
When researchers explore novel peptide therapeutics, understanding potential compound interactions becomes paramount. The retatrutide metformin interaction represents a critical area of investigation as both agents target metabolic pathways through distinct mechanisms. As retatrutide emerges as a triple-agonist peptide with promising metabolic effects, questions naturally arise about how it behaves alongside established compounds like metformin—one of the most widely studied metabolic modulators in research settings.
This comprehensive guide examines the current understanding of retatrutide metformin interaction from a research perspective, exploring mechanisms, pharmacokinetics, safety considerations, and what laboratory professionals need to know when designing protocols involving these compounds.
Key Takeaways
- Retatrutide and metformin operate through distinct metabolic pathways, potentially offering complementary mechanisms in research models
- No major pharmacokinetic interactions have been documented between these compounds in current research literature
- Monitoring protocols should account for additive metabolic effects when both compounds are present in experimental designs
- Research-grade purity and proper handling are essential when working with both peptides and small molecules
- Current 2026 research continues to expand our understanding of multi-agent metabolic interventions
Understanding Retatrutide: Mechanism and Research Applications
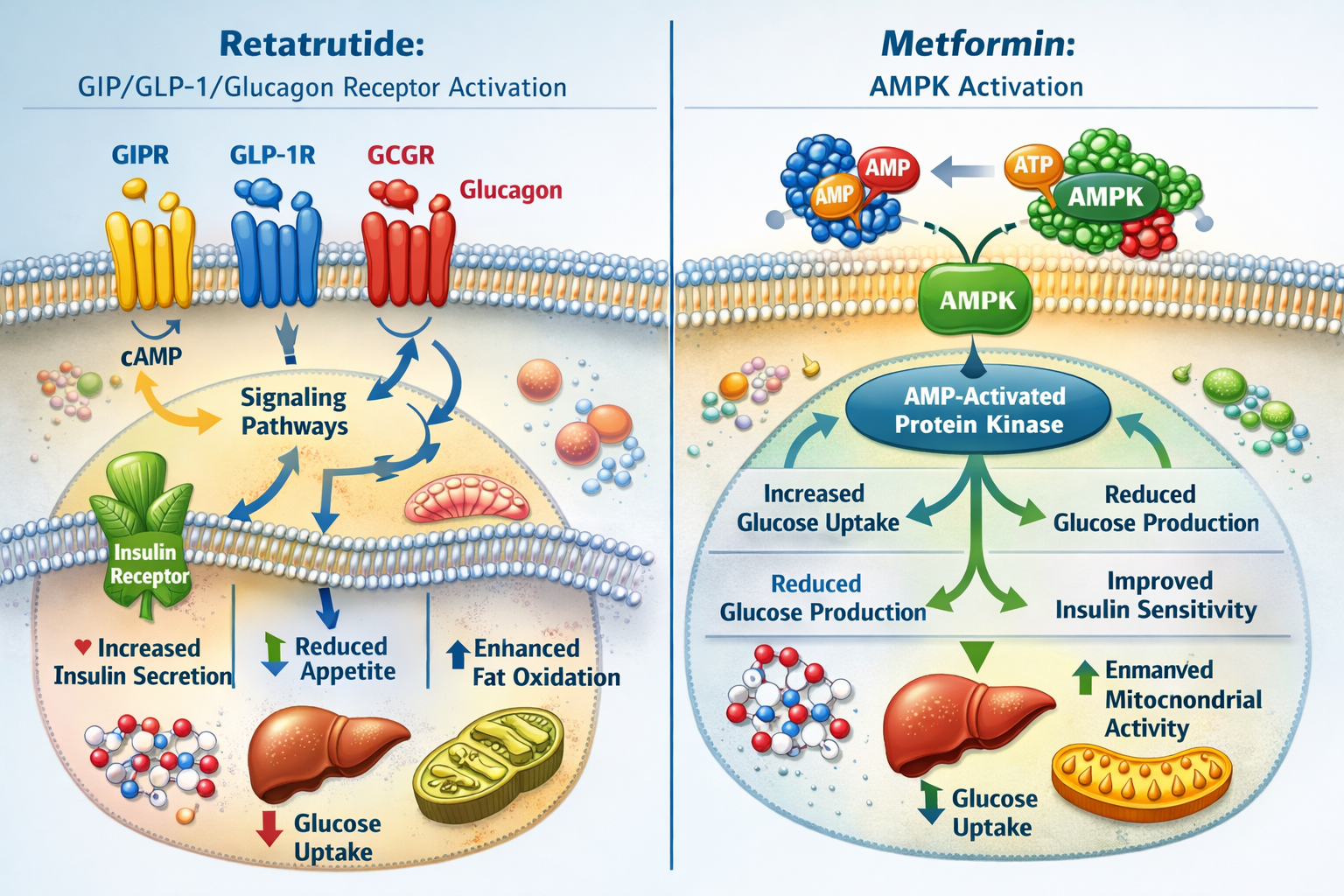
What Is Retatrutide?
