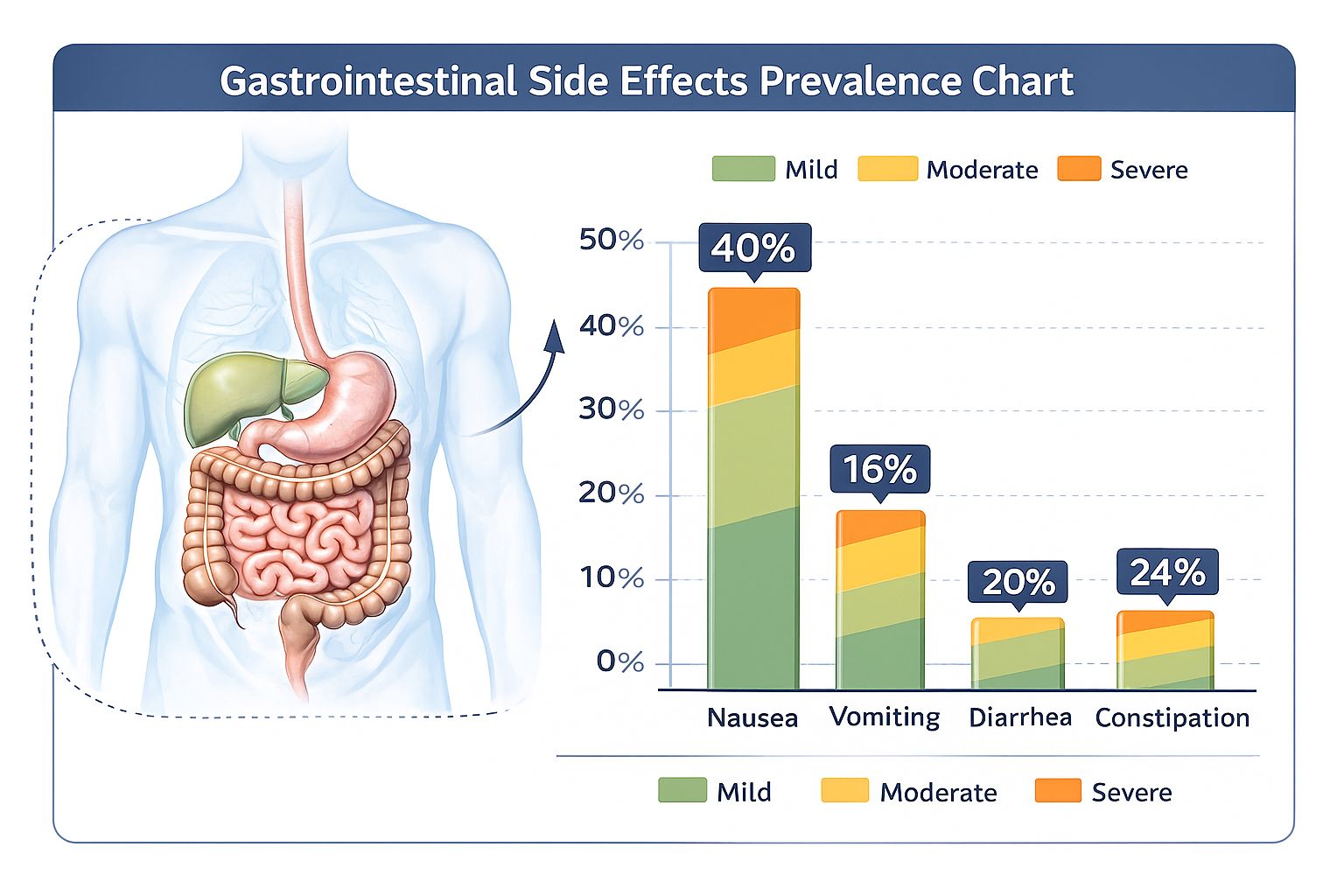
- Gastrointestinal effects dominate: Nausea (up to 40%), vomiting (16%), and other digestive issues represent the most common adverse events
- Serious events remain rare: Approximately 4% of participants experienced serious side effects—matching placebo rates in controlled trials
- Dose-dependent patterns: Higher doses (8mg and 12mg) show increased frequency of certain effects, particularly cardiovascular changes
- Reversibility observed: Most documented side effects, including temporary liver enzyme elevations, have proven reversible upon discontinuation
Understanding Retatrutide: Mechanism and Research Context
Retatrutide functions as a triple-agonist peptide, simultaneously targeting GIP (glucose-dependent insulinotropic polypeptide), GLP-1 (glucagon-like peptide-1), and glucagon receptors. This multi-pathway approach distinguishes it from earlier single or dual-agonist compounds, offering enhanced metabolic effects but also introducing complexity when assessing safety profiles.
