When exploring novel therapeutic peptides in laboratory settings, understanding the intersection of retatrutide pregnancy safety becomes paramount for responsible research protocols. As retatrutide emerges as a triple-agonist peptide targeting GLP-1, GIP, and glucagon receptors, researchers must navigate critical safety considerations that extend beyond standard laboratory procedures—particularly regarding reproductive health implications and contraindications during pregnancy.
This comprehensive guide examines the current scientific understanding of retatrutide in the context of pregnancy, reproductive safety protocols, and essential considerations for research applications. Whether you’re a research professional evaluating peptide safety profiles or seeking to understand the contraindications associated with this compound, this resource provides evidence-based information grounded in current scientific literature and regulatory guidance.
Key Takeaways
- Retatrutide is strictly contraindicated during pregnancy and classified for research use only, not approved for human therapeutic applications
- Preclinical studies indicate potential reproductive toxicity, requiring strict pregnancy prevention protocols in any research involving reproductive-age subjects
- Adequate washout periods (typically 3-5 months based on pharmacokinetics) must be observed before conception attempts in research contexts
- Research-grade retatrutide from reputable suppliers like PEPTIDE PRO ensures quality control and proper labeling for laboratory applications
- Current evidence suggests multi-receptor agonism may impact fetal development, placental function, and maternal metabolic homeostasis
Understanding Retatrutide: Mechanism and Research Applications
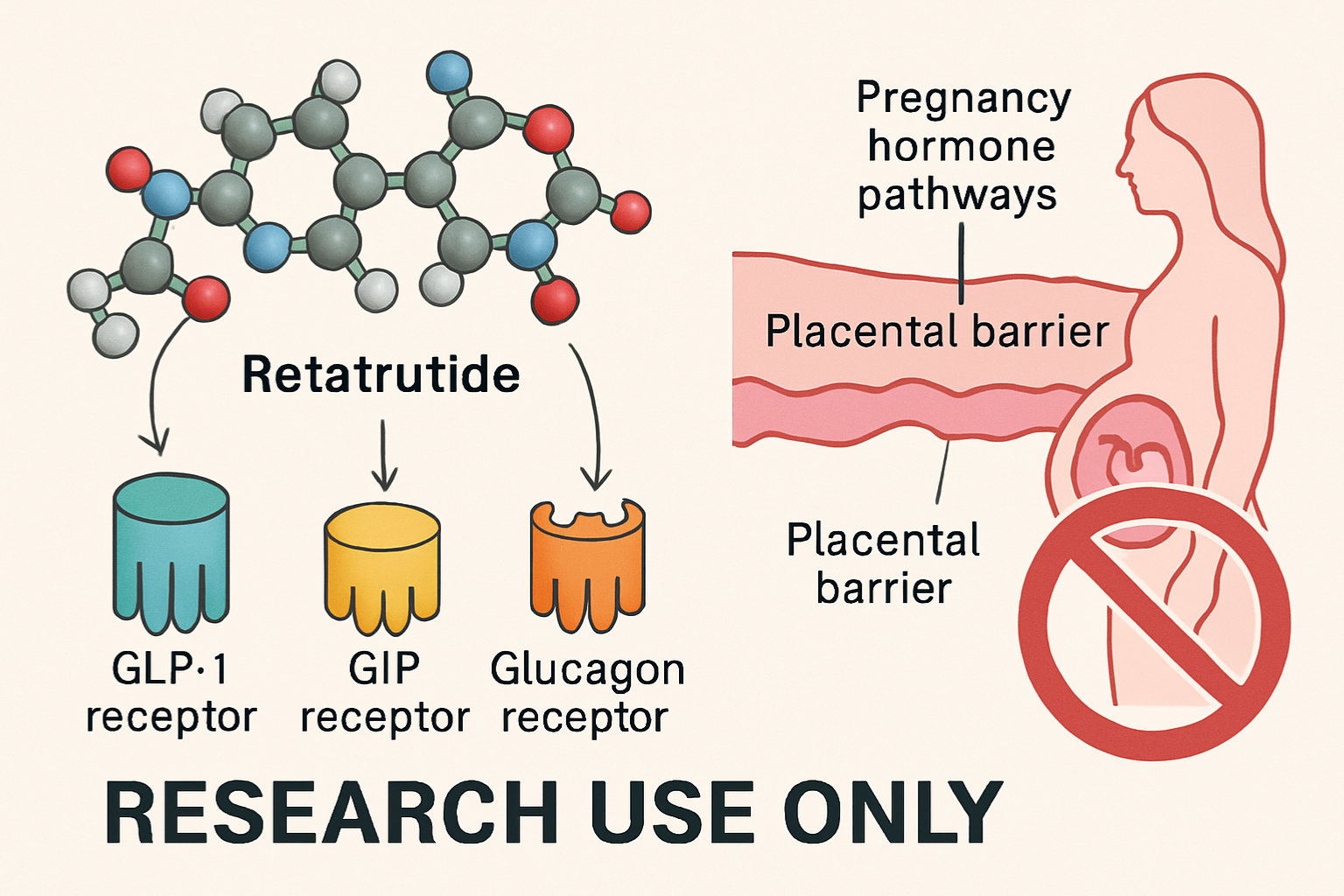
What Is Retatrutide?
Retatrutide represents a novel class of triple-agonist peptides that simultaneously activates three distinct receptor pathways: glucagon-like peptide-1 (GLP-1), glucose-dependent insulinotropic polypeptide (GIP), and glucagon receptors. This multi-targeted approach distinguishes retatrutide from earlier single or dual-agonist compounds, creating unique metabolic effects that have attracted significant research interest.
Key characteristics of retatrutide include:
- Molecular structure: Synthetic peptide analog with modifications enhancing receptor affinity and half-life
- Receptor targeting: Triple-agonist activity across GLP-1R, GIPR, and GCGR
- Pharmacokinetics: Extended half-life allowing for less frequent dosing in research protocols