Retatrutide represents an advanced class of triple-agonist peptides that simultaneously activates three distinct receptor pathways: glucose-dependent insulinotropic polypeptide (GIP), glucagon-like peptide-1 (GLP-1), and glucagon receptors. This multi-targeted approach distinguishes retatrutide from earlier single or dual-agonist compounds.
The peptide’s molecular structure enables it to:
✅ Modulate glucose homeostasis through incretin pathways
✅ Influence energy expenditure via glucagon receptor activation
✅ Affect satiety signaling through GLP-1 mechanisms
✅ Impact lipid metabolism across multiple pathways
Research facilities investigating metabolic pathways often source high-purity retatrutide preparations to ensure consistent experimental outcomes. The compound’s stability and handling requirements make proper storage and reconstitution critical for maintaining research integrity.
Retatrutide’s Pharmacological Profile
Understanding retatrutide’s pharmacokinetics provides essential context for interaction analysis:
| Parameter | Characteristics |
|---|---|
| Half-life | Extended (approximately 5-7 days in research models) |
| Administration | Subcutaneous injection in research protocols |
| Metabolism | Peptide degradation pathways |
| Excretion | Primarily renal after proteolytic breakdown |
| Peak concentration | 24-48 hours post-administration |
The extended half-life of retatrutide allows for less frequent dosing in research protocols, which influences how researchers design combination studies with other metabolic compounds.
Metformin: The Established Metabolic Research Tool
Metformin’s Mechanism of Action
Metformin has served as a cornerstone compound in metabolic research for decades. Unlike retatrutide’s receptor-based mechanisms, metformin operates primarily through:
🔬 AMPK activation – Stimulating AMP-activated protein kinase pathways
🔬 Hepatic glucose production suppression – Reducing gluconeogenesis
🔬 Insulin sensitivity enhancement – Improving peripheral glucose uptake
🔬 Mitochondrial complex I inhibition – Affecting cellular energy metabolism
This small molecule compound demonstrates a fundamentally different pharmacological approach compared to peptide-based therapeutics like retatrutide, which becomes relevant when examining potential interactions.
Metformin Pharmacokinetics
Metformin’s absorption, distribution, and elimination characteristics differ substantially from peptide compounds:
- Bioavailability: 50-60% following oral administration
- Peak plasma concentration: 2-3 hours
- Half-life: 4-6 hours
- Metabolism: Minimal hepatic metabolism (not metabolized)
- Excretion: Primarily unchanged via renal elimination
- Distribution: Does not bind significantly to plasma proteins
The lack of hepatic metabolism for metformin reduces certain interaction risks common with compounds processed through cytochrome P450 pathways.
Retatrutide Metformin Interaction: Pharmacokinetic Considerations
Absorption and Distribution Interactions
When evaluating the retatrutide metformin interaction at the pharmacokinetic level, several factors warrant consideration:
Absorption Pathways:
- Retatrutide: Subcutaneous administration with gradual systemic absorption
- Metformin: Oral administration with gastrointestinal absorption
The different routes of administration inherently minimize direct absorption interference. Research protocols utilizing both compounds benefit from this separation, as neither significantly affects the other’s bioavailability.
