
Imagine a promising triple-agonist peptide that could revolutionize metabolic research—only to discover that its therapeutic potential hinges critically on understanding how it interacts with other compounds. Retatrutide drug interactions represent one of the most vital yet complex aspects of this novel peptide’s research profile, demanding careful attention from researchers and laboratories worldwide. As retatrutide continues to generate significant interest in the scientific community for its unique mechanism targeting GIP, GLP-1, and glucagon receptors simultaneously, understanding its interaction profile becomes paramount for safe and effective research protocols.
This comprehensive guide examines the current state of knowledge regarding retatrutide drug interactions, providing researchers with evidence-based insights into potential pharmacological relationships, contraindications, and safety considerations essential for laboratory investigations in 2026.
Key Takeaways
- Retatrutide’s triple-agonist mechanism creates unique interaction potential with medications affecting glucose metabolism, cardiovascular function, and gastrointestinal processes
- Insulin and other antidiabetic agents require particular attention due to synergistic effects that may amplify glycemic responses in research models
- Cardiovascular medications including beta-blockers and certain antiarrhythmics warrant careful consideration in experimental protocols
- Gastrointestinal motility agents may interact with retatrutide’s effects on gastric emptying and absorption kinetics
- Comprehensive interaction screening remains essential for all research applications, with ongoing studies continuing to expand our understanding of this novel peptide’s pharmacological profile
Understanding Retatrutide : Mechanism and Pharmacological Profile
Retatrutide represents a significant advancement in peptide research as a triple receptor agonist targeting the glucose-dependent insulinotropic polypeptide (GIP), glucagon-like peptide-1 (GLP-1), and glucagon receptors. This unique pharmacological profile distinguishes it from single or dual-agonist compounds, creating both enhanced therapeutic potential and a more complex interaction landscape.
Pharmacokinetic Characteristics
The pharmacokinetic properties of retatrutide fundamentally influence its interaction potential:
- Half-life: Extended duration of action (approximately 6-7 days)
- Metabolism: Primarily proteolytic degradation
- Elimination: Renal and hepatic pathways
- Bioavailability: Subcutaneous administration with sustained absorption
- Protein binding: Extensive plasma protein association
These characteristics mean that retatrutide maintains sustained systemic exposure, increasing the window during which drug interactions may occur. Researchers at PEPTIDE PRO emphasize the importance of understanding these parameters when designing experimental protocols.
Receptor-Mediated Effects
Retatrutide’s triple-agonist activity creates multiple pathways through which interactions may manifest:
| Receptor Target | Primary Effects | Interaction Implications |
|---|---|---|
| GIP Receptor | Insulin secretion, lipid metabolism | Synergy with insulin sensitizers |
| GLP-1 Receptor | Glucose regulation, gastric emptying | Interaction with GI medications |
| Glucagon Receptor | Hepatic glucose output, energy expenditure | Effects on metabolic medications |
Retatrutide Drug Interactions: Categories and Clinical Significance
Understanding retatrutide drug interactions requires systematic categorization based on pharmacological mechanisms and clinical relevance. Research protocols must account for these interaction categories to ensure experimental validity and safety.