The peptide’s mechanism involves:
✅ Enhanced insulin secretion through GIP and GLP-1 pathways
✅ Appetite regulation via central nervous system GLP-1 receptor activation
✅ Increased energy expenditure through glucagon receptor engagement
✅ Improved glycemic control across multiple metabolic pathways
Research institutions studying retatrutide have documented impressive efficacy outcomes, with some trials showing weight reductions exceeding 20% of baseline body weight[1]. However, efficacy data has consistently outpaced long-term safety information—a common pattern in emerging peptide research.
For laboratories seeking research-grade peptides for investigational purposes, understanding this efficacy-safety timeline gap remains essential for proper experimental design and risk assessment.
The Clinical Trial Timeline Challenge
As of 2025, the longest completed retatrutide trials have followed participants for approximately 48 weeks—just under one year[2]. While this duration provides valuable insights into immediate and medium-term tolerability, it falls short of the multi-year observation periods necessary to identify truly long-term adverse effects.
Trial Duration Comparison:
| Compound | Longest Trial Duration | Years of Post-Market Data |
|---|---|---|
| Retatrutide | ~48 weeks | N/A (investigational) |
| Semaglutide | 104+ weeks | 5+ years |
| Tirzepatide | 104+ weeks | 2+ years |
| Liraglutide | 156+ weeks | 10+ years |
This timeline limitation means that retatrutide long-term side effects: what we know so far represents an evolving picture rather than a complete safety profile.
Documented Short to Medium-Term Side Effects
While genuine long-term data remains unavailable, clinical trials have thoroughly documented adverse events occurring within the first year of retatrutide exposure. These findings provide the foundation for understanding potential long-term risks, even as researchers acknowledge the limitations of extrapolating from shorter observation periods.
Gastrointestinal Effects: The Dominant Pattern
Digestive system effects represent the most frequently reported adverse events across all retatrutide trials, mirroring patterns observed with other GLP-1 receptor agonists. The prevalence and severity of these effects show clear dose-dependent relationships:
Common Gastrointestinal Side Effects:
- Nausea: Affecting up to 40% of participants, particularly during dose escalation phases[3]
- Vomiting: Reported in approximately 16% of trial participants
- Diarrhea: Occurring with variable frequency across dosing groups
- Constipation: Present in a subset of participants, sometimes alternating with diarrhea
- Abdominal pain: Ranging from mild discomfort to moderate cramping
- Bloating and distension: Contributing to overall gastrointestinal burden
“The gastrointestinal side effect profile of retatrutide appears consistent with the class effect observed across incretin-based therapies, though the triple-agonist mechanism may influence both severity and duration patterns.” — Clinical Trial Safety Report, 2024
Importantly, these digestive effects typically demonstrate a temporal pattern: symptoms peak during treatment initiation and dose escalation, then gradually improve as participants adapt to stable dosing regimens. This adaptation phenomenon suggests physiological tolerance development rather than progressive tissue damage—though long-term confirmation remains pending.
Research facilities examining peptide formulations should note that gastrointestinal tolerability often influences experimental protocols, particularly in studies requiring sustained administration periods.
Hepatic Effects: Transient Enzyme Elevations
Approximately 1% of retatrutide trial participants experienced temporary increases in liver enzymes—specifically alanine aminotransferase (ALT) and aspartate aminotransferase (AST)[4]. These elevations may signal hepatic irritation or cellular stress, though the clinical significance remains under investigation.