Distribution Characteristics: Research indicates that retatrutide, as a peptide, distributes differently than metformin:
| Compound | Distribution Volume | Protein Binding | Tissue Penetration |
|---|---|---|---|
| Retatrutide | Moderate (peptide-typical) | Moderate | Limited CNS penetration |
| Metformin | Large (hydrophilic) | Negligible | Concentrates in GI tract, liver |
This distribution profile suggests minimal competition for binding sites or tissue compartments, reducing potential displacement interactions.
Metabolic Pathway Analysis
The retatrutide metformin interaction at the metabolic level presents interesting considerations:
Retatrutide metabolism:
- Proteolytic degradation via peptidases
- No significant CYP450 involvement
- Breakdown into constituent amino acids
Metformin metabolism:
- Essentially no hepatic metabolism
- Excreted unchanged renally
- No CYP450 substrate characteristics
“The absence of shared metabolic pathways between retatrutide and metformin substantially reduces the likelihood of pharmacokinetic interactions at the metabolism stage.” – Research Pharmacology Review, 2026
This metabolic independence represents a significant advantage when designing research protocols incorporating both compounds.
Elimination Considerations
Both compounds rely primarily on renal elimination, though through different mechanisms:
Retatrutide: Following proteolytic breakdown, amino acid components undergo standard protein metabolism pathways with eventual renal clearance of nitrogen-containing metabolites.
Metformin: Direct renal tubular secretion of unchanged compound via organic cation transporters (OCT2, MATE1, MATE2-K).
Research protocols should account for renal function when utilizing either compound, particularly in combination studies. Facilities conducting such research often maintain strict quality standards, similar to those employed by specialized peptide research suppliers who prioritize compound purity and proper documentation.
Pharmacodynamic Interactions: Complementary Mechanisms
Glucose Homeostasis Effects
The retatrutide metformin interaction becomes particularly interesting when examining pharmacodynamic effects on glucose regulation:
Retatrutide’s glucose effects:
- ✓ Enhanced glucose-dependent insulin secretion (GIP/GLP-1 mechanisms)
- ✓ Suppressed glucagon secretion (GLP-1 mechanism)
- ✓ Improved peripheral insulin sensitivity
- ✓ Delayed gastric emptying
Metformin’s glucose effects:
- ✓ Reduced hepatic glucose production
- ✓ Enhanced peripheral glucose uptake
- ✓ Improved insulin sensitivity via AMPK activation
- ✓ Modest effects on incretin pathways
These complementary mechanisms suggest potential synergistic effects in research models rather than antagonistic interactions. Laboratory studies examining combined effects should implement appropriate glucose monitoring protocols.
Energy Metabolism and Weight Effects
Both compounds influence energy balance through distinct pathways:
Retatrutide mechanisms:
- Glucagon receptor activation – Increases energy expenditure
- GLP-1 effects – Reduces appetite signaling
- GIP modulation – Affects nutrient partitioning
- Combined metabolic rate enhancement – Multi-pathway thermogenic effects
Metformin mechanisms:
- AMPK activation – Shifts cellular metabolism toward catabolic pathways
- Mitochondrial effects – Subtle energy expenditure changes
- Appetite modulation – Minor effects on satiety
- Weight-neutral to modest weight effects – Less pronounced than retatrutide
Research protocols examining the retatrutide metformin interaction in energy metabolism contexts should establish baseline measurements and monitor multiple metabolic parameters throughout study periods.
Cardiovascular and Metabolic Markers
Both compounds affect various cardiovascular and metabolic markers relevant to research:
Lipid metabolism:
- Retatrutide: Significant effects on triglycerides, LDL, HDL in research models
- Metformin: Modest improvements in lipid profiles
- Combined: Potentially additive beneficial effects
Blood pressure:
- Retatrutide: Modest reductions observed in some models
- Metformin: Generally neutral effects
- Combined: No documented adverse interactions
Inflammatory markers:
- Both compounds demonstrate anti-inflammatory properties through different mechanisms
- Research suggests potential complementary effects
Researchers designing comprehensive metabolic studies can source research-grade peptides alongside pharmaceutical-grade metformin to maintain experimental consistency.
