
When working with research-grade peptides like retatrutide, proper storage isn’t just a recommendation—it’s essential for maintaining molecular integrity and research validity. Does retatrutide need refrigeration? The answer depends critically on whether the peptide is in its lyophilized (freeze-dried) form or has been reconstituted with bacteriostatic water. Understanding these storage requirements can mean the difference between reliable research results and compromised compounds that waste both time and resources.
Retatrutide, a novel triple agonist peptide targeting GIP, GLP-1, and glucagon receptors, has gained significant attention in metabolic research. As researchers and laboratories increasingly work with this compound, questions about proper storage conditions have become paramount. This comprehensive guide addresses every aspect of retatrutide refrigeration requirements, from initial storage through reconstitution and beyond.
Key Takeaways
- Lyophilized retatrutide can be stored at room temperature (below 25°C) short-term but benefits from refrigeration at 2-8°C for extended stability
- Reconstituted retatrutide requires strict refrigeration at 2-8°C and should be used within 28 days for optimal stability
- Never freeze reconstituted peptides—freezing damages the molecular structure and renders the compound ineffective
- Proper storage conditions directly impact research validity, peptide potency, and experimental reproducibility
- Temperature-controlled shipping and appropriate packaging are essential for maintaining peptide integrity during transit
Understanding Retatrutide: What Makes This Peptide Unique?
Retatrutide represents a significant advancement in peptide research, functioning as a triple receptor agonist with activity at glucose-dependent insulinotropic polypeptide (GIP), glucagon-like peptide-1 (GLP-1), and glucagon receptors [1]. This multi-targeted mechanism distinguishes it from single or dual agonist compounds, making proper handling and storage even more critical for research applications.
The Molecular Structure and Stability Considerations
The peptide’s complex molecular architecture includes specific amino acid sequences that confer its receptor-binding properties. Like most research peptides, retatrutide is sensitive to:
- Temperature fluctuations that can denature protein structures