- Research applications: Metabolic studies, obesity research, diabetes modeling, and energy homeostasis investigations
The compound’s mechanism involves complex signaling cascades affecting glucose metabolism, energy expenditure, appetite regulation, and lipid metabolism—all systems that intersect with pregnancy physiology in potentially significant ways.
Current Research Status and Regulatory Classification
As of 2025, retatrutide remains in investigational stages for potential therapeutic applications, with ongoing clinical trials examining safety and efficacy profiles. However, it’s crucial to emphasize that retatrutide is not approved for human use outside controlled research settings and is explicitly labeled as “For Research Use Only” by responsible suppliers.
Research institutions and laboratories sourcing retatrutide must adhere to strict protocols ensuring:
✅ Proper storage conditions (typically -20°C for lyophilized peptides)
✅ Documented chain of custody and usage logs
✅ Compliance with institutional review board (IRB) guidelines
✅ Clear labeling preventing misuse or unauthorized administration
✅ Quality assurance through certificates of analysis (COAs)
Reputable suppliers like PEPTIDE PRO provide research-grade peptides with comprehensive documentation, ensuring researchers can maintain proper protocols and safety standards.
Retatrutide Pregnancy Contraindications: Why This Matters
Reproductive Toxicity Concerns in Preclinical Studies
The intersection of retatrutide pregnancy safety represents a critical area of concern based on preclinical evidence from animal studies. While comprehensive human pregnancy data remains limited due to ethical constraints, animal reproduction studies have revealed several concerning findings:
Developmental toxicity observations:
| Finding Category | Observed Effects | Significance Level |
|---|---|---|
| Embryo-fetal development | Skeletal abnormalities, reduced fetal weight | High concern |
| Maternal toxicity | Weight loss, metabolic disruption | Moderate-High |
| Placental function | Altered nutrient transfer, reduced placental weight | High concern |
| Post-natal development | Growth retardation, developmental delays | Moderate concern |
| Fertility parameters | Altered estrous cycles, reduced conception rates | Moderate concern |
These findings, while derived from animal models, raise substantial red flags regarding potential human reproductive risks. The multi-receptor agonism characteristic of retatrutide creates complex metabolic alterations that may fundamentally disrupt the delicate hormonal and metabolic balance required for healthy pregnancy.
