
The landscape of weight management treatment in the UK has undergone a dramatic transformation in 2025, with tirzepatide emerging as one of the most discussed pharmaceutical interventions for obesity and type 2 diabetes. As thousands of patients seek access through the National Health Service, understanding Tirzepatide & the NHS: Eligibility, Criteria and Access has become essential for both healthcare professionals and individuals exploring treatment options. The complexities of NHS prescribing guidelines, regional variations in availability, and stringent eligibility requirements create a challenging pathway that demands comprehensive understanding.
This article provides an authoritative examination of how tirzepatide is accessed through the NHS, who qualifies for treatment, and what alternatives exist for those who don’t meet the strict criteria. Whether you’re a healthcare provider navigating prescribing protocols or a patient considering treatment options, this guide delivers the professional insights needed to make informed decisions.
Key Takeaways
- NHS eligibility for tirzepatide requires a BMI of 35+ with weight-related comorbidities or 30+ with type 2 diabetes, alongside participation in specialist weight management programmes
- Access pathways vary significantly across England, Scotland, Wales, and Northern Ireland, with most patients requiring referral to tier 3 or 4 specialist services
- Treatment duration on the NHS is typically limited to 12-24 months, with continuation dependent on achieving specific weight loss targets (typically 5% in 3 months, 15% in 12 months)
- Private prescription alternatives exist but involve substantial ongoing costs (£200-400+ monthly), while research-grade peptides serve entirely different purposes
- NICE guidance continues to evolve throughout 2025, with regional Integrated Care Boards (ICBs) implementing varying local policies that affect availability
Understanding Tirzepatide: Mechanism and Clinical Applications

Tirzepatide represents a significant advancement in metabolic medicine, functioning as a dual glucose-dependent insulinotropic polypeptide (GIP) and glucagon-like peptide-1 (GLP-1) receptor agonist. This innovative mechanism distinguishes it from earlier single-pathway medications, offering enhanced efficacy in both glycaemic control and weight reduction.[1]
How Tirzepatide Works
The medication operates through multiple complementary pathways:
Dual Receptor Activation: By simultaneously targeting both GIP and GLP-1 receptors, tirzepatide creates a synergistic effect that surpasses single-agonist therapies. The GIP component enhances insulin secretion and may reduce food intake, while the GLP-1 component slows gastric emptying, increases satiety, and improves insulin sensitivity.[2]
Metabolic Effects: Clinical trials have demonstrated average weight reductions of 15-22% of body weight over 72 weeks, with corresponding improvements in cardiovascular risk markers including blood pressure, lipid profiles, and inflammatory markers.[3]
Glycaemic Control: For patients with type 2 diabetes, tirzepatide consistently achieves HbA1c reductions of 2.0-2.5%, often bringing patients to target levels below 7% without significant hypoglycaemia risk.[4]
Licensed Indications in the UK
As of 2025, tirzepatide holds regulatory approval from the Medicines and Healthcare products Regulatory Agency (MHRA) for:
- Type 2 diabetes management as an adjunct to diet and exercise
- Chronic weight management in adults with obesity (BMI ≥30 kg/m²) or overweight (BMI ≥27 kg/m²) with at least one weight-related comorbidity
The medication is administered via weekly subcutaneous injection, with dosing escalated gradually from 2.5mg to maintenance doses of 5mg, 10mg, or 15mg depending on individual response and tolerability.
NHS Eligibility Criteria for Tirzepatide Prescription
Understanding Tirzepatide & the NHS: Eligibility, Criteria and Access requires careful examination of the specific criteria that determine who can receive this treatment through public healthcare. The NHS operates under strict guidelines developed by the National Institute for Health and Care Excellence (NICE), with additional local variations implemented by regional commissioning bodies.