- Light exposure that may degrade certain amino acid residues
- Moisture when in lyophilized form
- Bacterial contamination after reconstitution
- Freeze-thaw cycles that disrupt molecular integrity
At PEPTIDE PRO, we supply retatrutide in its most stable lyophilized form, carefully processed under controlled conditions to maximize shelf life and maintain research-grade purity.
Why Storage Conditions Matter for Research Validity
Research outcomes depend entirely on compound integrity. When peptides degrade due to improper storage, researchers may observe:
- Reduced biological activity in assays
- Inconsistent results between experiments
- False negative findings
- Wasted research materials and funding
- Compromised data quality
“Proper peptide storage isn’t optional—it’s fundamental to research integrity. Temperature control, light protection, and contamination prevention form the foundation of reliable peptide research.” — Laboratory Best Practices Guide
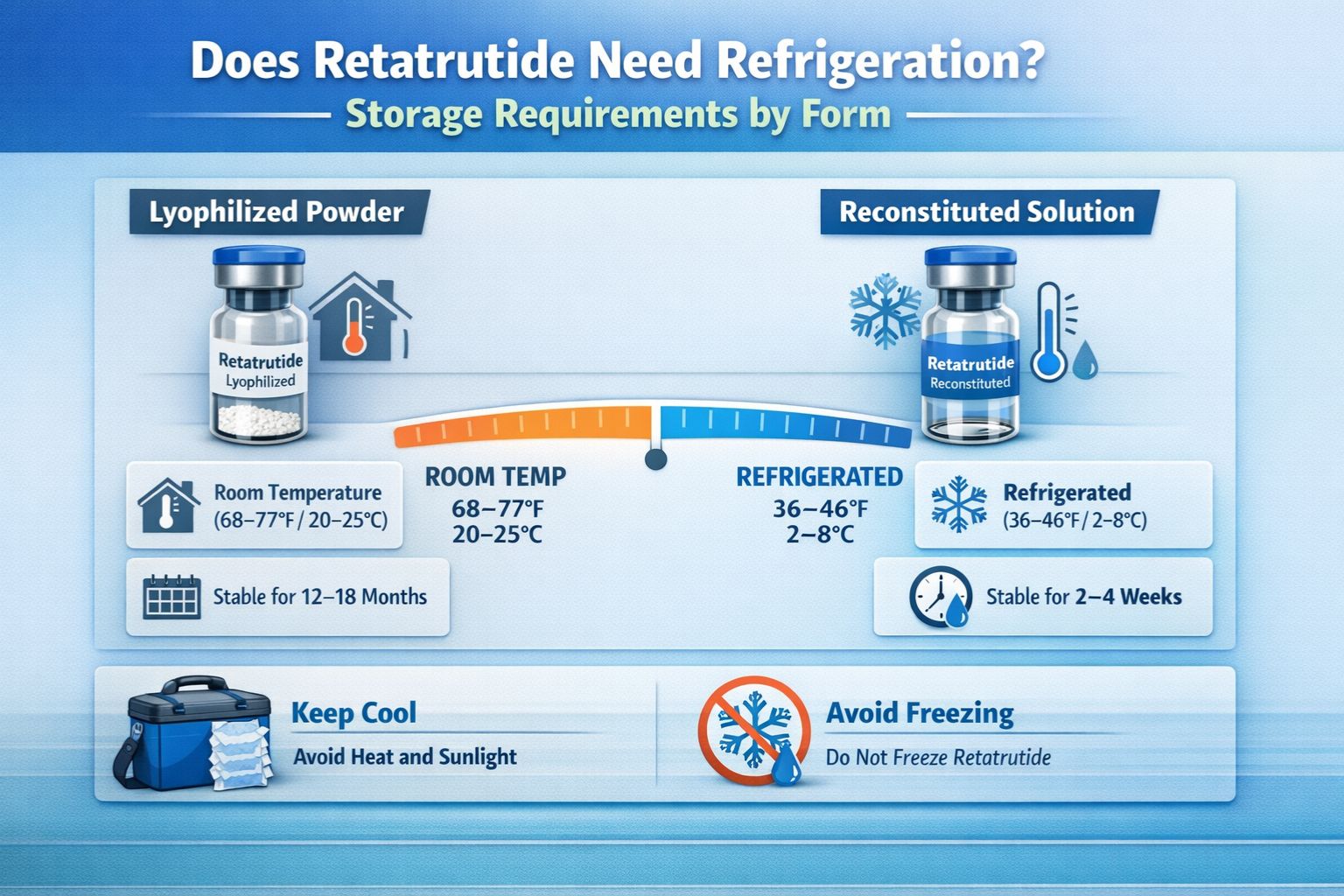
Does Retatrutide Need Refrigeration? Storage Requirements by Form
The refrigeration requirements for retatrutide vary significantly depending on whether the peptide is in its lyophilized or reconstituted state. Understanding these distinctions is crucial for maintaining compound stability.
Lyophilized (Freeze-Dried) Retatrutide Storage
Lyophilized retatrutide offers remarkable stability when stored correctly. In this dehydrated powder form, the peptide exhibits:
Short-Term Storage (Up to 30 Days)
- ✅ Room temperature storage acceptable (below 25°C)
- ✅ Keep in original sealed pen peptide
- ✅ Protect from direct light
- ✅ Store in low-humidity environment
- ✅ Avoid temperature fluctuations
Long-Term Storage (Beyond 30 Days)
️ Refrigeration at 2-8°C recommended
- ❄️ Freezer storage at -20°C optimal for extended periods (6-12 months)
- 📦 Keep in original packaging with desiccant
- 🔒 Seal tightly to prevent moisture exposure
- 📝 Label with receipt date and storage conditions
When you order retatrutide from PEPTIDE PRO, the compound arrives in pharmaceutical-grade pen peptides with appropriate sealing to maintain stability during shipping and initial storage.
Reconstituted Retatrutide Storage: Critical Refrigeration Required
Once retatrutide has been reconstituted with bacteriostatic water or another appropriate diluent, storage requirements become significantly more stringent.