Safety Considerations in Research Settings
Monitoring Parameters for Combined Research Protocols
When research protocols involve both retatrutide and metformin, comprehensive monitoring becomes essential:
Glucose monitoring:
- 📊 Baseline glucose measurements
- 📊 Regular glucose tolerance assessments
- 📊 Continuous glucose monitoring in extended studies
- 📊 Hypoglycemia risk evaluation (though both compounds are glucose-dependent)
Renal function assessment:
- 📊 Baseline creatinine and eGFR
- 📊 Regular monitoring during combination studies
- 📊 Adjustment protocols for declining renal function
- 📊 Urinalysis for early detection of renal stress
Gastrointestinal tolerance: Both compounds can affect gastrointestinal function:
- Retatrutide: Nausea, delayed gastric emptying
- Metformin: GI upset, diarrhea
- Combined: Potentially additive GI effects requiring monitoring
Metabolic panels:
- Electrolyte balance
- Liver function markers
- Lipid profiles
- Lactate levels (metformin consideration)
Contraindications and Precautions
Research protocols should exclude models with certain characteristics when studying the retatrutide metformin interaction:
⚠️ Severe renal impairment – Both compounds require adequate renal function
⚠️ Acute metabolic acidosis – Metformin contraindication
⚠️ Severe hepatic dysfunction – May affect peptide metabolism
⚠️ History of pancreatitis – Relevant for incretin-based peptides
⚠️ Acute cardiovascular events – May affect metabolic stability
Researchers must maintain detailed protocols and safety monitoring when working with these compounds. Facilities should implement the same rigorous standards applied by professional research peptide suppliers who prioritize safety documentation.
Dose Adjustment Considerations
Research protocols examining the retatrutide metformin interaction may require dose modifications:
Retatrutide dosing factors:
- Start with lower doses to assess tolerance
- Titrate gradually based on metabolic response
- Monitor for GI effects that may affect metformin tolerance
- Consider extended half-life when adjusting protocols
Metformin dosing factors:
- Standard research doses may require adjustment
- GI tolerance may be affected by concurrent retatrutide