Mechanisms of Potential Pregnancy Risk
Understanding why retatrutide poses pregnancy risks requires examining the physiological roles of its target receptors during gestation:
🔬 GLP-1 Receptor Activation:
- Modulates insulin secretion and glucose homeostasis
- Affects placental glucose transport mechanisms
- May alter maternal-fetal nutrient partitioning
- Influences pancreatic beta-cell function during pregnancy
🔬 GIP Receptor Activation:
- Regulates lipid metabolism and fat storage
- Affects bone metabolism (relevant for fetal skeletal development)
- Modulates inflammatory responses
- Influences adipocyte differentiation
🔬 Glucagon Receptor Activation:
- Promotes hepatic glucose production
- Affects amino acid metabolism
- Influences ketone body production
- May disrupt maternal metabolic adaptation to pregnancy
The simultaneous activation of these pathways creates metabolic states potentially incompatible with the profound physiological adaptations pregnancy requires. Maternal weight loss, altered glucose dynamics, and disrupted nutrient availability could compromise fetal development at critical stages.
Safety Protocols for Research Involving Retatrutide
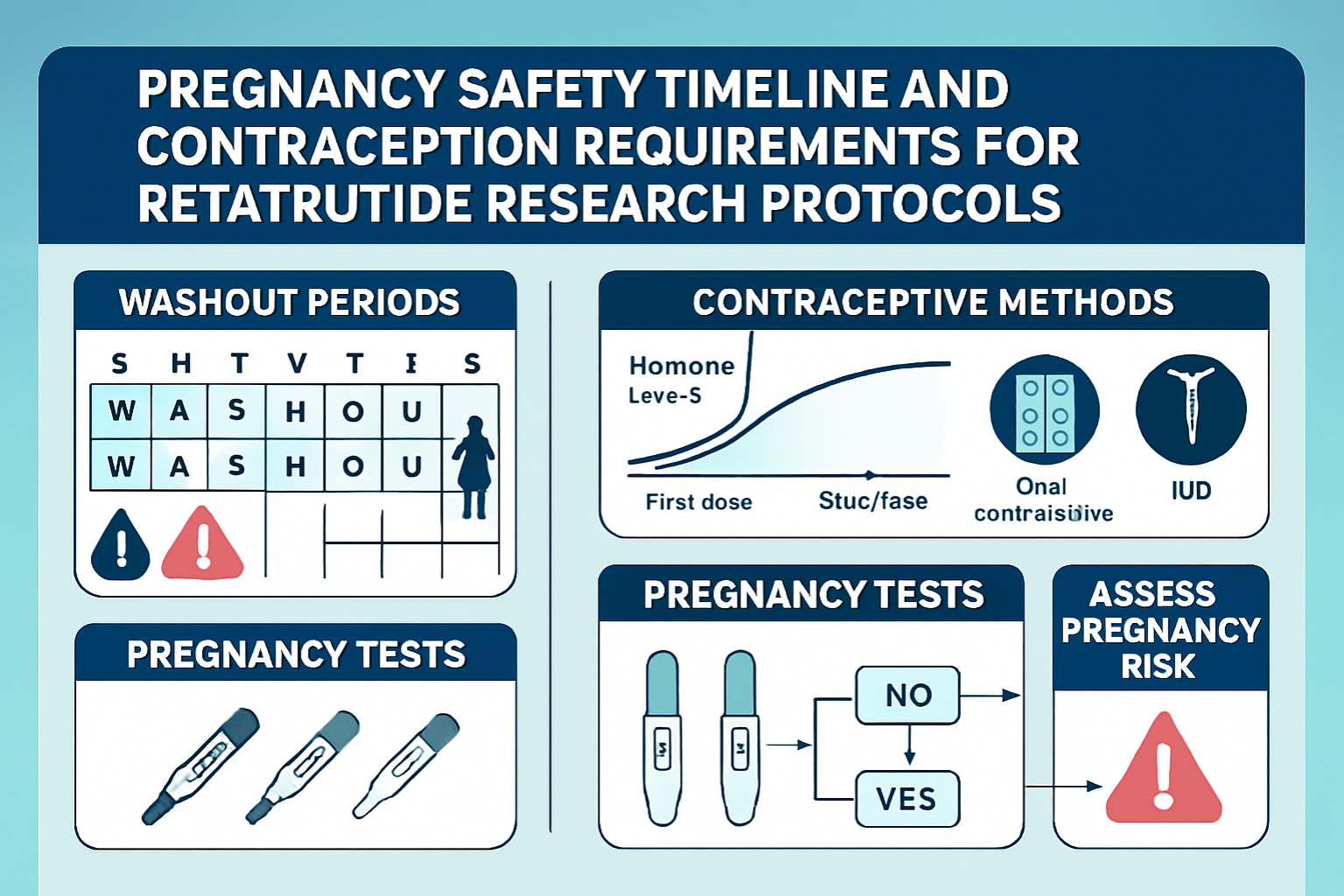
Pregnancy Prevention Requirements in Research Settings
Any research protocol involving retatrutide and participants of reproductive potential must implement rigorous pregnancy prevention measures. These protocols typically mirror pharmaceutical industry standards for teratogenic substances:
Mandatory contraception requirements:
- Dual contraceptive methods (barrier plus hormonal or IUD)
- Pre-study pregnancy testing (serum β-hCG with sensitivity ≤25 mIU/mL)
- Regular pregnancy monitoring throughout study participation
- Post-study contraception continuation during washout period
- Immediate study discontinuation if pregnancy occurs
Research institutions must document comprehensive informed consent processes ensuring participants understand reproductive risks and contraception requirements. The contact information for research peptide suppliers should include safety data sheets and handling protocols addressing these concerns.
Washout Periods and Conception Planning
Given retatrutide’s extended pharmacokinetic profile, adequate washout periods are essential before conception attempts. Current guidance suggests:
⏱️ Recommended Washout Timeline:
- Minimum washout: 3 months after final dose
- Conservative approach: 5 months for enhanced safety margin
- Pharmacokinetic basis: Approximately 5 half-lives for >97% elimination