Mandatory Refrigeration Parameters
| Storage Aspect | Requirement | Rationale |
|---|---|---|
| Temperature | 2-8°C (36-46°F) | Prevents bacterial growth and peptide degradation |
| Duration | Use within 28 days | Bacteriostatic properties decline over time |
| Light Protection | Store in dark or amber pen peptide | Prevents photodegradation |
| Positioning | Upright storage preferred | Minimizes rubber stopper contact |
| Freezing | ❌ NEVER freeze | Ice crystals destroy peptide structure |
Why refrigeration is non-negotiable for reconstituted peptides:
- Bacterial contamination prevention: Even with bacteriostatic water, refrigeration inhibits microbial growth
- Peptide bond stability: Lower temperatures slow hydrolysis and oxidation reactions
- Structural integrity: Cool conditions maintain the three-dimensional protein configuration
- Solubility maintenance: Prevents precipitation and aggregation
Temperature Monitoring Best Practices
For laboratories working with valuable research compounds like retatrutide, implementing temperature monitoring protocols ensures storage compliance:
- 📊 Use calibrated thermometers or digital temperature loggers
- 🔔 Set alarms for temperature excursions beyond 2-8°C range
- 📝 Maintain temperature logs for quality assurance
- 🔄 Verify refrigerator performance regularly
- 🚫 Avoid storing peptides in refrigerator doors (temperature fluctuates)
Reconstitution and Post-Mixing Storage Guidelines
The reconstitution process represents a critical transition point where storage requirements change dramatically. Proper technique and immediate refrigeration are essential.
Step-by-Step Reconstitution Protocol
Materials needed:
- Lyophilized retatrutide pen peptide
- Bacteriostatic water (0.9% benzyl alcohol)
- Sterile syringes and needles
- Alcohol swabs
- Refrigerated storage immediately available
Reconstitution procedure:
- Preparation Phase
- Remove retatrutide pen peptide from storage
- Allow to reach room temperature (15-20 minutes)
- Clean pen peptide top with alcohol swab
- Prepare bacteriostatic water syringe
- Mixing Phase
- Inject bacteriostatic water slowly down pen peptide side (not directly onto powder)
- Allow liquid to gently dissolve powder without shaking
- Swirl gently if needed—never shake vigorously
- Ensure complete dissolution before use
- Immediate Storage
- ⚡ Transfer to refrigerator within 5 minutes of reconstitution
- Label pen peptide with reconstitution date and concentration
- Store at 2-8°C consistently
- Protect from light with amber pen peptide or foil wrap
Common Reconstitution Mistakes That Affect Storage Stability
❌ Shaking the pen peptide vigorously – Creates foam and can denature peptides ❌ Using incorrect diluent – Tap water or saline without preservative promotes bacterial growth ❌ Delayed refrigeration – Room temperature exposure accelerates degradation ❌ Reconstituting entire inventory – Only mix what you’ll use within 28 days ❌ Freezing reconstituted solution – Destroys molecular structure irreversibly
Multi-Dose Vial Management
When working with reconstituted retatrutide over several days or weeks:
- 🧊 Return to refrigerator immediately after each use
- ⏱️ Minimize time at room temperature (under 15 minutes per access)
- 🧼 Swab pen peptide top with alcohol before each needle insertion
- 📅 Track usage dates and discard after 28 days
️ Inspect for cloudiness, particles, or discoloration before each use
Research facilities should establish standard operating procedures (SOPs) for peptide reconstitution and storage to ensure consistency across team members.
Transportation and Shipping Considerations for Retatrutide
Maintaining proper temperature during transportation is equally critical as storage. Peptides can experience significant degradation during shipping if not properly packaged.