Primary Eligibility Requirements
The baseline criteria for NHS-funded tirzepatide treatment include:
Body Mass Index (BMI) Thresholds:
- BMI ≥35 kg/m² with at least one weight-related comorbidity (hypertension, type 2 diabetes, dyslipidaemia, obstructive sleep apnoea, cardiovascular disease), OR
- BMI ≥30 kg/m² specifically for patients with type 2 diabetes requiring improved glycaemic control
Adjusted Criteria for Specific Populations:
- For individuals from South Asian, Chinese, other Asian, Middle Eastern, Black African, or African-Caribbean backgrounds, BMI thresholds are reduced by 2.5 kg/m² due to increased metabolic risk at lower BMI levels[5]
- Age restrictions typically apply (18-75 years), though some specialist services may consider treatment outside these boundaries
Weight-Related Comorbidities
Qualifying comorbidities that satisfy NHS criteria include:
| Comorbidity | Clinical Definition |
|---|---|
| Type 2 Diabetes | HbA1c ≥48 mmol/mol or established diagnosis with ongoing treatment |
| Hypertension | Blood pressure ≥140/90 mmHg or current antihypertensive medication |
| Dyslipidaemia | Total cholesterol >5.0 mmol/L or current lipid-lowering therapy |
| Obstructive Sleep Apnoea | Confirmed diagnosis via sleep study (AHI ≥5) |
| Non-alcoholic Fatty Liver Disease | Confirmed via imaging or biopsy with evidence of steatosis |
| Cardiovascular Disease | Previous MI, stroke, or established coronary artery disease |
Pre-Treatment Requirements
Before NHS prescription of tirzepatide, patients must typically demonstrate:
✅ Engagement with lifestyle interventions: Documented participation in structured weight management programmes for at least 3-6 months, including dietary modification and increased physical activity
✅ Psychological assessment: Evaluation to ensure understanding of treatment expectations, commitment to lifestyle changes, and absence of contraindications such as active eating disorders
✅ Failed previous interventions: Evidence that conventional approaches (diet, exercise, behavioural therapy) have not achieved adequate weight loss (typically defined as <5% body weight reduction)