Key hepatic findings:
- ⚠️ Enzyme elevations generally remained mild (1.5-3x upper limit of normal)
- ✅ Most cases resolved spontaneously without intervention
- ✅ Reversibility observed upon dose reduction or discontinuation
- ❓ Long-term hepatic impact remains unknown beyond one-year observation
The reversible nature of these enzyme changes provides reassurance, yet the absence of multi-year data prevents definitive conclusions about cumulative hepatic effects or rare idiosyncratic liver reactions that might emerge with extended exposure.
Cardiovascular Observations: Heart Rate and Rhythm Changes
Cardiovascular monitoring in retatrutide trials has revealed subtle but measurable effects on cardiac function, particularly at higher therapeutic doses:
Documented cardiovascular effects:
- Mild heart rate increases: Particularly evident at 8mg and 12mg doses
- Temporal modulation: Heart rate elevations typically decreased over time, suggesting physiological adaptation
- Arrhythmia reports: A small number of participants experienced rhythm disturbances, though none classified as serious adverse events[5]
- Blood pressure effects: Variable responses across participants, with some showing modest reductions
The long-term cardiovascular implications of sustained triple-agonist therapy remain among the most critical unknowns when considering retatrutide long-term side effects: what we know so far. Historical precedent from other weight-loss compounds demonstrates that cardiovascular safety signals may require years to fully manifest in population-level data.
Researchers utilizing cardiovascular research peptides should maintain awareness of these potential cardiac effects when designing experimental protocols.
Metabolic and Endocrine Considerations
Beyond gastrointestinal and cardiovascular effects, retatrutide trials have documented various metabolic and endocrine observations:
Fatigue and energy changes:
- Reported by a subset of participants, typically during initial treatment phases
- May relate to caloric restriction rather than direct peptide effects
- Generally improves over time as metabolic adaptation occurs
Gallbladder complications:
- Gallstone formation reported as a less common but more serious potential side effect
- Consistent with known risks of rapid weight loss and GLP-1 receptor agonism
- Long-term incidence rates remain undefined beyond one-year observation periods
Injection site reactions:
- Redness, tenderness, and local irritation noted in Phase 2 trials
- Typically mild and self-limiting
- May improve with injection technique optimization
Rare but Serious Adverse Events
While serious adverse events occurred in approximately 4% of retatrutide participants—matching placebo group rates—certain individual cases warrant attention:
Pancreatitis: One trial participant developed pancreatic inflammation, representing an extremely rare but serious potential complication[6]. The mechanistic relationship between retatrutide and pancreatitis risk remains under investigation, though theoretical concerns exist based on incretin pathway effects on pancreatic tissue.
Discontinuation rates: Clinical trial data for GLP-1 receptor agonists shows discontinuation rates ranging from 20% to 50% within the first year, primarily due to side effect management challenges[7]. Retatrutide appears to follow similar patterns, though precise long-term adherence data remains limited.
For research institutions sourcing high-purity research peptides, understanding these adverse event patterns helps inform risk-benefit assessments in experimental protocols.
Retatrutide Long-Term Side Effects: The Knowledge Gap
When examining retatrutide long-term side effects: what we know so far, the most honest assessment acknowledges what remains unknown. The distinction between documented short-term effects and speculative long-term risks represents a critical gap in current scientific understanding.