How PEPTIDE PRO Ensures Temperature Control During Delivery
When you order from PEPTIDE PRO, our commitment to peptide integrity extends through the entire delivery process:
- 📦 Temperature-appropriate packaging with insulation materials
- 🧊 Cold packs or gel refrigerants for temperature-sensitive shipments
- ⚡ Same-day dispatch for orders placed before 1pm (Mon-Fri)
- 📍 Full tracking to minimize delivery delays
️ Seasonal packaging adjustments based on ambient temperatures
Receiving Your Retatrutide Shipment: Immediate Steps
Upon receiving your peptide delivery:
- Inspect packaging immediately for signs of temperature compromise
- Check pen peptide integrity – ensure seals are intact and no moisture inside
- Transfer to appropriate storage within 30 minutes of receipt
- Document receipt date on pen peptide label
- Report any concerns to PEPTIDE PRO customer support immediately
International Shipping and Extended Transit Times
For international orders or remote locations where transit may exceed 48 hours:
- Consider ordering during moderate weather seasons
- Communicate specific delivery timing requirements
- Understand that lyophilized peptides tolerate brief temperature variations better than reconstituted forms
- Plan reconstitution timing around delivery schedules
Our extensive peptide range includes various compounds with similar storage requirements, and our team can provide specific guidance for your research needs.
Comparing Retatrutide Storage to Other Research Peptides
Understanding how retatrutide storage compares to other common research peptides provides valuable context for laboratory storage planning.
Storage Requirements Across Popular Research Peptides
| Peptide | Lyophilized Storage | Reconstituted Storage | Special Considerations |
|---|---|---|---|
| Retatrutide | 2-8°C or -20°C long-term | 2-8°C, 28 days max | Triple agonist, light-sensitive |
| Semaglutide | 2-8°C preferred | 2-8°C, 28 days max | GLP-1 agonist, similar stability |
| Tirzepatide | 2-8°C or -20°C | 2-8°C, 28 days max | Dual agonist, refrigeration critical |
| BPC-157 | Room temp acceptable | 2-8°C, 30 days max | Relatively stable peptide |
| TB-500 | 2-8°C recommended | 2-8°C, 28 days max | Thymosin beta-4 fragment |
| CJC-1295 | -20°C optimal | 2-8°C, 21 days max | GHRH analog, more temperature-sensitive |
Why Some Peptides Are More Storage-Sensitive
Peptide stability varies based on:
- Amino acid composition – Methionine and cysteine residues are oxidation-prone
- Sequence length – Longer peptides generally more fragile
- Structural complexity – Modified peptides may have additional stability concerns
- Hydrophobicity – Affects aggregation tendencies
- Post-translational modifications – Acylation, PEGylation affect stability profiles
Retatrutide’s triple agonist activity and specific modifications place it in the category requiring careful temperature control, similar to other metabolic research peptides.
Signs of Peptide Degradation: When Storage Conditions Fail
Recognizing degradation signs helps researchers avoid using compromised compounds that could invalidate research findings.
Visual Inspection Indicators
For lyophilized retatrutide:
- ❌ Discoloration (yellowing or browning)
- ❌ Cake collapse or unusual texture
- ❌ Moisture inside sealed pen peptide
- ❌ Crystallization or unusual formations
- ✅ Should appear as white to off-white powder
For reconstituted retatrutide:
- ❌ Cloudiness or turbidity
- ❌ Visible particles or precipitates
- ❌ Color changes
- ❌ Unusual odor
- ✅ Should be clear, colorless solution
Performance-Based Degradation Signs
Beyond visual inspection, degraded peptides may exhibit:
- Reduced solubility during reconstitution
- Unexpected pH changes
- Decreased biological activity in assays
- Inconsistent results compared to fresh preparations
- Failed positive controls in experiments
What to Do If You Suspect Degradation
If you observe any degradation indicators:
- Do not use the peptide for critical research
- Document observations with photos and notes
- Review storage logs to identify potential temperature excursions
- Contact supplier – PEPTIDE PRO provides support for storage-related concerns
- Consider ordering fresh material to ensure research validity
Laboratory Storage Infrastructure: Setting Up for Success
Proper storage infrastructure represents an investment in research quality and peptide longevity.