✅ Medical optimization: Stable management of existing comorbidities, with diabetes patients often required to have tried metformin and at least one other glucose-lowering agent
Exclusion Criteria
The NHS will not prescribe tirzepatide to individuals with:
- Personal or family history of medullary thyroid carcinoma
- Multiple endocrine neoplasia syndrome type 2 (MEN2)
- Severe gastrointestinal disease
- Pregnancy or planned pregnancy within treatment period
- History of pancreatitis (relative contraindication requiring specialist assessment)
- Severe renal impairment (eGFR <30 mL/min/1.73m²)
Understanding these criteria is crucial when considering treatment pathways and options available through different channels.
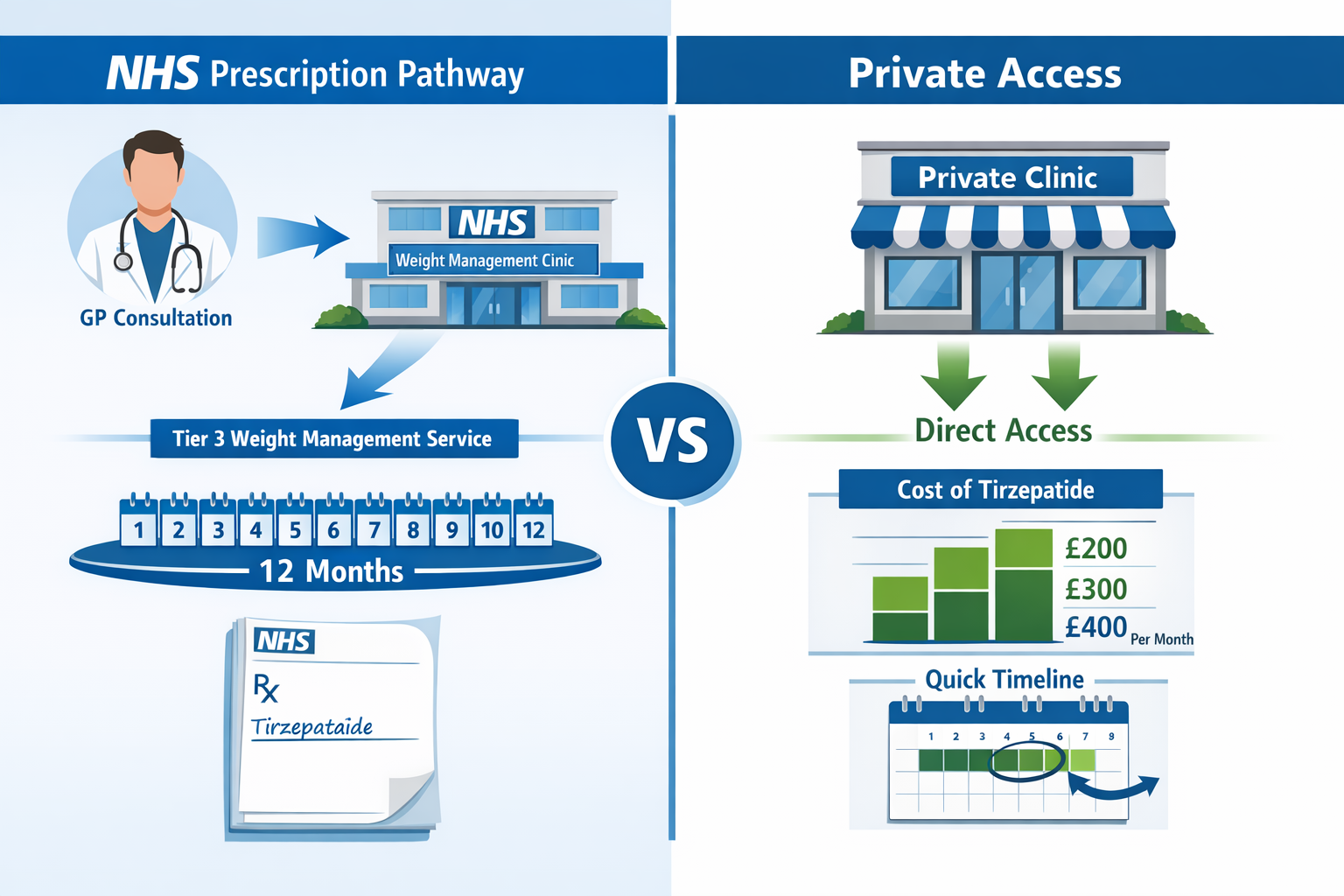
NHS Access Pathways: How to Get Tirzepatide Prescribed
Navigating Tirzepatide & the NHS: Eligibility, Criteria and Access involves understanding the complex referral pathways and service tiers that govern weight management treatment in the UK. Access is rarely straightforward, with most patients requiring progression through multiple healthcare levels.
The Tiered Approach to Weight Management
The NHS operates a structured four-tier system for obesity management:
Tier 1: Universal Services Primary prevention delivered through public health initiatives, community programmes, and general lifestyle advice. This tier is non-specialist and available to all.
Tier 2: Lifestyle Weight Management Services Community-based programmes offering structured diet, physical activity, and behavioural support. Typically 12-week interventions delivered by trained practitioners. Most areas require completion of Tier 2 before progression.
Tier 3: Specialist Weight Management Services Multidisciplinary teams including specialist physicians, dietitians, psychologists, and physiotherapists. This is where tirzepatide prescribing typically occurs. Services assess complex cases, manage pharmacotherapy, and prepare patients for potential bariatric surgery.
Tier 4: Bariatric Surgery Surgical interventions for severe obesity, usually requiring prior engagement with Tier 3 services.