- Individual considerations: Body composition, dosing history, metabolic factors
During washout periods, continued pregnancy testing and contraception remain essential. Researchers should document:
- Final administration date and dosage
- Contraceptive methods employed during washout
- Pregnancy test results at regular intervals
- Any adverse events or metabolic changes
- Clearance confirmation before conception attempts
These protocols protect both research integrity and participant safety, reflecting the serious nature of retatrutide pregnancy contraindications.
Retatrutide Pregnancy Exposure: Risk Assessment and Management
What Happens If Pregnancy Occurs During Research?
Despite rigorous prevention protocols, unintended pregnancy exposure may occasionally occur in research settings. Immediate response protocols should include:
🚨 Immediate Actions:
- Cease retatrutide administration immediately upon pregnancy confirmation
- Document exposure details: gestational age at exposure, cumulative dosing, duration
- Initiate enhanced monitoring: high-risk obstetric referral, detailed ultrasound assessment
- Report to regulatory authorities: institutional review boards, drug safety databases
- Provide comprehensive counseling: reproductive toxicology consultation, informed decision support
The limited human pregnancy exposure data for retatrutide necessitates individualized risk assessment. Factors influencing risk magnitude include:
- Timing of exposure: First trimester (organogenesis) represents highest risk period
- Dose and duration: Cumulative exposure correlates with potential toxicity
- Maternal metabolic status: Degree of weight loss or metabolic disruption
- Concurrent medications: Polypharmacy may compound risks
Monitoring and Follow-Up Recommendations
Pregnancies with retatrutide exposure require enhanced surveillance throughout gestation and postpartum:
Recommended monitoring protocol:
| Trimester | Monitoring Elements | Frequency |
|---|---|---|
| First | Serial β-hCG, progesterone, detailed anatomy scan | Weekly to bi-weekly |
| Second | Growth ultrasounds, anatomical surveys, glucose tolerance | Every 3-4 weeks |
| Third | Fetal growth monitoring, biophysical profiles, Doppler studies | Every 2-3 weeks |
| Postpartum | Neonatal assessment, developmental screening | Standard plus 6-month follow-up |
Long-term pediatric follow-up may be warranted given potential developmental effects observed in animal studies. Pregnancy registries, when available, provide valuable data for understanding human reproductive outcomes following exposure.