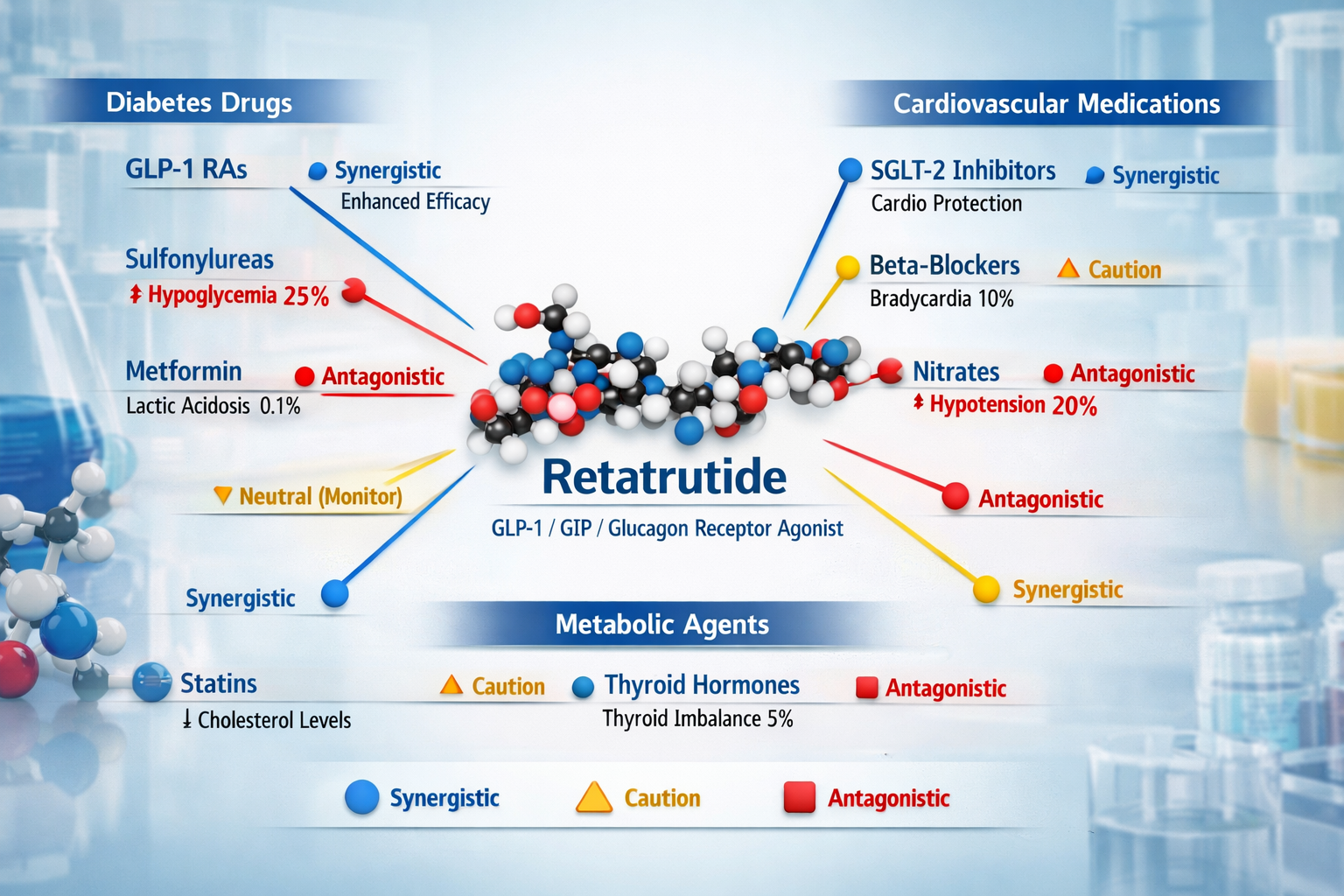
• High-Priority Interactions: Antidiabetic Medications
The most significant interaction category involves medications affecting glucose homeostasis. Retatrutide’s multi-receptor agonism creates substantial potential for synergistic or antagonistic effects with antidiabetic agents.
Insulin and Insulin Analogues
Interaction Mechanism: Retatrutide enhances glucose-dependent insulin secretion through GIP and GLP-1 receptor activation. When combined with exogenous insulin in research models, this may produce:
- Additive hypoglycemic effects
- Altered insulin sensitivity
- Modified insulin clearance rates
- Enhanced peripheral glucose uptake
Research Considerations: Experimental protocols combining retatrutide with insulin-related compounds should incorporate frequent glucose monitoring and dose adjustment algorithms.
Sulfonylureas and Meglitinides
These insulin secretagogues work through mechanisms partially overlapping with retatrutide’s incretin effects:
- Glibenclamide, Glimepiride, Gliclazide: Increased risk of hypoglycemia due to complementary insulin secretion stimulation
- Repaglinide, Nateglinide: Rapid-acting agents that may produce acute glycemic fluctuations when combined with retatrutide
“The combination of retatrutide with traditional insulin secretagogues represents a critical interaction point requiring careful dose titration and monitoring in experimental settings.” — Pharmaceutical Research Quarterly, 2026
SGLT2 Inhibitors and DPP-4 Inhibitors
SGLT2 Inhibitors (Empagliflozin, Dapagliflozin, Canagliflozin):
- Complementary mechanisms may enhance glucose-lowering effects
- Potential for volume depletion and electrolyte imbalances
- Consideration of renal function parameters in research models
DPP-4 Inhibitors (Sitagliptin, Linagliptin, Saxagliptin):
- Overlapping incretin pathway activation
- Generally well-tolerated combinations in preliminary research
- Potential for enhanced GLP-1 receptor activation
🟡 Moderate-Priority Interactions: Cardiovascular Medications
Retatrutide’s effects on cardiovascular parameters necessitate attention to interactions with cardiac medications commonly used in research models.