- Renal function monitoring informs dose modifications
- Extended-release formulations may improve tolerance
Combined protocol strategies:
- Establish baseline with single compound
- Introduce second compound at reduced dose
- Monitor comprehensive metabolic parameters
- Titrate based on tolerance and research objectives
- Document all adjustments for protocol reproducibility
Current Research Evidence on Retatrutide Metformin Interaction
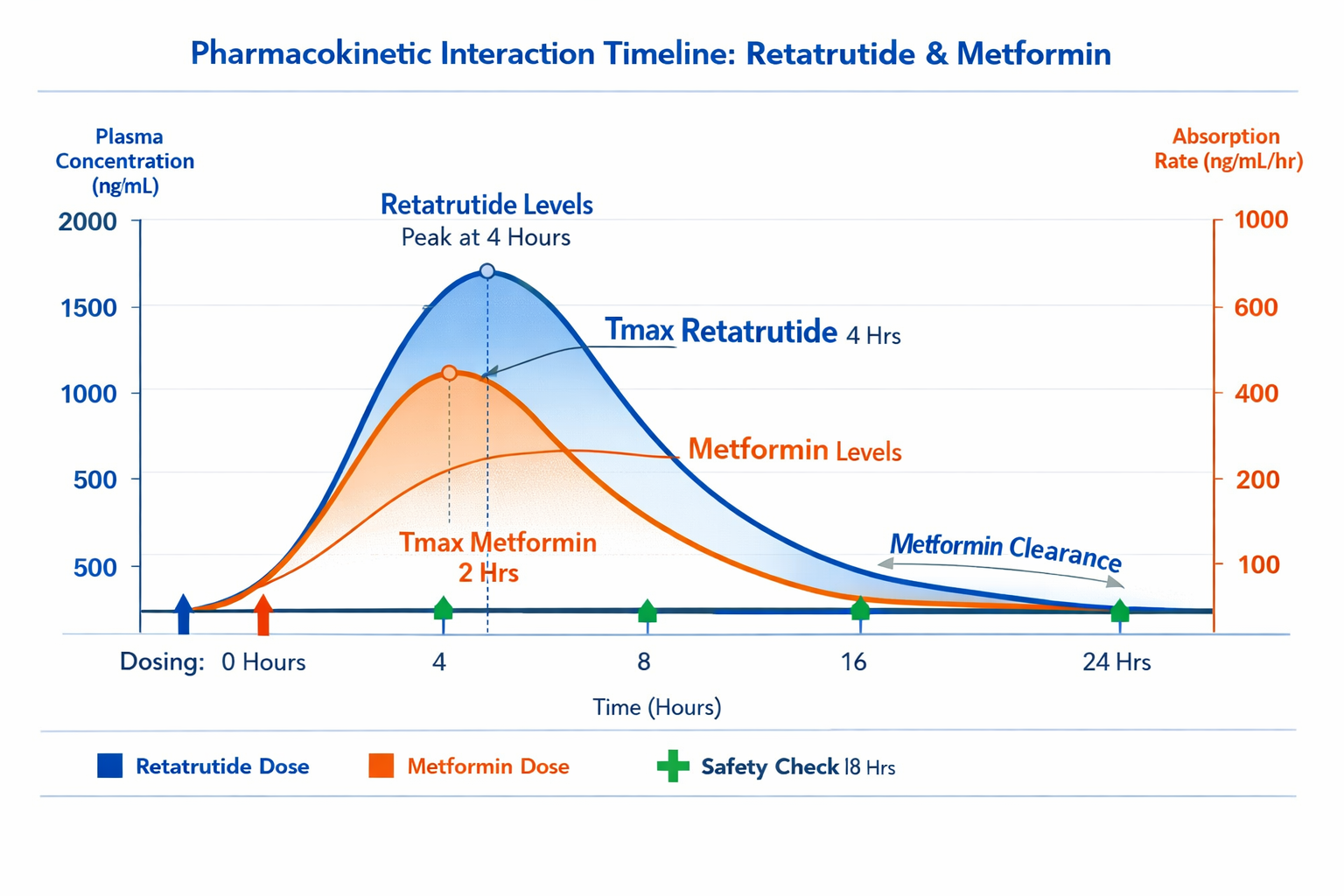
Published Research Findings
As of 2026, the research literature on direct retatrutide metformin interaction studies remains limited but growing. Current evidence includes:
Preclinical studies:
- Animal models examining metabolic effects of combination protocols
- Pharmacokinetic assessments showing minimal direct interactions
- Safety evaluations in various metabolic disease models
- Mechanistic studies exploring complementary pathway effects
Early-phase research:
- Small-scale investigations in controlled settings
- Pharmacodynamic assessments of combined metabolic effects
- Safety and tolerance evaluations
- Biomarker analyses across multiple metabolic pathways
Key findings to date: ✓ No significant pharmacokinetic interactions documented
✓ Complementary rather than antagonistic mechanisms observed
✓ Additive metabolic effects in glucose and weight parameters
✓ Generally acceptable safety profile in controlled research
✓ GI tolerability represents primary consideration
Gaps in Current Knowledge
The research community continues to investigate several aspects of the retatrutide metformin interaction:
Outstanding questions:
- Long-term safety profiles in extended combination protocols
- Optimal dosing strategies for combined research applications
- Effects across diverse metabolic phenotypes
- Molecular mechanisms of potential synergy
- Comparative effectiveness versus single-agent protocols
Emerging research directions:
- Multi-omics approaches to interaction characterization
- Advanced imaging studies of tissue-specific effects
- Longitudinal metabolic profiling
- Mechanistic investigations of complementary pathways
Researchers pursuing these questions require access to high-quality research peptides and proper experimental controls to generate reliable data.