Essential Equipment for Peptide Storage
Refrigeration systems:
- 🏥 Medical-grade refrigerators with precise temperature control
- 📊 Digital temperature displays with min/max recording
- 🔔 Alarm systems for temperature excursions
- 🚪 Glass doors (optional) for inventory visibility without opening
- 🔒 Lockable units for controlled access
Freezer storage (for long-term lyophilized storage):
- ❄️ -20°C or -80°C freezers depending on duration needs
- 📦 Organized storage boxes with dividers
️ Clear labeling systems with date tracking
- 🧊 Frost-free or manual defrost considerations
Supporting equipment:
️ Calibrated thermometers (certified annually)
- 📝 Temperature logging systems (digital or manual)
- 🧴 Desiccant containers for lyophilized storage
- 🔦 Amber pen peptides or foil wraps for light protection
- 🧤 Sterile handling supplies
Storage Organization Best Practices
Efficient organization prevents storage errors and extends peptide viability:
Labeling protocols:
- Peptide name and concentration
- Receipt or reconstitution date
- Expiration or use-by date
- Storage temperature requirements
- Lot or batch number
Inventory management:
- First-in, first-out (FIFO) rotation
- Separate storage for lyophilized vs. reconstituted
- Dedicated sections for different peptide types
- Regular inventory audits
- Digital tracking systems for larger collections
Access controls:
- Limit personnel with refrigerator access
- Training requirements for handling protocols
- Documentation of each access event for critical materials
- Standard operating procedures posted visibly
Cost-Benefit Analysis: Proper Storage vs. Peptide Replacement
Investing in proper storage infrastructure and protocols delivers significant return on investment compared to replacing degraded peptides.
Hidden Costs of Improper Storage
When peptides degrade due to storage failures, researchers face:
Direct costs:
- 💰 Replacement peptide purchase (£275+ for retatrutide 40mg)
- 📦 Additional shipping charges
- ⏱️ Delayed research timelines
- 🔄 Repeated experiments with fresh material
Indirect costs:
- 📊 Compromised data requiring exclusion
- 📄 Publication delays or rejections
- 👥 Research team time wasted on invalid experiments
- 🎓 Student or postdoc project setbacks
- 💼 Grant deliverable delays
Investment in Proper Storage Infrastructure
One-time equipment costs:
- Medical-grade refrigerator: £500-2,000
- Temperature monitoring system: £100-500
- Organization supplies: £50-200
- Total initial investment: £650-2,700
Ongoing operational costs:
- Electricity (minimal, ~£50-100/year)
- Calibration and maintenance: £100-200/year
- Replacement supplies: £50-100/year
- Annual operating cost: £200-400
Return on Investment Calculation
For a laboratory using 10 pen peptides of retatrutide annually (£2,750 value):
- Without proper storage: 20-30% degradation rate = £550-825 annual loss
- With proper storage: <5% degradation rate = £137 annual loss
- Net annual savings: £413-688
- ROI timeline: Infrastructure pays for itself in 1-4 years
Beyond financial considerations, proper storage ensures research integrity—a value that cannot be quantified but is fundamental to scientific advancement.
Regulatory and Compliance Considerations
Research institutions must maintain proper storage documentation to comply with various regulatory frameworks and quality standards.
Good Laboratory Practice (GLP) Requirements
Facilities operating under GLP guidelines must:
- 📋 Maintain detailed storage SOPs
️ Document continuous temperature monitoring
- 🔍 Conduct regular equipment calibration
- 📊 Keep comprehensive inventory records
- ✍️ Log all peptide receipt, storage, and disposal events
Quality Management Systems
ISO-certified laboratories and those following quality management principles should:
- Establish storage validation protocols
- Perform periodic storage condition audits
- Maintain chain-of-custody documentation
- Implement corrective action procedures for storage deviations
- Train personnel on storage requirements with documented competency
Research-Use-Only Compliance
Important regulatory notice: Retatrutide and all peptides from PEPTIDE PRO are strictly for research use only—not for human or animal consumption.