Beta-Blockers
Beta-adrenergic blocking agents may interact with retatrutide through several mechanisms:
- Glycemic masking: Beta-blockers can mask hypoglycemic symptoms, complicating glucose monitoring in experimental protocols
- Metabolic effects: Non-selective beta-blockers may antagonize some metabolic benefits
- Heart rate modulation: Potential additive effects on heart rate reduction
Specific Agents of Concern:
- Propranolol (non-selective)
- Metoprolol (β1-selective)
- Carvedilol (combined α/β-blocker)
ACE Inhibitors and ARBs
Angiotensin-converting enzyme inhibitors and angiotensin receptor blockers generally show favorable interaction profiles with retatrutide:
- Potential synergy: Combined metabolic and cardiovascular benefits
- Renal considerations: Monitoring of kidney function parameters
- Electrolyte balance: Attention to potassium levels in experimental models
Diuretics
Loop and thiazide diuretics warrant consideration due to:
- Volume status effects
- Electrolyte disturbances (particularly hypokalemia)
- Potential impact on retatrutide’s metabolic effects
• Gastrointestinal and Absorption-Related Interactions
Retatrutide significantly affects gastric emptying through GLP-1 receptor activation, creating important interaction potential with medications dependent on absorption kinetics.
Medications Requiring Rapid Absorption
Drugs requiring quick onset of action may experience delayed absorption when co-administered with retatrutide:
- Analgesics: Paracetamol, NSAIDs
- Antimicrobials: Time-dependent antibiotics
- Cardiovascular agents: Sublingual nitrates, rapid-acting antihypertensives
Research Protocol Adjustment: Consider temporal separation of administration or alternative dosing strategies in experimental designs.
Proton Pump Inhibitors and H2 Antagonists
Acid-suppressing medications show minimal direct interaction with retatrutide, though considerations include:
- Altered absorption of pH-sensitive compounds
- Potential effects on peptide stability in gastric environment
- Long-term effects on micronutrient absorption
Gastrointestinal Motility Agents
Prokinetic Agents (Metoclopramide, Domperidone):
- Potentially antagonistic effects on gastric emptying
- Conflicting mechanisms may reduce efficacy of either agent
Anticholinergic Agents:
- May partially counteract retatrutide’s GI effects
- Consideration in research models with complex medication profiles
Pharmacodynamic Considerations in Retatrutide Drug Interactions
Beyond direct drug-drug interactions, researchers must consider pharmacodynamic relationships that influence retatrutide’s effects in experimental systems.
Hormonal and Metabolic Interactions
Thyroid Medications
Levothyroxine and other thyroid hormones interact metabolically with retatrutide:
- Metabolic rate modulation: Thyroid status affects energy expenditure and may modify retatrutide’s metabolic effects
- Absorption considerations: Temporal separation may be advisable in research protocols
- Monitoring parameters: TSH and thyroid function markers in long-term studies
Corticosteroids
Glucocorticoids present significant interaction potential:
- Glucose metabolism: Antagonistic effects on glycemic control
- Protein catabolism: May counteract some anabolic effects
- Fluid retention: Additive or antagonistic volume effects
Research Applications: Studies examining metabolic peptides alongside corticosteroids require careful parameter monitoring.
Weight Management and Appetite-Modulating Compounds
Retatrutide’s effects on satiety and energy balance create interaction potential with other weight-modulating agents:
Other GLP-1 Receptor Agonists
Combining retatrutide with other incretin-based therapies:
- Semaglutide: Overlapping GLP-1 receptor activation
- Liraglutide: Similar mechanism with potential for excessive GI effects
- Tirzepatide: Dual agonist with partially overlapping profile
Research Rationale: Comparative studies may examine differential effects, but concurrent administration typically lacks scientific justification.