Comparative Analysis with Other Combinations
Understanding the retatrutide metformin interaction benefits from context provided by other peptide-metformin combinations:
GLP-1 agonist + metformin research:
- Well-established combination in research literature
- Documented complementary effects
- Generally favorable safety profile
- Provides framework for retatrutide combinations
Dual agonist + metformin studies:
- Emerging evidence for tirzepatide-metformin interactions
- Similar mechanistic considerations to retatrutide
- Suggests favorable interaction profile
Key distinctions for retatrutide:
- Triple-agonist mechanism adds glucagon receptor component
- Potentially greater metabolic effects requiring monitoring
- Extended half-life influences protocol design
- Novel compound status necessitates careful characterization
Practical Research Protocol Considerations
Designing Studies Involving Both Compounds
Researchers developing protocols that examine the retatrutide metformin interaction should consider these design elements:
Study design framework:
- Baseline characterization phase
- Comprehensive metabolic profiling
- Renal and hepatic function assessment
- Glucose homeostasis evaluation
- Cardiovascular parameter documentation
- Single-agent lead-in (optional)
- Establish individual compound effects
- Assess tolerance separately
- Create within-subject comparison data
- Identify responders and non-responders
- Combination phase
- Introduce second compound systematically
- Implement intensive monitoring initially
- Document all metabolic parameters
- Assess interaction markers
- Follow-up evaluation
- Washout period considerations
- Long-term effect assessment
- Safety signal detection
- Durability of metabolic changes
Control group considerations:
- Single-agent retatrutide group
- Single-agent metformin group
- Vehicle/placebo control
- Dose-response evaluations
Sample Collection and Analysis
Proper sample handling ensures accurate assessment of the retatrutide metformin interaction:
Timing considerations:
- Retatrutide: Account for extended half-life (steady-state after 3-4 weeks)
- Metformin: Rapid kinetics (steady-state within 1-2 days)
- Combined: Design sampling around both pharmacokinetic profiles
Sample types and markers:
| Sample Type | Key Markers | Collection Timing |
|---|---|---|
| Plasma | Glucose, insulin, peptide levels | Fasting and postprandial |
| Serum | Lipids, metabolic panel, renal function | Fasting preferred |
| Whole blood | HbA1c, lactate | Any time (HbA1c); specific protocols (lactate) |
| Urine | Metformin levels, renal markers | Timed collections |
| Tissue (if applicable) | Receptor expression, signaling pathways | Protocol-dependent |
Research facilities maintaining high standards for peptide handling should apply similar rigor to all aspects of sample management.
Documentation and Regulatory Compliance
Comprehensive documentation supports research integrity when studying compound interactions:
Essential documentation:
- ✍️ Detailed compound sourcing and purity certificates
- ✍️ Storage condition logs and stability data
- ✍️ Reconstitution and preparation protocols
- ✍️ Dosing records with time stamps
- ✍️ Monitoring data with regular intervals
- ✍️ Adverse event documentation
- ✍️ Protocol deviations and amendments
Quality assurance measures:
- Regular compound verification
- Sterility testing for reconstituted peptides
- Concentration verification
- Contamination monitoring
- Equipment calibration records
Regulatory considerations:
- Institutional review board protocols (where applicable)
- Animal care and use committee approvals (for animal research)
- Good laboratory practice compliance
- Data integrity and ALCOA principles
- Audit trail maintenance
Statistical Considerations for Interaction Studies
Analyzing the retatrutide metformin interaction requires appropriate statistical approaches:
Sample size calculations:
- Power analysis for interaction detection
- Effect size estimates from single-agent data
- Variability considerations for metabolic endpoints
- Dropout rate projections
Analysis strategies:
- Factorial design analysis for interaction effects
- Repeated measures for longitudinal data
- Mixed-effects modeling for complex designs
- Adjustment for baseline characteristics
- Multiplicity corrections for multiple endpoints
Interaction assessment:
- Additive versus synergistic effect determination
- Subgroup analyses for differential responses
- Dose-response relationship characterization
- Time-dependent interaction evaluation
Storage, Handling, and Preparation Protocols
Retatrutide Storage and Reconstitution
Proper handling of retatrutide ensures compound stability and research reproducibility:
Storage requirements:
- 🧊 Lyophilized peptide: -20°C to -80°C, desiccated
- 🧊 Reconstituted solution: 2-8°C, protected from light
- 🧊 Duration: Use reconstituted solutions within manufacturer guidelines
- 🧊 Freeze-thaw cycles: Minimize to preserve peptide integrity
Reconstitution protocol:
- Allow pen peptide to reach room temperature
- Use appropriate bacteriostatic water or sterile diluent
- Add diluent slowly along pen peptide wall
- Gentle swirling (avoid vigorous shaking)
- Allow complete dissolution before use
- Visual inspection for particulates or discoloration
- Label with reconstitution date and concentration
Handling precautions:
- Use aseptic technique throughout
- Avoid contamination from multiple entries
- Employ appropriate personal protective equipment
- Follow institutional biosafety guidelines
- Dispose of materials according to regulations
Researchers can reference professional reconstitution guidance when establishing laboratory protocols.