Step-by-Step Access Process
Step 1: GP Consultation and Initial Assessment
The journey begins with your general practitioner who will:
- Calculate BMI and assess weight-related comorbidities
- Review medical history and current medications
- Discuss lifestyle interventions attempted
- Provide referral to Tier 2 services if appropriate
Step 2: Tier 2 Programme Participation
Most ICBs require documented engagement with lifestyle programmes:
- 12-16 week structured intervention
- Regular attendance and participation records
- Measurement of weight loss outcomes
- Assessment of readiness for further intervention
Step 3: Specialist Referral (Tier 3)
If Tier 2 interventions prove insufficient, GPs can refer to specialist services where:
- Comprehensive medical assessment occurs
- Psychological evaluation is conducted
- Suitability for pharmacotherapy is determined
- Treatment plans are developed
Step 4: Tirzepatide Initiation
Once approved by the specialist team:
- Baseline measurements are recorded (weight, BMI, HbA1c, lipids, blood pressure)
- Injection technique training is provided
- Starting dose (2.5mg weekly) is prescribed
- Follow-up schedule is established
Step 5: Ongoing Monitoring and Review
Treatment continuation requires:
- Monthly reviews initially, then quarterly
- Achievement of weight loss targets (5% at 3 months, 15% at 12 months)
- Assessment of side effects and tolerability
- Continued engagement with lifestyle modifications
Regional Variations in Access
Tirzepatide & the NHS: Eligibility, Criteria and Access varies significantly across the UK’s devolved healthcare systems:
England: Access determined by local Integrated Care Boards (ICBs), with significant postcode variation. Some ICBs have restricted tirzepatide to specialist centres only, while others permit broader primary care prescribing under shared care protocols.
Scotland: NHS Scotland follows similar NICE guidance but with interpretation by regional health boards. Access through specialist weight management services is generally more consistent than in England.
Wales: Tirzepatide availability through NHS Wales remains limited as of early 2025, with many health boards restricting access pending further health economic evaluations.
Northern Ireland: The Health and Social Care Board has implemented cautious rollout, with tirzepatide primarily available through specialist diabetes and endocrinology services.
Typical Waiting Times
Realistic timelines for NHS access include:
- GP to Tier 2 referral: 2-8 weeks
- Tier 2 programme completion: 12-16 weeks
- Tier 2 to Tier 3 referral: 4-12 weeks
- Tier 3 initial assessment: 8-20 weeks
- Treatment initiation: 2-4 weeks post-approval
Total timeline: 6-12 months from initial GP consultation to first tirzepatide injection, with significant regional variation.
For those seeking to understand the broader context of peptide research and applications, PEPTIDE PRO provides educational resources on research-grade compounds, though it’s crucial to distinguish between clinical therapeutics and research materials.
Treatment Protocols and Continuation Criteria
Once tirzepatide treatment begins through the NHS, specific protocols govern dosing, monitoring, and continuation. Understanding these requirements is essential for maintaining access to this medication.
Dosing Schedule and Titration
The standard NHS protocol follows manufacturer guidance:
| Week | Dose | Purpose |
|---|---|---|
| 1-4 | 2.5mg | Initiation and tolerability assessment |
| 5-8 | 5.0mg | First maintenance dose option |
| 9-12 | 7.5mg | Intermediate escalation (if needed) |
| 13-16 | 10mg | Higher maintenance dose |
| 17-20 | 12.5mg | Further escalation option |
| 21+ | 15mg | Maximum approved dose |
Dose escalation is individualized based on:
- Weight loss response
- Glycaemic control (for diabetes patients)
- Gastrointestinal tolerability
- Patient preference and treatment goals
Monitoring Requirements
NHS protocols mandate regular assessment:
Baseline Measurements:
- Weight, height, BMI, waist circumference
- Blood pressure
- HbA1c, fasting glucose
- Lipid profile (total cholesterol, HDL, LDL, triglycerides)
- Liver function tests
- Renal function (eGFR, creatinine)
- Thyroid function (TSH)
Ongoing Monitoring:
- Monthly (first 3 months): Weight, blood pressure, side effect assessment
- Quarterly: Comprehensive metabolic panel, HbA1c (diabetes patients)
- Annually: Full baseline panel repeated
Continuation Criteria: The “Stop Rules”
NHS funding for tirzepatide continuation depends on achieving specific outcomes:
3-Month Review:
- Minimum 5% body weight reduction required
- Failure to achieve this target typically results in treatment discontinuation
- Exceptions may be made for significant HbA1c improvement in diabetes patients
12-Month Review:
- Minimum 15% body weight reduction from baseline expected for obesity indication
- Diabetes patients may continue with lower weight loss if HbA1c targets achieved (<7% or 53 mmol/mol)
- Assessment of comorbidity improvement (blood pressure, lipids, sleep apnoea)
Beyond 12 Months:
- Treatment typically limited to 24 months maximum on NHS
- Continuation beyond 2 years requires exceptional clinical justification
- Maintenance of weight loss and continued engagement with lifestyle measures required
Managing Side Effects
Common adverse effects influence treatment continuation:
Gastrointestinal Effects (most common):
- Nausea (20-40% of patients)
- Diarrhoea (15-25%)
- Constipation (10-20%)
- Vomiting (5-15%)
Management strategies include:
- Slower dose escalation
- Dietary modifications (smaller meals, avoiding high-fat foods)
- Temporary dose reduction
- Anti-emetic medication if needed
Serious Adverse Events requiring immediate medical review:
- Severe abdominal pain (possible pancreatitis)
- Visual changes or neck mass (thyroid concerns)
- Persistent vomiting with dehydration
- Acute kidney injury symptoms
Treatment Cessation and Transition
When NHS-funded tirzepatide ends:
Planned Discontinuation:
- Gradual dose reduction not typically required (unlike some medications)
- Intensive lifestyle support to maintain weight loss
- Consideration of alternative weight management strategies
- Monitoring for weight regain
Weight Maintenance Post-Treatment:
- Continued engagement with Tier 2/3 services
- Regular follow-up (3-6 monthly)
- Possible consideration of alternative anti-obesity medications
- Bariatric surgery referral if significant weight regain occurs
Private Prescription and Alternative Access Routes
For individuals who don’t meet NHS criteria or face prohibitive waiting times, private prescription represents an alternative pathway for accessing tirzepatide. However, this route involves substantial financial commitment and different regulatory considerations.
Private Healthcare Access
Private Specialist Consultation:
- Initial assessment with private endocrinologist or obesity specialist: £200-400
- Comprehensive metabolic screening: £150-300
- Ongoing consultation fees: £100-200 per review
Medication Costs:
- Tirzepatide (Mounjaro®) pricing varies by dose and supplier
- Monthly costs typically range: £200-400+
- Annual treatment cost: £2,400-4,800+
- No NHS prescription charge applies; full commercial price paid
Private Clinic Models:
Several private healthcare models have emerged in 2025:
- Traditional Private Hospitals: Formal specialist-led services with comprehensive medical oversight, highest cost but most thorough care
- Specialist Weight Loss Clinics: Dedicated obesity medicine practices offering streamlined access, mid-range pricing
- Online Prescription Services: Telemedicine platforms providing remote consultations and home delivery, often lower cost but variable quality of care
Regulatory Considerations for Private Prescription
Private prescribers must still adhere to:
- MHRA licensing conditions
- General Medical Council (GMC) prescribing standards
- Clinical guidelines regarding appropriate use
- Informed consent and patient safety protocols
Due diligence when seeking private prescription:
✅ Verify prescriber credentials (GMC registration) ✅ Ensure comprehensive medical assessment occurs ✅ Confirm medication source legitimacy (licensed pharmacy) ✅ Understand monitoring protocols and ongoing support ✅ Review terms regarding treatment discontinuation
Medical Tourism and International Access
Some UK residents explore international options:
Considerations:
- Regulatory standards vary significantly between countries
- Medication authenticity and quality cannot be guaranteed
- Legal implications of importing prescription medications
- Absence of ongoing medical supervision
- Insurance and liability concerns
Risks:
- Counterfeit or substandard products
- Inappropriate dosing or contamination
- Lack of medical support for adverse events
- Potential legal consequences
Research-Grade Peptides: A Critical Distinction
It’s essential to distinguish between licensed pharmaceutical tirzepatide and research-grade peptides. Companies like PEPTIDE PRO supply high-purity research compounds for legitimate scientific investigation, clearly labelled “For Research Use Only.”