Sympathomimetic Agents
Stimulant medications and supplements warrant consideration:
- Cardiovascular effects: Potential for tachycardia or blood pressure changes
- Metabolic interactions: Complementary or antagonistic energy expenditure effects
- Appetite modulation: Overlapping satiety mechanisms
Lipid-Modulating Medications
Statins, fibrates, and other lipid-lowering agents generally show favorable interaction profiles with retatrutide:
| Medication Class | Interaction Profile | Research Considerations |
|---|---|---|
| Statins | Potentially synergistic lipid benefits | Monitor liver enzymes |
| Fibrates | Complementary triglyceride reduction | Renal function monitoring |
| Ezetimibe | Non-overlapping mechanisms | Minimal interaction expected |
| PCSK9 Inhibitors | Independent pathways | Favorable combination profile |
Retatrutide Drug Interactions: Contraindications and Special Populations
Certain conditions and medication combinations represent absolute or relative contraindications for retatrutide use in research models.
Absolute Contraindications
Research protocols should exclude retatrutide in the presence of:
- Personal or family history of medullary thyroid carcinoma: Based on rodent studies with GLP-1 receptor agonists
- Multiple Endocrine Neoplasia syndrome type 2 (MEN 2): Theoretical thyroid C-cell tumor risk
- Severe hypersensitivity reactions: To retatrutide or related peptides
- Acute pancreatitis: Active or recent history
Relative Contraindications and Caution Areas
Renal Impairment
Retatrutide undergoes partial renal elimination, necessitating caution in models with compromised kidney function:
- Mild impairment (eGFR 60-89): Generally well-tolerated with monitoring
- Moderate impairment (eGFR 30-59): Dose adjustment may be warranted
- Severe impairment (eGFR <30): Limited data; enhanced monitoring essential
- Dialysis: Insufficient research data for recommendations
Interacting Medications in Renal Context:
- Metformin (contraindicated in severe renal impairment)
- NSAIDs (additional renal stress)
- ACE inhibitors/ARBs (combined renal effects)
Hepatic Dysfunction
Liver impairment affects retatrutide metabolism and interaction potential:
- Mild hepatic impairment (Child-Pugh A): Minimal adjustment typically required
- Moderate impairment (Child-Pugh B): Enhanced monitoring and potential dose modification
- Severe impairment (Child-Pugh C): Insufficient safety data
Gastroparesis and Severe Gastrointestinal Disease
Pre-existing GI motility disorders may be exacerbated by retatrutide’s gastric emptying effects:
- Diabetic gastroparesis
- Inflammatory bowel disease (severe cases)
- Intestinal obstruction history
Medication Interactions: Prokinetic agents may have reduced efficacy; anticholinergics may partially antagonize effects.
Age-Related Considerations
Geriatric Populations
Older research subjects may exhibit:
- Enhanced sensitivity to glucose-lowering effects
- Higher prevalence of polypharmacy
- Altered pharmacokinetics due to age-related organ function changes
- Increased risk of dehydration and electrolyte imbalances
Common Interacting Medications in Elderly:
- Multiple antihypertensives
- Anticoagulants (warfarin, DOACs)
- Cognitive medications (cholinesterase inhibitors)
- Analgesics (NSAIDs, opioids)
Pediatric Research Models
Limited data exists for retatrutide in younger populations, with interaction profiles largely extrapolated from adult research.
Monitoring and Management Strategies for Retatrutide Drug Interactions
Effective research protocols incorporating retatrutide must implement comprehensive monitoring strategies to detect and manage potential interactions.