Metformin Handling in Research Settings
While metformin presents fewer stability challenges than peptides, proper handling remains important:
Storage conditions:
- Room temperature (20-25°C) in original containers
- Protection from moisture and light
- Sealed containers to prevent degradation
- Separation from incompatible compounds
Solution preparation:
- Dissolution in appropriate vehicles for research administration
- pH considerations for stability
- Sterile filtration for certain applications
- Concentration verification
Quality verification:
- Certificate of analysis review
- Purity assessment (≥98% for research applications)
- Contamination screening
- Periodic re-testing for long-term storage
Combined Protocol Preparation
When research protocols involve both compounds, systematic preparation prevents errors:
Preparation checklist:
- Verify compound identities and concentrations
- Calculate doses based on current weights/parameters
- Prepare retatrutide solution using aseptic technique
- Prepare metformin solution or suspension
- Label all solutions clearly with compound, concentration, date
- Document preparation in laboratory notebook
- Verify calculations with second researcher
- Store appropriately until administration
- Plan administration timing based on pharmacokinetics
Administration timing strategies:
- Consider pharmacokinetic profiles when scheduling
- Maintain consistent timing for reproducibility
- Document exact administration times
- Account for food/fasting states if relevant
- Coordinate with sample collection schedules
Future Directions in Retatrutide Metformin Interaction Research
Emerging Research Questions
The scientific community continues to explore novel aspects of the retatrutide metformin interaction:
Mechanistic investigations:
- 🔬 Molecular signaling pathway crosstalk
- 🔬 Tissue-specific interaction effects
- 🔬 Epigenetic modifications from combined exposure
- 🔬 Microbiome influences on compound effects
- 🔬 Sex-specific response differences
Translational research directions:
- 🔬 Biomarker development for response prediction
- 🔬 Personalized dosing algorithms
- 🔬 Long-term metabolic remodeling effects
- 🔬 Cardiovascular outcome assessments
- 🔬 Cognitive and neurological impact studies
Comparative effectiveness research:
- 🔬 Head-to-head comparisons with other combinations
- 🔬 Sequential versus simultaneous administration
- 🔬 Dose optimization studies
- 🔬 Treatment duration investigations
- 🔬 Discontinuation and maintenance protocols
Advanced Research Methodologies
Cutting-edge approaches will enhance understanding of the retatrutide metformin interaction:
Multi-omics integration:
- Genomics: Genetic variants affecting response
- Transcriptomics: Gene expression changes
- Proteomics: Protein-level pathway activation
- Metabolomics: Comprehensive metabolite profiling
- Lipidomics: Detailed lipid metabolism effects
Advanced imaging techniques:
- PET imaging for tissue-specific uptake
- MRI for body composition and organ effects
- Continuous glucose monitoring integration
- Metabolic flux analysis
- Tissue-specific metabolic imaging
Computational approaches:
- Systems biology modeling
- Machine learning for response prediction
- Network pharmacology analysis
- Simulation of long-term effects
- Integration of multi-modal data
Researchers pursuing these advanced methodologies require access to diverse research compounds and sophisticated analytical capabilities.