Key Distinctions:
| Aspect | Pharmaceutical Tirzepatide | Research-Grade Peptides |
|---|---|---|
| Regulatory Status | MHRA-approved medicine | Not approved for human use |
| Intended Use | Clinical treatment under medical supervision | Laboratory research only |
| Quality Standards | GMP pharmaceutical manufacturing | Research-grade purity standards |
| Prescription Required | Yes, from licensed prescriber | No (not for human consumption) |
| Legal Use | Patient treatment | Scientific research applications |
| Medical Oversight | Mandatory clinical supervision | Not applicable (research context) |
Important: Research-grade peptides, including those available through specialized suppliers, are strictly for in vitro research purposes and are not intended, approved, or safe for human consumption. Using research chemicals outside legitimate laboratory settings carries serious health, legal, and ethical risks.
For researchers seeking high-purity compounds for legitimate scientific studies, PEPTIDE PRO’s extensive catalogue provides research-grade materials with appropriate documentation, certificates of analysis, and clear usage restrictions.
Cost-Benefit Analysis: NHS vs Private Access
Understanding the financial and practical implications of different access routes helps inform decision-making regarding Tirzepatide & the NHS: Eligibility, Criteria and Access.
NHS Route: Total Cost of Access
Direct Costs:
- Prescription charges: £9.90 per prescription (England) or free in Scotland, Wales, Northern Ireland
- Typical 3-month supply: £29.70 (or covered by prepayment certificate at £31.25/quarter)
- 12-month treatment cost: £118.80 (or £111.60 with annual prepayment certificate)
Indirect Costs:
- Time investment: Multiple appointments, programme attendance
- Travel to specialist centres (potentially significant for rural patients)
- Opportunity costs of extended waiting periods
Total NHS Patient Cost: £100-150 annually (excluding time and travel)
Private Route: Comprehensive Cost Analysis
Year One Costs:
- Initial consultations and investigations: £500-800
- Medication (12 months): £2,400-4,800
- Follow-up consultations (4-6 visits): £400-800
- Additional monitoring bloods: £200-400
Total Private Year One Cost: £3,500-6,800
Subsequent Years:
- Medication: £2,400-4,800
- Consultations: £300-600
- Monitoring: £150-300
Total Private Ongoing Annual Cost: £2,850-5,700
Value Proposition Comparison
NHS Advantages:
- ✅ Minimal financial burden
- ✅ Comprehensive multidisciplinary support
- ✅ Integration with broader health services
- ✅ Evidence-based protocols and safety monitoring
NHS Disadvantages:
- ❌ Strict eligibility criteria
- ❌ Extended waiting times (6-12+ months)
- ❌ Regional availability variations
- ❌ Limited treatment duration (typically 12-24 months maximum)
- ❌ Mandatory participation in structured programmes
Private Advantages:
- ✅ Rapid access (often within weeks)
- ✅ Flexible eligibility (clinical judgment-based)
- ✅ Extended treatment duration possible
- ✅ Personalized service and convenience
Private Disadvantages:
- ❌ Substantial ongoing financial commitment
- ❌ Variable quality of care between providers
- ❌ Potential for less rigorous screening
- ❌ Limited integration with NHS records
Health Economic Perspective
From a societal perspective, tirzepatide demonstrates cost-effectiveness through:
Direct Healthcare Savings:
- Reduced diabetes medication requirements
- Decreased cardiovascular event rates
- Lower incidence of obesity-related complications
- Reduced need for bariatric surgery
Indirect Economic Benefits:
- Improved work productivity
- Reduced disability and sick leave
- Enhanced quality of life
- Decreased informal care burden
NICE health economic modeling suggests tirzepatide is cost-effective at willingness-to-pay thresholds of £20,000-30,000 per quality-adjusted life year (QALY) for appropriate patient populations.[6]
Future Outlook: Evolving NHS Policy and Access in 2025
The landscape of Tirzepatide & the NHS: Eligibility, Criteria and Access continues to evolve throughout 2025, with several developments likely to influence availability and prescribing patterns.