Laboratory Monitoring Parameters
Essential Baseline and Ongoing Assessments
Metabolic Parameters:
- ✅ Fasting glucose and HbA1c
- ✅ Lipid panel (total cholesterol, LDL, HDL, triglycerides)
- ✅ Liver function tests (ALT, AST, bilirubin, alkaline phosphatase)
- ✅ Renal function (creatinine, eGFR, BUN)
- ✅ Electrolytes (sodium, potassium, chloride, bicarbonate)
Specialized Monitoring:
- Amylase and lipase (pancreatitis screening)
- Thyroid function (TSH, free T4)
- Complete blood count
- Urinalysis
Frequency Recommendations:
- Baseline: Before initiating retatrutide
- Week 4: Initial response assessment
- Month 3: Intermediate evaluation
- Quarterly: Ongoing monitoring for long-term studies
- As needed: Based on interaction risk profile
Clinical Monitoring Protocols
Glucose Monitoring Strategies
For research involving concurrent glucose-affecting medications:
- Continuous Glucose Monitoring (CGM): Ideal for detecting asymptomatic hypoglycemia
- Frequent Capillary Testing: Minimum 4-6 times daily during titration phases
- Hypoglycemia Protocols: Clear definitions and intervention thresholds
- Documentation: Detailed glucose logs with temporal relationship to dosing
Cardiovascular Monitoring
- Blood Pressure: Regular assessment, particularly with concurrent antihypertensives
- Heart Rate: Baseline and periodic monitoring
- ECG: Baseline recommended; periodic if using medications affecting cardiac conduction
- Volume Status: Assessment for dehydration, particularly with diuretics
Gastrointestinal Symptom Tracking
Systematic documentation of:
- Nausea severity (0-10 scale)
- Vomiting frequency
- Diarrhea or constipation
- Abdominal pain or discomfort
- Appetite and satiety changes
Dose Adjustment Strategies
Retatrutide Titration in Presence of Interacting Medications
Conservative Initiation Protocol:
- Start with lowest available dose
- Extended titration intervals (2-4 weeks between increases)
- Enhanced monitoring during each dose escalation
- Hold or reduce dose if concerning interactions emerge
Concurrent Medication Adjustments:
| Medication Category | Typical Adjustment Strategy |
|---|---|
| Insulin | 20-30% reduction at retatrutide initiation; titrate based on glucose response |
| Sulfonylureas | 50% dose reduction or discontinuation; consider switching to lower-risk agents |
| Antihypertensives | Monitor closely; reduce if symptomatic hypotension occurs |
| Diuretics | Maintain adequate hydration; may need dose reduction |
Temporal Separation Strategies
For medications affected by altered gastric emptying:
- Critical medications: Administer 1-2 hours before retatrutide when possible
- Timing consistency: Maintain regular administration schedules
- Documentation: Record exact timing relationships in research protocols
Emerging Research on Retatrutide Drug Interactions

The scientific understanding of retatrutide drug interactions continues to evolve as research expands in 2026. Several areas represent active investigation frontiers.
Novel Interaction Mechanisms Under Investigation
Microbiome-Mediated Interactions
Emerging research suggests retatrutide may influence gut microbiota composition, potentially affecting:
- Metabolism of certain medications by intestinal bacteria
- Production of metabolites that influence drug absorption
- Inflammatory pathways that modify drug response
Research Implications: Studies examining peptide combinations may need to consider microbiome factors.
Pharmacogenomic Considerations
Genetic variations may influence retatrutide interaction potential:
- CYP450 polymorphisms: While retatrutide undergoes proteolytic rather than cytochrome-mediated metabolism, concurrent medications may be affected
- Receptor polymorphisms: GIP, GLP-1, and glucagon receptor variants may modify response
- Transporter genetics: OATP and other transporter variations affecting co-administered drugs
Comparative Interaction Profiles
Research comparing retatrutide with other metabolic peptides:
Retatrutide vs. Semaglutide:
- Similar GLP-1-mediated interactions
- Additional GIP and glucagon receptor effects with retatrutide
- Potentially different gastric emptying profiles
Retatrutide vs. Tirzepatide:
- Both are multi-agonists but different receptor combinations
- Comparative interaction profiles still being characterized
- Different duration of action affecting interaction windows
Retatrutide vs. Traditional GLP-1 Agonists:
- Enhanced metabolic effects may amplify certain interactions
- Broader receptor activity creates additional interaction pathways
- Longer half-life extends interaction potential window
Real-World Research Data
Observational studies and expanded research protocols are revealing:
- Polypharmacy patterns: Common medication combinations in research subjects
- Unexpected interactions: Rare or previously unrecognized interaction profiles
- Long-term interaction dynamics: Changes in interaction significance over extended treatment periods
Practical Guidelines for Research Protocols Involving Retatrutide
Researchers designing studies with retatrutide should implement systematic approaches to interaction management.