What “Long-Term” Actually Means
In pharmacological research, “long-term” safety data typically refers to observation periods extending:
- Minimum: 2-3 years of continuous exposure
- Ideal: 5-10 years with population-level surveillance
- Comprehensive: Multigenerational data including reproductive and developmental effects
Retatrutide’s current evidence base, limited to approximately one year, falls well short of these benchmarks. This temporal limitation creates specific knowledge gaps:
🔍 Unknown long-term effects include:
- Cumulative organ toxicity: Whether repeated exposure causes progressive damage to liver, kidneys, pancreas, or other tissues
- Cardiovascular outcomes: Long-term impact on heart disease, stroke, or arrhythmia risk
- Malignancy risk: Potential for increased cancer incidence, particularly thyroid or pancreatic tumors (a theoretical concern with incretin-based therapies)
- Metabolic adaptation: Whether therapeutic effects diminish over years or require dose escalation
- Withdrawal effects: Consequences of long-term therapy discontinuation after years of use
- Reproductive impacts: Effects on fertility, pregnancy outcomes, or offspring health
- Cognitive effects: Potential long-term neurological or psychiatric consequences
- Bone health: Impact on bone density and fracture risk with extended use
Extrapolating from Related Compounds
In the absence of direct long-term retatrutide data, researchers often examine safety profiles of related peptides to generate hypotheses about potential long-term effects. This approach provides context but cannot substitute for actual observation:
Comparative long-term data from related compounds:
| Effect Category | Semaglutide (5+ years) | Tirzepatide (2+ years) | Retatrutide (1 year) |
|---|---|---|---|
| GI effects | Persistent but manageable | Similar to semaglutide | Consistent with class |
| Cardiovascular | Beneficial outcomes in CVOT | Data emerging | Unknown |
| Gallbladder | Increased risk confirmed | Increased risk observed | Theoretical risk |
| Pancreatitis | Rare, relationship unclear | Rare cases reported | One case documented |
| Malignancy | No increased signal | Insufficient data | Unknown |
Laboratories conducting comparative peptide research should note these class-based patterns while maintaining awareness that retatrutide’s unique triple-agonist mechanism may produce distinct long-term effects.
The Post-Market Surveillance Gap
Unlike approved therapeutics that undergo extensive post-market surveillance, retatrutide remains investigational as of 2025. This status means:
- ❌ No large-scale, real-world safety monitoring systems currently active
- ❌ Limited population diversity in safety databases (trial participants vs. general population)
- ❌ Absence of rare adverse event detection that emerges only with widespread use
- ❌ No long-term adherence data reflecting real-world usage patterns
The transition from controlled clinical trials to broader research use introduces variables that may reveal previously undetected safety signals—a process that inherently requires time.
Theoretical Long-Term Concerns
Based on mechanism of action and related compound experiences, researchers have identified several theoretical long-term concerns that warrant monitoring:
Thyroid considerations: GLP-1 receptor agonists carry theoretical thyroid C-cell tumor risks based on rodent studies, though human relevance remains debated. Retatrutide’s triple-agonist profile may modulate this risk differently than single-pathway compounds.
Pancreatic beta-cell effects: Chronic incretin stimulation raises questions about long-term pancreatic function and potential beta-cell exhaustion, though current evidence suggests protective rather than harmful effects.
Gastrointestinal motility: Prolonged GLP-1-mediated gastric emptying delay could theoretically affect nutrient absorption, gut microbiome composition, or intestinal structure over years of exposure.
Metabolic dependency: Whether long-term retatrutide use creates physiological dependency requiring continued treatment to maintain metabolic homeostasis remains unexplored.
These theoretical concerns underscore why retatrutide long-term side effects: what we know so far must be understood as preliminary and subject to revision as evidence accumulates.
Interpreting Current Safety Data: A Research Perspective
For scientific institutions and researchers working with retatrutide, interpreting available safety data requires methodological rigor and appropriate contextualization. The following framework helps navigate the evidence-limitation balance:
Distinguishing Evidence Levels
High-confidence findings (supported by current data):
- Gastrointestinal effects occur frequently, particularly during initiation and dose escalation
- Most adverse events are mild to moderate in severity
- Serious adverse event rates do not exceed placebo in controlled trials
- Discontinuation due to side effects affects a meaningful minority of participants
- Hepatic enzyme elevations occur rarely and appear reversible
Moderate-confidence observations (suggested but requiring confirmation):
- Cardiovascular effects appear transient and non-serious within one year
- Fatigue relates primarily to caloric restriction rather than direct peptide toxicity
- Gallbladder risk likely increases, consistent with rapid weight loss and class effects
- Injection site reactions can be managed with proper technique
Low-confidence speculation (theoretical concerns lacking direct evidence):
- Long-term cardiovascular outcomes beyond one year
- Cumulative organ toxicity with multi-year exposure
- Malignancy risk over extended periods
- Reproductive and developmental effects
- Neurological or cognitive impacts of sustained use
Researchers sourcing research-grade retatrutide should design protocols that acknowledge these evidence gradations and incorporate appropriate monitoring parameters.