Anticipated Policy Changes
NICE Guidance Updates:
- Expanded indications under review, potentially including lower BMI thresholds
- Cardiovascular outcome data influencing cost-effectiveness assessments
- Possible extension of treatment duration recommendations based on long-term trial data
NHS England Commissioning:
- Potential standardization of access criteria across ICBs to reduce postcode lottery
- Development of shared care protocols enabling GP prescribing under specialist guidance
- Integration with digital weight management platforms
Supply and Manufacturing Considerations
Global Supply Challenges:
- Unprecedented demand has created periodic supply constraints
- Manufacturer capacity expansion ongoing throughout 2025
- NHS prioritization protocols during shortage periods
Biosimilar Development:
- Patent landscape may enable biosimilar development in coming years
- Potential for reduced costs and improved access if biosimilars emerge
- Regulatory pathways for peptide biosimilars remain complex
Emerging Alternatives and Combination Therapies
Next-Generation Medications:
- Triple agonists (GIP/GLP-1/glucagon) in late-stage development
- Oral GLP-1 formulations advancing through trials
- Longer-acting formulations (monthly or less frequent dosing)
Combination Approaches:
- Tirzepatide plus other anti-obesity medications
- Integration with digital therapeutics and continuous glucose monitoring
- Personalized medicine approaches based on genetic and metabolic profiling
Research Directions
The scientific community continues investigating:
Expanded Indications:
- Non-alcoholic steatohepatitis (NASH)
- Cardiovascular disease prevention
- Chronic kidney disease
- Polycystic ovary syndrome (PCOS)
- Obstructive sleep apnoea
Mechanistic Understanding:
- Optimal GIP/GLP-1 receptor activation ratios
- Central nervous system effects on appetite regulation
- Metabolic adaptations during treatment and post-discontinuation
For those interested in the fundamental science underlying these therapeutic peptides, research institutions utilize high-purity research-grade compounds to investigate mechanisms of action, receptor binding characteristics, and metabolic effects in controlled laboratory settings.
Patient Advocacy and Access Equity
Addressing Health Inequalities:
- Recognition that obesity disproportionately affects socioeconomically disadvantaged populations
- Efforts to ensure equitable access regardless of geography or socioeconomic status
- Cultural sensitivity in treatment approaches for diverse populations
Patient Support Organizations:
- Obesity UK and similar charities advocating for improved access
- Peer support networks for individuals navigating treatment pathways
- Educational initiatives to reduce stigma and improve understanding
Practical Guidance for Patients and Healthcare Providers

For Patients Considering Tirzepatide
Step 1: Assess Your Eligibility
Honestly evaluate whether you meet NHS criteria:
- Calculate your BMI accurately
- Document weight-related health conditions
- Review previous weight loss attempts
- Consider timing and commitment required
Step 2: Engage with Your GP
Prepare for productive consultation:
- Bring weight history documentation
- List previous interventions attempted
- Describe impact of weight on health and quality of life
- Ask about local referral pathways
Step 3: Commit to Lifestyle Modification
Treatment success requires:
- Sustainable dietary changes (not extreme restriction)
- Regular physical activity (150+ minutes weekly)
- Behavioral strategies for long-term maintenance
- Psychological support if needed
Step 4: Set Realistic Expectations
Understand that:
- Access may take 6-12+ months through NHS
- Treatment is time-limited (typically 12-24 months)
- Side effects are common initially
- Weight maintenance post-treatment requires ongoing effort
- Not everyone achieves dramatic results
Step 5: Consider All Options
Evaluate:
- NHS pathway (if eligible)
- Private prescription (if financially viable)
- Alternative evidence-based treatments
- Bariatric surgery (for severe obesity)
For Healthcare Providers
Primary Care Responsibilities:
✅ Accurate Assessment: Calculate BMI correctly, using ethnicity-adjusted thresholds where appropriate
✅ Comprehensive Documentation: Record weight history, previous interventions, comorbidities, and functional impact
✅ Appropriate Referral: Understand local pathways and refer to correct tier based on patient complexity
✅ Realistic Counseling: Set appropriate expectations regarding access timelines, treatment duration, and outcomes
✅ Holistic Approach: Address obesity within broader context of patient’s health, not as isolated problem
Specialist Service Considerations:
✅ Multidisciplinary Assessment: Ensure psychological, nutritional, and medical evaluation
✅ Individualized Treatment Plans: Tailor approach to patient circumstances, preferences, and goals
✅ Rigorous Monitoring: Implement systematic follow-up with clear continuation criteria
✅ Safety Surveillance: Maintain vigilance for adverse effects, particularly pancreatitis and thyroid abnormalities
✅ Transition Planning: Prepare patients for treatment cessation with maintenance strategies
Shared Care Protocols:
Where implemented, shared care between specialists and GPs requires:
- Clear delineation of responsibilities
- Established communication channels
- Defined monitoring schedules
- Protocols for managing complications
- Criteria for re-referral to specialist
Navigating Tirzepatide & the NHS: Eligibility, Criteria and Access in 2025 requires understanding complex eligibility criteria, regional variations, and structured care pathways. While tirzepatide represents a significant therapeutic advancement for obesity and type 2 diabetes, NHS access remains restricted to patients meeting specific BMI thresholds with documented comorbidities, who have engaged with lifestyle interventions and specialist weight management services.