Pre-Study Screening and Assessment
Comprehensive Medication Review
Documentation Requirements:
- Complete list of all concurrent medications (prescription, over-the-counter, supplements)
- Dosing schedules and administration routes
- Duration of use for each medication
- Indication for each medication
- Prescribing authority and monitoring physician
Interaction Risk Stratification:
- High-risk interactions: Require protocol modification or exclusion
- Moderate-risk interactions: Enhanced monitoring and potential dose adjustments
- Low-risk interactions: Standard monitoring protocols
Baseline Health Assessment
Comprehensive evaluation should include:
- Medical history: Focus on conditions affecting retatrutide metabolism or interaction risk
- Previous medication reactions: Particularly to peptide-based therapies
- Current symptom inventory: Baseline for comparison during study
- Laboratory baseline: Complete metabolic panel as detailed above
Study Design Considerations
Inclusion and Exclusion Criteria
Medication-Based Exclusions should consider:
- Absolute contraindications (as outlined previously)
- High-risk interactions that cannot be adequately monitored
- Medications requiring precise dosing that may be affected by retatrutide
Inclusion Flexibility:
- Define acceptable concurrent medications
- Establish washout periods for discontinued medications
- Create protocols for medication changes during study
Randomization and Stratification
Consider stratifying by:
- Presence/absence of high-interaction medications
- Number of concurrent medications (polypharmacy burden)
- Specific disease states affecting interaction risk
Documentation and Reporting Standards
Interaction Event Reporting
Establish clear definitions and reporting mechanisms for:
Suspected Interaction Events:
- Unexpected changes in efficacy parameters
- New or worsening adverse effects temporally related to medication changes
- Laboratory abnormalities suggesting interaction
Documentation Elements:
- Date and time of event
- All medications involved
- Temporal relationship to dosing
- Severity grading
- Intervention and outcome
- Causality assessment
Regulatory and Ethical Considerations
Research protocols must address:
- Informed consent regarding interaction risks
- Ethics committee review of interaction management protocols
- Adverse event reporting to appropriate regulatory bodies
- Data safety monitoring board oversight for high-risk studies
Resources and Support for Retatrutide Research
Researchers working with retatrutide benefit from access to comprehensive resources and expert support.
Quality Research Materials
PEPTIDE PRO provides research-grade peptides with:
- Exceptional Purity: Rigorous quality control ensuring consistent experimental results
- Comprehensive Documentation: Certificate of Analysis (COA) and detailed product information
- Proper Storage: Temperature-controlled handling from production to delivery
- Expert Support: Technical guidance for research applications
Educational Resources
Researchers should consult:
- Peer-reviewed literature: Current publications on retatrutide pharmacology and interactions
- Pharmaceutical databases: Micromedex, Lexicomp, or similar interaction checking tools
- Regulatory guidance: FDA, EMA, and other regulatory body publications
- Professional organizations: Endocrine Society, American Diabetes Association research guidelines
Interaction Checking Tools
Recommended Resources:
- Clinical Pharmacology Database: Comprehensive interaction monographs
- Micromedex: Evidence-based interaction severity ratings
- Lexicomp: Detailed mechanism and management recommendations
- UpToDate: Clinical context and practical management strategies
Consultation and Collaboration
Complex interaction scenarios may benefit from:
- Clinical pharmacology consultation: Expert review of interaction potential
- Therapeutic drug monitoring: For medications with narrow therapeutic windows
- Multidisciplinary team input: Endocrinology, cardiology, nephrology as appropriate
For researchers seeking high-purity retatrutide and related compounds for their investigations, PEPTIDE PRO’s extensive catalogue offers laboratory-tested quality with fast UK shipping and international delivery options.
Future Directions in Retatrutide Interaction Research
As retatrutide advances through expanded research phases, several key areas will shape our understanding of its interaction profile.