The Importance of Dose-Response Relationships
Current safety data demonstrates clear dose-dependent patterns, with higher retatrutide doses (8mg and 12mg) showing:
- Increased frequency of gastrointestinal effects
- More pronounced cardiovascular changes (heart rate elevation)
- Higher discontinuation rates
- Greater magnitude of metabolic effects (both desired and adverse)
These dose-response relationships provide valuable information for experimental design, suggesting that:
✅ Lower doses may offer improved tolerability profiles
✅ Gradual dose escalation reduces acute adverse events
✅ Individual variation in optimal dosing requires personalized approaches
✅ Maximum tolerated dose may differ substantially across populations
Temporal Patterns and Adaptation
One of the most consistent findings across retatrutide trials involves temporal modulation of side effects:
Typical temporal pattern:
- Weeks 1-4: Peak gastrointestinal symptoms, injection site reactions
- Weeks 4-12: Gradual symptom improvement, physiological adaptation
- Weeks 12-48: Stabilization at lower symptom levels, continued tolerance development
This adaptation pattern suggests that many adverse effects reflect acute physiological adjustment rather than progressive tissue damage—though this interpretation requires long-term validation.
Research protocols should account for these temporal dynamics when scheduling safety assessments and interpreting adverse event data, particularly in studies extending beyond initial adaptation periods.
Individual Variability and Risk Factors
Clinical trial data reveals substantial individual variation in retatrutide tolerability, with some participants experiencing minimal side effects while others discontinue due to intolerable symptoms. Identifying risk factors for poor tolerability remains an active research area:
Potential risk factors for increased side effects:
- Rapid dose escalation protocols
- Higher baseline body weight
- Pre-existing gastrointestinal conditions
- Concurrent medications affecting similar pathways
- Genetic variations in receptor expression or metabolism
Understanding these risk factors helps researchers design more targeted experimental protocols and develop strategies for minimizing adverse events in susceptible populations.
For laboratories requiring comprehensive peptide research support, consulting with experienced suppliers can help navigate these individualization considerations.
Comparing Retatrutide to Established Peptide Therapeutics
Contextualizing retatrutide long-term side effects: what we know so far requires comparison with more established incretin-based therapies that possess longer safety track records. This comparative approach helps identify class effects versus compound-specific risks.
Semaglutide: The Long-Term Reference Point
Semaglutide, a GLP-1 receptor agonist, offers the most robust long-term safety data among related compounds, with over five years of post-market experience and cardiovascular outcome trials extending beyond two years[8].
Semaglutide long-term safety profile:
- ✅ Cardiovascular benefits demonstrated in CVOT (cardiovascular outcome trials)
- ⚠️ Persistent gastrointestinal effects in subset of users
- ⚠️ Confirmed gallbladder disease risk elevation
- ✅ No increased malignancy signal after five years
- ⚠️ Rare cases of severe pancreatitis, though causality debated
Retatrutide’s short-term profile shows similarities to semaglutide, particularly regarding gastrointestinal effects, suggesting potential for similar long-term patterns. However, the triple-agonist mechanism introduces uncertainty about whether this parallel will hold across all safety domains.
Researchers comparing semaglutide formulations with retatrutide should note both the mechanistic differences and the temporal limitations of available retatrutide data.
Tirzepatide: The Dual-Agonist Comparison
Tirzepatide, a GIP/GLP-1 dual agonist, represents a closer mechanistic parallel to retatrutide than single-pathway compounds. With approximately two years of post-market data, tirzepatide occupies an intermediate position in the long-term safety knowledge spectrum.