The journey from initial GP consultation to treatment initiation typically spans 6-12 months, with ongoing access dependent on achieving measurable outcomes—minimum 5% weight loss at 3 months and 15% at 12 months for obesity indications. Regional variations across England, Scotland, Wales, and Northern Ireland create disparities in availability, with some areas offering more streamlined access than others.
For those who don’t meet NHS criteria or face prohibitive waiting times, private prescription offers an alternative, albeit at substantial cost (£3,500-6,800 in the first year). However, the distinction between licensed pharmaceutical products and research-grade peptides must be clearly understood—companies like PEPTIDE PRO provide high-purity compounds strictly for legitimate scientific research, not for human consumption.
Key Action Steps
If you’re seeking NHS access:
- Schedule GP consultation to assess eligibility
- Engage fully with Tier 2 lifestyle programmes
- Document all interventions and outcomes
- Request specialist referral if appropriate
- Prepare for 6-12 month timeline
If you’re considering private treatment:
- Research reputable providers with verified credentials
- Budget for £3,000-7,000 annual costs
- Ensure comprehensive medical assessment occurs
- Verify medication source legitimacy
- Establish clear monitoring protocols
If you’re a healthcare provider:
- Familiarize yourself with local ICB policies
- Implement systematic obesity assessment in routine practice
- Understand referral pathways and timelines
- Set realistic patient expectations
- Consider shared care opportunities where available
The evolving landscape of obesity pharmacotherapy promises improved access and outcomes in coming years, with policy developments, supply improvements, and emerging alternatives likely to expand treatment options. However, sustainable weight management ultimately requires comprehensive lifestyle modification supported by appropriate medical intervention—medication alone, whether accessed through NHS or private routes, represents only one component of effective long-term obesity treatment.
For those interested in the broader scientific context of peptide therapeutics, educational resources provide insights into peptide biochemistry, handling protocols, and research applications, though always within the context of legitimate scientific investigation rather than clinical use.
Understanding your options, eligibility, and the realities of both NHS and private access empowers informed decision-making in partnership with qualified healthcare professionals. Whether through public or private healthcare systems, the goal remains the same: sustainable health improvement through evidence-based, medically supervised intervention.
References
[1] Frias JP, et al. “Tirzepatide versus Semaglutide Once Weekly in Patients with Type 2 Diabetes.” New England Journal of Medicine 2021;385:503-515.
[2] Nauck MA, et al. “GIP and GLP-1 receptor agonism in type 2 diabetes and obesity.” Diabetes, Obesity and Metabolism 2023;25(Suppl 1):3-16.
[3] Jastreboff AM, et al. “Tirzepatide Once Weekly for the Treatment of Obesity.” New England Journal of Medicine 2022;387:205-216.
[4] Rosenstock J, et al. “Efficacy and safety of a novel dual GIP and GLP-1 receptor agonist tirzepatide in patients with type 2 diabetes (SURPASS-1): a double-blind, randomised, phase 3 trial.” Lancet 2021;398:143-155.
[5] National Institute for Health and Care Excellence. “Obesity: identification, assessment and management.” Clinical guideline [CG189]. Updated 2023.
[6] National Institute for Health and Care Excellence. “Tirzepatide for treating type 2 diabetes.” Technology appraisal guidance [TA924]. Published September 2023.