Research-Grade Retatrutide: Quality and Safety Considerations
Sourcing High-Purity Research Peptides
The quality of research-grade retatrutide directly impacts both experimental validity and safety profiles. Researchers must prioritize suppliers demonstrating:
✓ Quality Indicators:
- Purity verification: HPLC analysis showing ≥98% purity
- Certificate of Analysis (COA): Batch-specific documentation
- Proper storage: Temperature-controlled facilities and shipping
- Clear labeling: “For Research Use Only” designation
- Regulatory compliance: Adherence to research chemical regulations
PEPTIDE PRO exemplifies these standards, providing research-grade peptides with comprehensive quality documentation and professional service supporting the scientific community. Their commitment to purity and transparency ensures researchers work with compounds meeting rigorous specifications.
Storage and Handling Protocols
Proper retatrutide handling preserves compound integrity and prevents degradation that could alter safety profiles:
📦 Storage Guidelines:
- Lyophilized peptide: Store at -20°C in original sealed pen peptides
- Reconstituted solution: Refrigerate at 2-8°C, use within recommended timeframe
- Light protection: Store in amber pen peptides or light-protected containers
- Contamination prevention: Aseptic technique for all handling
- Documentation: Maintain logs of storage conditions and stability
Degraded or improperly stored peptides may exhibit altered biological activity, potentially creating unpredictable effects. Research protocols should include stability testing and regular quality verification to ensure compound integrity throughout studies.
Comparing Retatrutide to Other Metabolic Peptides in Pregnancy Context
GLP-1 Agonists and Reproductive Safety
Retatrutide’s GLP-1 receptor activity invites comparison with established GLP-1 agonists regarding pregnancy safety:
Comparative pregnancy data:
- Semaglutide: Limited human data; animal studies show developmental toxicity; contraindicated in pregnancy
- Liraglutide: Some inadvertent pregnancy exposures documented; generally discontinued when pregnancy detected
- Tirzepatide (GLP-1/GIP dual agonist): Similar preclinical concerns; pregnancy contraindication
- Retatrutide (triple agonist): Most complex mechanism; theoretical highest risk due to multi-pathway effects
Researchers working with various metabolic peptides can explore options like semaglutide research compounds or tirzepatide formulations while maintaining awareness that all these compounds share pregnancy contraindications due to similar mechanisms.
Novel Weight Management Peptides: Shared Safety Concerns
The emerging class of multi-agonist weight management peptides presents consistent reproductive safety patterns:
Common safety themes:
�
� Maternal weight loss: Potentially compromises fetal nutrition �
� Metabolic reprogramming: May disrupt pregnancy adaptations �
� Appetite suppression: Could lead to inadequate nutrient intake �
� Glucose dynamics: Alters maternal-fetal glucose transfer �
� Hormonal effects: May influence reproductive hormone balance
These shared concerns underscore why retatrutide pregnancy contraindications align with broader class effects. Researchers should apply similar safety protocols across related compounds, including newer agents like mazdutide or survodutide.