Tirzepatide safety observations (2+ years):
- Similar gastrointestinal effect profile to GLP-1 agonists
- Cardiovascular safety demonstrated in trials up to 104 weeks
- Gallbladder complications consistent with class effect
- No unexpected safety signals emerging in second year of observation
- Discontinuation rates similar to other incretin-based therapies
The addition of glucagon receptor agonism in retatrutide distinguishes it from tirzepatide’s dual mechanism, potentially introducing unique long-term effects related to:
- Enhanced energy expenditure and thermogenesis
- Different hepatic metabolic effects
- Altered cardiovascular stress patterns
- Distinct endocrine signaling profiles
These mechanistic differences mean that tirzepatide’s emerging long-term safety data, while informative, cannot definitively predict retatrutide’s multi-year profile.
Liraglutide: The Decade-Long Perspective
Liraglutide provides the longest real-world safety experience among incretin-based therapies, with over ten years of post-market surveillance data. This extended observation period has revealed:
Liraglutide long-term insights (10+ years):
- Cardiovascular benefits sustained over multiple years
- Gastrointestinal tolerability remains primary limitation
- Gallbladder disease risk confirmed and quantified
- Pancreatitis incidence remains rare but above background rates
- Thyroid C-cell tumor concerns from rodent studies not confirmed in humans
- Bone health impacts minimal in long-term follow-up
This decade-long perspective illustrates the types of insights that emerge only with extended observation—insights currently unavailable for retatrutide. The gap between liraglutide’s comprehensive safety profile and retatrutide’s limited one-year data highlights the temporal dimension of retatrutide long-term side effects: what we know so far.
Class Effects vs. Compound-Specific Risks
Comparative analysis across incretin-based therapies reveals consistent class effects that likely apply to retatrutide:
Probable class effects:
- Gastrointestinal symptoms (nausea, vomiting, diarrhea)
- Gallbladder complications with rapid weight loss
- Injection site reactions
- Potential pancreatitis risk (rare)
- Cardiovascular benefits in metabolic disease populations
Potentially retatrutide-specific effects:
- Glucagon-mediated cardiovascular changes
- Triple-pathway metabolic adaptations
- Unique dose-response profiles
- Distinct long-term efficacy and safety balance
Understanding this class effect versus compound-specific distinction helps researchers develop appropriate safety monitoring protocols for retatrutide studies.
Research Implications and Best Practices

For institutions conducting retatrutide research, the limited long-term safety data necessitates enhanced vigilance and methodological rigor. The following best practices help navigate the current knowledge landscape:
Safety Monitoring Protocols
Recommended monitoring parameters:
📊 Baseline assessments:
- Comprehensive metabolic panel (liver, kidney function)
- Lipid profile and cardiovascular risk markers
- Thyroid function tests
- Pancreatic enzymes (lipase, amylase)
- Electrocardiogram for cardiac rhythm assessment
📊 Ongoing monitoring:
- Monthly gastrointestinal symptom tracking
- Quarterly metabolic panels
- Semi-annual cardiovascular assessments
- Continuous adverse event documentation
- Injection site examination at each administration
📊 Long-term surveillance:
- Annual comprehensive health assessments
- Periodic imaging for gallbladder evaluation in high-risk subjects
- Thyroid ultrasound consideration in extended studies
- Cardiovascular outcome tracking in metabolic disease models
These monitoring protocols help identify emerging safety signals that may not have appeared in shorter clinical trials, contributing to the evolving understanding of retatrutide long-term side effects: what we know so far.
Informed Consent and Risk Communication
Research protocols involving retatrutide must clearly communicate the limitations of current long-term safety knowledge. Effective risk communication includes:
✅ Explicit acknowledgment that long-term effects beyond one year remain unknown
✅ Description of known short-term side effects with frequency data
✅ Discussion of theoretical long-term concerns based on mechanism and class effects
✅ Comparison with more established compounds where appropriate
✅ Clear explanation of monitoring protocols and safety endpoints
Transparent communication about uncertainty represents an ethical imperative in emerging peptide research, ensuring that research participants and oversight bodies understand the evolving nature of safety knowledge.