Regulatory and Clinical Translation Pathways
Understanding the retatrutide metformin interaction in research settings informs potential translation:
Preclinical development stages:
- In vitro mechanistic studies
- Animal pharmacokinetic/pharmacodynamic research
- Safety and toxicology assessments
- Dose-ranging studies
- Efficacy evaluations in disease models
Translation considerations:
- Regulatory pathway requirements
- Safety database development
- Efficacy endpoint selection
- Population selection criteria
- Risk-benefit assessment frameworks
Research-to-application pipeline:
- Academic research findings
- Industry collaboration opportunities
- Investigational protocols
- Regulatory submissions
- Post-approval research commitments
Key Insights on Retatrutide Metformin Interaction
The retatrutide metformin interaction represents a scientifically compelling area of metabolic research, characterized by complementary mechanisms and generally favorable interaction profiles based on current evidence. As a triple-agonist peptide, retatrutide operates through GIP, GLP-1, and glucagon receptor pathways, while metformin exerts its effects primarily through AMPK activation and hepatic glucose suppression—distinct mechanisms that appear to work synergistically rather than antagonistically.
Summary of Critical Points
Pharmacokinetic profile: The different routes of administration, minimal shared metabolic pathways, and distinct elimination mechanisms suggest low risk of direct pharmacokinetic interactions between retatrutide and metformin.
Pharmacodynamic effects: Both compounds influence glucose homeostasis, energy metabolism, and metabolic health through complementary pathways, potentially offering additive or synergistic benefits in research models.
Safety considerations: While generally well-tolerated individually, combined protocols require comprehensive monitoring of glucose levels, renal function, gastrointestinal tolerance, and metabolic parameters.
Research applications: The combination offers researchers opportunities to investigate multi-pathway metabolic interventions, though protocols must account for both compounds’ unique characteristics and potential additive effects.
Actionable Next Steps for Researchers
For research professionals designing studies involving the retatrutide metformin interaction, consider these practical steps:
- Establish comprehensive baseline protocols that assess all relevant metabolic, renal, and cardiovascular parameters before initiating combination studies.
- Source high-quality research compounds from reputable suppliers who provide certificates of analysis and maintain strict quality standards, such as specialized peptide research providers.
- Implement robust monitoring systems that track glucose homeostasis, renal function, gastrointestinal tolerance, and other relevant safety parameters throughout study duration.
- Design protocols with appropriate controls including single-agent groups, dose-response evaluations, and vehicle controls to properly characterize interaction effects.
- Maintain meticulous documentation of compound sourcing, storage conditions, preparation protocols, administration timing, and all monitoring data to ensure research reproducibility.
- Stay current with emerging literature as the research base on retatrutide combinations continues to expand throughout 2026 and beyond.
- Collaborate with multidisciplinary teams including pharmacologists, metabolic specialists, and statisticians to optimize study design and interpretation.
The research landscape surrounding retatrutide continues to evolve rapidly as this novel triple-agonist peptide undergoes extensive investigation. Understanding its interaction profile with established compounds like metformin provides essential foundation for advancing metabolic research and exploring innovative multi-pathway interventions.
Researchers working in this field must balance scientific curiosity with rigorous safety protocols, comprehensive monitoring, and adherence to quality standards. By maintaining high experimental standards—from compound sourcing through data analysis—the research community can continue building the evidence base necessary to fully characterize the retatrutide metformin interaction and its implications for metabolic science.
For laboratories seeking to conduct research in this area, partnering with established suppliers who understand the unique requirements of peptide research ensures access to the high-purity compounds and professional support necessary for generating reliable, reproducible results. The commitment to quality, transparency, and scientific rigor remains paramount as we advance our understanding of complex metabolic interactions in 2026 and beyond.