Future Research Directions and Knowledge Gaps
Areas Requiring Further Investigation
Despite growing interest in retatrutide, significant knowledge gaps remain regarding reproductive safety:
Priority research questions:
- Pharmacokinetic studies: Precise half-life determination in diverse populations
- Placental transfer: Does retatrutide cross the placental barrier?
- Lactation safety: Presence in breast milk and neonatal exposure risk
- Paternal exposure: Effects on sperm quality and male fertility
- Long-term developmental outcomes: Childhood and adolescent health following in utero exposure
- Mechanism clarification: Which receptor pathway(s) drive reproductive toxicity?
Addressing these questions requires carefully designed preclinical studies, pregnancy exposure registries, and long-term epidemiological follow-up—all conducted with rigorous ethical oversight.
Evolving Regulatory Landscape
As retatrutide progresses through development stages, regulatory frameworks will evolve. Researchers should monitor:
- FDA pregnancy category updates (if therapeutic approval pursued)
- International regulatory guidance from EMA, MHRA, and other authorities
- Professional society recommendations from endocrinology and reproductive medicine organizations
- Institutional policy changes regarding research peptide handling
Staying informed ensures research protocols remain compliant with current best practices and regulatory requirements.
Practical Guidance for Research Professionals
Implementing Comprehensive Safety Protocols
Research institutions incorporating retatrutide into study protocols should develop comprehensive standard operating procedures (SOPs) addressing:
📋 Essential SOP Components:
1. Participant Screening:
- Detailed reproductive history
- Pregnancy intention assessment
- Contraception counseling and documentation
- Baseline pregnancy testing
2. Ongoing Monitoring:
- Regular pregnancy testing schedules
- Contraception compliance verification
- Adverse event reporting systems
- Metabolic parameter tracking
3. Emergency Protocols:
- Immediate response to positive pregnancy test
- Consultation pathways (reproductive toxicology, high-risk obstetrics)
- Documentation requirements
- Regulatory reporting procedures
4. Staff Training:
- Recognition of pregnancy signs/symptoms
- Proper handling and administration techniques
- Emergency response protocols
- Ethical considerations and informed consent
Documentation and Regulatory Compliance
Meticulous documentation protects both participants and institutions:
Required documentation elements:
✍️ Informed consent forms explicitly addressing pregnancy risks
✍️ Contraception method documentation and compliance logs
✍️ Pregnancy test results with dates and sensitivity specifications
✍️ Adverse event reports following standardized formats
✍️ Protocol deviation documentation if pregnancy occurs
✍️ Communication logs with regulatory authorities
These records demonstrate institutional commitment to participant safety and regulatory compliance, essential for maintaining research integrity and ethical standards.
Alternative Research Approaches and Considerations
In Vitro and Ex Vivo Models
Given the significant retatrutide pregnancy safety concerns, alternative research models offer valuable approaches for investigating reproductive effects without human exposure:
🔬 Alternative Model Systems:
- Placental perfusion models: Ex vivo human placenta studies examining transfer kinetics
- Embryoid body cultures: Stem cell-derived models for developmental toxicity screening
- Organoid systems: 3D tissue cultures mimicking reproductive organs
- Animal models: Carefully designed studies with appropriate species selection
- Computational modeling: In silico predictions of pharmacokinetic and toxicological parameters
These approaches can provide mechanistic insights while minimizing human reproductive risk, advancing scientific understanding without compromising safety.
Responsible Research Peptide Sourcing
Researchers committed to ethical and safe peptide research should partner with suppliers demonstrating:
Supplier evaluation criteria:
| Criterion | Importance | Verification Method |
|---|---|---|
| Quality documentation | Critical | Request batch-specific COAs |
| Regulatory compliance | Critical | Verify “Research Use Only” labeling |
| Storage/shipping protocols | High | Review temperature monitoring procedures |
| Customer support | High | Assess responsiveness to technical inquiries |
| Transparency | High | Evaluate willingness to provide detailed information |
PEPTIDE PRO exemplifies these standards, offering high-purity research peptides with comprehensive support for the scientific community. Their commitment to quality and transparency supports responsible research practices.