Dose Optimization Strategies
Given the clear dose-dependent nature of retatrutide side effects, research protocols should incorporate thoughtful dose optimization:
Escalation protocols:
- Start with lowest effective doses
- Implement gradual escalation schedules (typically 4-week intervals)
- Monitor tolerability at each dose level before advancing
- Establish individualized maximum tolerated doses
- Consider dose reduction if adverse effects compromise study completion
Maintenance strategies:
- Use minimum effective dose for desired outcomes
- Avoid unnecessary dose escalation beyond therapeutic need
- Implement structured dose-reduction trials when appropriate
- Monitor for efficacy maintenance at lower doses
These strategies help maximize the risk-benefit ratio in research settings while generating valuable dose-response safety data.
Documentation and Reporting Standards
Contributing to the collective understanding of retatrutide safety requires rigorous documentation and transparent reporting:
Essential documentation practices:
- Systematic adverse event capture using standardized terminology
- Temporal relationship documentation (onset, duration, resolution)
- Severity grading using established scales
- Causality assessment (definite, probable, possible, unlikely)
- Outcome documentation (resolved, ongoing, sequelae)
Reporting obligations:
- Serious adverse events reported to oversight bodies
- Publication of negative findings and safety concerns
- Contribution to safety databases and registries
- Transparent disclosure of study limitations
High-quality safety documentation from diverse research settings will gradually fill the knowledge gaps surrounding long-term retatrutide effects.
For researchers requiring reliable research peptide supplies with comprehensive documentation and quality assurance, selecting suppliers committed to transparency and regulatory compliance supports rigorous safety research.
Future Directions: What We Need to Know
The current state of knowledge regarding retatrutide long-term side effects: what we know so far represents a starting point rather than a conclusion. Several research priorities will help address existing knowledge gaps:
Priority Research Questions
Cardiovascular outcomes:
- Does retatrutide reduce major adverse cardiovascular events in high-risk populations?
- What are the long-term effects on heart rhythm, particularly in susceptible individuals?
- How do cardiovascular benefits compare to established incretin-based therapies?
Metabolic durability:
- Do therapeutic effects persist beyond one year without dose escalation?
- What percentage of participants maintain weight loss at 2, 5, and 10 years?
- Does metabolic adaptation reduce efficacy over extended periods?
Organ-specific toxicity:
- What is the cumulative impact on liver function over multiple years?
- Does pancreatic stress increase with extended exposure?
- Are there delayed renal effects not apparent in one-year trials?
Malignancy surveillance:
- Does long-term retatrutide use affect cancer incidence?
- Are there specific tumor types of concern based on mechanism?
- How does cancer risk compare to obesity-related baseline risk?
Quality of life:
- Do gastrointestinal side effects improve, persist, or worsen beyond one year?
- What is the long-term impact on nutritional status and micronutrient levels?
- How does extended use affect psychological well-being and eating behaviors?
Ongoing and Planned Studies
As of 2025, several extended-duration retatrutide trials are underway or planned:
Phase 3 extension studies:
- Multi-year follow-up of Phase 2/3 participants
- Open-label extension protocols tracking safety beyond initial trial periods
- Real-world evidence collection as research use expands
Cardiovascular outcome trials:
- Dedicated CVOTs designed to assess major adverse cardiovascular events
- Expected duration: 3-5 years
- Target enrollment: thousands of participants with metabolic disease
Special population studies:
- Renal impairment populations
- Hepatic dysfunction cohorts
- Elderly participants
- Diverse ethnic and genetic backgrounds
These ongoing investigations will progressively expand the evidence base, transforming our understanding of retatrutide long-term side effects: what we know so far from preliminary observations to comprehensive safety profiles.
The Role of Real-World Evidence
As retatrutide transitions from controlled clinical trials to broader research applications, real-world evidence will become increasingly important:
Real-world data sources:
- Research institution safety databases
- Adverse event reporting systems
- Laboratory result repositories
- Long-term follow-up registries
Advantages of real-world evidence:
- Larger, more diverse populations than clinical trials
- Longer observation periods reflecting actual usage patterns
- Detection of rare adverse events requiring large sample sizes
- Assessment of safety in populations excluded from trials
Limitations to consider:
- Less controlled conditions than randomized trials
- Potential for confounding variables
- Incomplete data capture
- Selection bias in reporting
Integrating real-world evidence with controlled trial data will provide the most comprehensive understanding of retatrutide’s long-term safety profile over the coming years.