Precision Medicine Approaches
Personalized Interaction Prediction:
- Integration of pharmacogenomic data
- Machine learning algorithms for interaction risk assessment
- Individualized monitoring protocols based on genetic and clinical factors
Biomarker Development:
- Identification of markers predicting interaction susceptibility
- Real-time monitoring technologies for early interaction detection
- Predictive models incorporating multiple risk factors
Technology Integration
Digital Health Tools:
- Mobile applications for medication tracking and interaction alerts
- Continuous monitoring devices for glucose, blood pressure, and other parameters
- Electronic health record integration for comprehensive medication review
Artificial Intelligence Applications:
- Pattern recognition in large datasets identifying novel interactions
- Predictive modeling for interaction severity and timing
- Decision support systems for dose optimization in complex medication regimens
Expanded Research Populations
Future studies will need to address:
- Diverse ethnic populations: Pharmacogenomic variations affecting interaction profiles
- Special populations: Pregnancy, lactation, pediatrics (where ethically appropriate)
- Complex comorbidities: Multiple disease states with polypharmacy
- Long-term interaction dynamics: Changes in interaction significance over years of use
Combination Therapy Optimization
Systematic investigation of:
- Synergistic combinations: Identifying medication pairings that enhance therapeutic benefits
- Antagonistic combinations: Understanding and avoiding counterproductive interactions
- Optimal sequencing: Determining best timing and order of medication introduction
- Dose optimization algorithms: Mathematical models for multi-drug regimen optimization
Understanding retatrutide drug interactions represents a critical component of responsible and effective research with this novel triple-agonist peptide. As we’ve explored throughout this comprehensive guide, retatrutide’s unique pharmacological profile—simultaneously activating GIP, GLP-1, and glucagon receptors—creates both remarkable research potential and complex interaction considerations.
Key Principles for Research Excellence
Successful research protocols incorporating retatrutide should be built on these foundational principles:
- Comprehensive Assessment: Thorough evaluation of all concurrent medications and health conditions before initiating retatrutide research
- Risk Stratification: Systematic categorization of interaction potential to guide monitoring intensity
- Proactive Monitoring: Implementation of robust laboratory and clinical monitoring protocols
- Flexible Protocols: Readiness to adjust doses, timing, or medication combinations based on emerging data
- Detailed Documentation: Meticulous recording of all medications, timing, and observed effects
- Ongoing Education: Commitment to staying current with evolving research on retatrutide interactions
Actionable Next Steps for Researchers
For Research Teams Planning Retatrutide Studies:
✅ Conduct comprehensive medication reviews for all potential subjects, identifying high-risk interactions early
✅ Develop detailed monitoring protocols tailored to your specific research population and concurrent medications
✅ Establish clear communication channels with clinical pharmacology experts for consultation on complex interaction scenarios
✅ Implement systematic documentation of all medication changes and suspected interaction events
✅ Source high-quality research materials from reputable suppliers like PEPTIDE PRO to ensure experimental consistency
For Individual Researchers:
✅ Review current literature on retatrutide pharmacology and emerging interaction data
✅ Familiarize yourself with interaction checking tools and databases
✅ Network with colleagues conducting similar research to share insights and experiences
✅ Consider additional training in clinical pharmacology or drug interactions
The Path Forward
As research with retatrutide expands in 2026 and beyond, our understanding of its interaction profile will continue to evolve. The triple-agonist mechanism that makes retatrutide so promising for metabolic research also necessitates heightened awareness of potential interactions across multiple physiological systems.
By maintaining rigorous standards for interaction assessment, monitoring, and management, the research community can maximize the scientific value of retatrutide studies while ensuring the highest levels of experimental integrity and safety. The comprehensive approach outlined in this guide provides a framework for navigating the complex landscape of retatrutide drug interactions, enabling researchers to design and execute studies that advance scientific knowledge while maintaining appropriate safeguards.
The future of retatrutide research is bright, with ongoing investigations continuing to reveal new insights into its therapeutic potential and interaction profile. By staying informed, implementing best practices, and maintaining a commitment to scientific rigor, researchers can contribute meaningfully to this exciting field while building the evidence base that will guide future applications of this novel peptide.
For researchers ready to advance their investigations with premium research-grade peptides, PEPTIDE PRO stands ready to support your work with exceptional purity, fast delivery, and professional service trusted by laboratories across the UK and worldwide.