Clinical Considerations Beyond Pregnancy
Reproductive Health Across the Lifespan
While pregnancy represents the most acute reproductive concern, retatrutide research must consider broader reproductive health implications:
🔄 Lifecycle Reproductive Considerations:
Pre-conception period:
- Effects on ovarian function and menstrual regularity
- Impact on fertility parameters (ovarian reserve, ovulation)
- Male reproductive health (spermatogenesis, testosterone levels)
- Optimal washout periods before conception attempts
Lactation period:
- Unknown excretion in breast milk
- Potential effects on milk production
- Neonatal exposure risks through breastfeeding
- Recommendations for breastfeeding mothers in research settings
Perimenopause/menopause:
- Interactions with hormone replacement therapy
- Effects on bone density (relevant given GIP receptor involvement)
- Metabolic changes during reproductive transition
Comprehensive reproductive health assessment should extend beyond pregnancy alone, encompassing the full spectrum of reproductive life stages.
Long-Term Metabolic Effects and Fertility
The metabolic reprogramming induced by retatrutide may have lasting implications for reproductive function:
Potential long-term effects:
- Hypothalamic-pituitary-gonadal axis: Disruption of reproductive hormone regulation
- Body composition changes: Alterations in fat distribution affecting hormone production
- Insulin sensitivity: Improvements potentially benefiting polycystic ovary syndrome (PCOS)
- Weight cycling: Repeated weight loss/regain cycles affecting fertility
These complex interactions require longitudinal research examining reproductive outcomes in individuals with prior retatrutide exposure, even outside pregnancy contexts.
Ethical Considerations in Retatrutide Research
Informed Consent and Reproductive Autonomy
Research involving reproductive-age individuals and compounds with pregnancy contraindications raises profound ethical considerations:
⚖️ Ethical Principles:
1. Autonomy: Participants must receive comprehensive information enabling informed reproductive decisions
2. Beneficence: Research design should maximize knowledge gain while minimizing reproductive risks
3. Justice: Inclusion/exclusion criteria should not unfairly burden or exclude specific populations
4. Non-maleficence: Protocols must prevent foreseeable reproductive harm through robust safety measures
Informed consent processes should explicitly address:
- Known and theoretical pregnancy risks
- Contraception requirements and compliance monitoring
- Washout period recommendations
- Immediate discontinuation protocols if pregnancy occurs
- Long-term reproductive health unknowns
Balancing Scientific Progress and Safety
The scientific community faces ongoing tension between advancing metabolic research and protecting reproductive health:
“The promise of novel therapeutic approaches must never compromise fundamental safety principles, particularly regarding vulnerable populations and reproductive health. Rigorous safety protocols aren’t obstacles to research—they’re essential foundations enabling ethical scientific progress.”
This balance requires:
✓ Conservative risk assessment when data are limited
✓ Transparent communication of uncertainties
✓ Prioritization of participant welfare over research convenience
✓ Continuous protocol refinement as new safety data emerge
✓ Institutional commitment to ethical oversight
Resources for Researchers and Institutions
Professional Organizations and Guidelines
Researchers working with retatrutide should consult authoritative resources:
📚 Key Resources:
- Teratology Information Services: Consultation for pregnancy exposure cases
- Institutional Review Boards: Local ethical oversight and protocol approval
- Regulatory Authorities: FDA, EMA, MHRA guidance documents
- Professional Societies: Endocrine Society, Society for Reproductive Investigation
- Pregnancy Registries: Enrollment pathways for exposed pregnancies
Continuing Education and Training
Staying current with evolving safety data requires ongoing professional development:
Recommended training areas:
🎓 Reproductive toxicology fundamentals
🎓 Peptide pharmacology and metabolism
🎓 Research ethics and informed consent
🎓 Adverse event recognition and reporting
🎓 Regulatory compliance requirements
Many institutions offer specialized training programs addressing research peptide safety, and suppliers like PEPTIDE PRO may provide educational resources supporting responsible research practices.