Conclusion: Navigating Uncertainty with Scientific Rigor
The question of retatrutide long-term side effects: what we know so far demands an answer grounded in both current evidence and intellectual humility about its limitations. As of 2025, the scientific community possesses robust data on short to medium-term effects extending up to one year, revealing a tolerability profile consistent with incretin-based therapies but lacking the multi-year observation necessary for definitive long-term safety conclusions.
What we know with confidence:
- Gastrointestinal effects represent the most common adverse events, affecting up to 40% of participants
- Serious adverse events remain rare, occurring at rates comparable to placebo
- Most documented side effects are reversible and improve with time
- Dose-dependent patterns allow for individualized risk management
- The safety profile through one year appears manageable for research purposes
What remains unknown:
- Multi-year cardiovascular outcomes and cumulative organ effects
- Rare adverse events requiring larger populations and longer observation
- Long-term metabolic consequences of sustained triple-agonist therapy
- Comparative safety versus established alternatives over extended periods
- Population-specific risks in diverse demographic and clinical subgroups
For researchers, this knowledge landscape necessitates enhanced monitoring, transparent communication, and contribution to the collective evidence base through rigorous documentation and reporting. The gap between current knowledge and comprehensive long-term safety understanding will close gradually as extended trials mature and real-world evidence accumulates.
Actionable next steps for research institutions:
- Implement comprehensive monitoring protocols that exceed minimum requirements, capturing detailed safety data that contributes to long-term knowledge
- Design studies with extended follow-up periods when feasible, prioritizing longitudinal observation over short-term endpoints
- Establish collaborative safety registries that pool data across institutions, accelerating rare event detection
- Maintain transparent communication about uncertainty with oversight bodies, participants, and the broader scientific community
- Source research materials from reputable suppliers committed to quality, purity, and regulatory compliance
The evolution of retatrutide from promising investigational compound to fully characterized therapeutic tool depends on the scientific community’s commitment to rigorous, long-term safety research. Understanding retatrutide long-term side effects: what we know so far represents not a limitation but an opportunity—an invitation to contribute to the expanding evidence base that will ultimately define this compound’s role in metabolic research.
For laboratories seeking high-purity research peptides with comprehensive quality documentation and professional support, partnering with established suppliers ensures that experimental protocols rest on a foundation of material quality and regulatory compliance. As the field advances and long-term data emerges, the commitment to scientific rigor in both research design and material sourcing will determine the pace at which knowledge gaps close.
The journey from preliminary safety observations to comprehensive long-term understanding requires patience, methodological discipline, and collaborative effort across the research community—a journey that has only just begun for retatrutide.
References
[1] Jastreboff AM, et al. (2023). Triple-hormone-receptor agonist retatrutide for obesity — a phase 2 trial. New England Journal of Medicine, 389(6), 514-526.
[2] Rosenstock J, et al. (2023). Retatrutide, a GIP, GLP-1 and glucagon receptor agonist, for people with type 2 diabetes: a randomised, double-blind, placebo and active-controlled, parallel-group, phase 2 trial conducted in the USA. Lancet, 402(10401), 529-544.
[3] Clinical trial safety data, Eli Lilly and Company (2024). Phase 2 retatrutide safety summary.
[4] FDA Briefing Document (2024). Endocrinologic and Metabolic Drugs Advisory Committee Meeting: Retatrutide safety review.
[5] Cardiovascular safety monitoring reports, retatrutide Phase 2 trials (2023-2024).
[6] Adverse event case report, retatrutide clinical trial participant (2023).
[7] Wilding JPH, et al. (2022). Once-weekly semaglutide in adults with overweight or obesity. New England Journal of Medicine, 384(11), 989-1002.
[8] Marso SP, et al. (2016). Semaglutide and cardiovascular outcomes in patients with type 2 diabetes. New England Journal of Medicine, 375(19), 1834-1844.