Retatrutide Pregnancy: Summary of Critical Points
Key Safety Messages
As this comprehensive review demonstrates, retatrutide pregnancy represents a critical contraindication requiring rigorous attention:
**
�
� Essential Safety Points:**
- Absolute contraindication: Retatrutide must not be used during pregnancy
- Preclinical evidence: Animal studies demonstrate reproductive toxicity
- Multi-pathway effects: Triple-agonist mechanism creates complex metabolic disruptions
- Prevention protocols: Dual contraception and regular pregnancy testing mandatory
- Washout periods: Minimum 3-5 months before conception attempts
- Research-only status: Not approved for human therapeutic use
- Quality sourcing: Use only research-grade peptides from reputable suppliers
- Documentation: Comprehensive records essential for safety and compliance
Risk Mitigation Strategies
Institutions and researchers can minimize reproductive risks through:
Comprehensive risk mitigation approach:
| Strategy Level | Specific Actions | Responsibility |
|---|---|---|
| Institutional | Develop robust SOPs, provide ethics training | Research administration |
| Protocol design | Include rigorous exclusion criteria, monitoring plans | Principal investigators |
| Participant screening | Thorough reproductive history, counseling | Research coordinators |
| Ongoing monitoring | Regular pregnancy testing, contraception verification | Clinical research staff |
| Emergency response | Immediate protocols for positive pregnancy tests | All research personnel |
| Supplier partnership | Source from quality-verified vendors | Procurement departments |
This multi-level approach creates redundant safety systems protecting participants and research integrity.
Conclusion: Advancing Research Responsibly
The emergence of retatrutide as a novel triple-agonist peptide presents exciting research opportunities in metabolic science, obesity research, and energy homeostasis investigation. However, these opportunities must be pursued within frameworks prioritizing participant safety—particularly regarding retatrutide pregnancy contraindications and reproductive health protection.
Key takeaways for responsible research:
✅ Recognize absolute pregnancy contraindication based on preclinical reproductive toxicity data
✅ Implement comprehensive prevention protocols including dual contraception and regular testing
✅ Source research-grade peptides from reputable suppliers like PEPTIDE PRO ensuring quality and proper labeling
✅ Maintain meticulous documentation supporting regulatory compliance and participant safety
✅ Plan adequate washout periods (3-5 months) before conception attempts
✅ Develop emergency protocols for unintended pregnancy exposure
✅ Stay informed about evolving safety data and regulatory guidance
Next Steps for Research Professionals
Researchers incorporating retatrutide into study protocols should:
- Review institutional policies regarding reproductive safety in research
- Develop comprehensive SOPs addressing pregnancy prevention and monitoring
- Establish supplier relationships with quality-verified peptide sources
- Train research staff on safety protocols and emergency procedures
- Implement robust informed consent processes addressing reproductive risks
- Create monitoring systems ensuring protocol compliance
- Establish consultation pathways with reproductive toxicology and high-risk obstetrics
Commitment to Scientific Excellence and Safety
The research community’s commitment to both scientific advancement and participant welfare defines ethical research practice. Understanding retatrutide pregnancy contraindications and implementing rigorous safety protocols demonstrates this dual commitment.
For researchers seeking high-purity, research-grade peptides with comprehensive quality documentation, PEPTIDE PRO offers an extensive catalogue of compounds suitable for diverse research applications. Their commitment to purity, transparency, and professional service supports the scientific community in conducting responsible, ethically sound research.
Research Use Only: All information in this article pertains to research applications only. Retatrutide is not approved for human therapeutic use and should never be used during pregnancy or by individuals attempting conception. Always consult institutional review boards and regulatory authorities when designing research protocols involving reproductive-age participants.
By maintaining unwavering commitment to safety protocols, transparent communication of risks, and ethical research practices, the scientific community can advance metabolic research while protecting the reproductive health of research participants—ensuring that scientific progress and human welfare advance together.