Proper labeling and storage documentation should clearly indicate:
- “For Research Use Only” designation
- Not approved for clinical or therapeutic use
- Appropriate handling by qualified personnel only
- Disposal according to institutional hazardous waste protocols
Frequently Asked Questions About Retatrutide Refrigeration
Can I store lyophilized retatrutide at room temperature?
Yes, short-term storage (up to 30 days) at room temperature below 25°C is generally acceptable for lyophilized retatrutide. However, refrigeration at 2-8°C or freezing at -20°C significantly extends stability for long-term storage beyond one month.
What happens if reconstituted retatrutide is accidentally frozen?
Freezing reconstituted peptides causes irreversible damage. Ice crystals disrupt the three-dimensional protein structure, resulting in loss of biological activity. If accidentally frozen, the peptide should be discarded and replaced with fresh material.
How long can reconstituted retatrutide stay at room temperature?
Reconstituted retatrutide should be minimized at room temperature—ideally less than 15 minutes during preparation and administration. Extended room temperature exposure (>2 hours) can promote bacterial growth and accelerate peptide degradation.
Does retatrutide need special packaging during summer shipping?
Yes, during warm weather months, temperature-controlled packaging becomes even more critical. PEPTIDE PRO adjusts packaging protocols seasonally, using additional insulation and cooling elements to maintain appropriate temperatures during transit.
Can I use retatrutide past the 28-day reconstitution window?
While bacteriostatic water provides some preservation, peptide stability and sterility cannot be guaranteed beyond 28 days after reconstitution. For research validity, fresh reconstitution is recommended for experiments conducted beyond this timeframe.
What’s the best way to transport reconstituted retatrutide between facilities?
Transport reconstituted peptides in insulated containers with ice packs or cooling elements, maintaining 2-8°C throughout transit. Minimize transport time and use validated coolers with temperature monitoring if available.
Should I refrigerate unopened retatrutide pen peptides immediately upon receipt?
For lyophilized retatrutide, immediate refrigeration isn’t critical if you plan to use within 30 days, though it’s beneficial for long-term storage. For reconstituted peptides (if pre-mixed), refrigeration is mandatory immediately upon receipt.
Advanced Storage Strategies for Research Facilities

Sophisticated research operations can implement additional strategies to maximize peptide stability and research efficiency.
Aliquoting for Extended Viability
Aliquoting strategy: Rather than reconstituting large volumes, consider:
- Divide lyophilized powder into smaller portions before reconstitution
- Reconstitute only needed amounts for immediate research needs
- Store remaining lyophilized aliquots at -20°C for extended stability
- Minimize freeze-thaw cycles by using single-use aliquots
Benefits:
- Reduces waste from expired reconstituted peptide
- Maintains fresh material for extended research timelines
- Minimizes contamination risk
- Provides backup material if primary stock degrades
Stability Testing Protocols
Research facilities can implement internal stability monitoring:
- Time-point sampling: Test peptide activity at regular intervals (0, 7, 14, 21, 28 days)
- Storage condition comparison: Evaluate different temperatures and containers
- Analytical methods: HPLC, mass spectrometry, or bioassays
- Documentation: Create stability profiles specific to your storage conditions
Backup Storage Systems
Critical research programs should consider redundancy:
- 🔄 Dual refrigeration units with automatic failover
- 📊 Remote monitoring with 24/7 alerts
- ⚡ Emergency backup power (UPS or generator)
- 🧊 Liquid nitrogen backup for irreplaceable materials
- 📋 Disaster recovery protocols for equipment failure
Environmental and Sustainability Considerations
Responsible peptide storage extends beyond research quality to environmental stewardship.
Energy-Efficient Storage Solutions
Modern refrigeration technology offers improved sustainability:
- 🌱 Energy Star-certified refrigerators reduce electricity consumption
- 🔋 LED lighting in refrigerated units
️ Optimized temperature cycling reduces compressor wear
- 📦 Proper door seals minimize cooling loss
- ♻️ Refrigerant selection with lower global warming potential
Waste Reduction Strategies
Minimize environmental impact through:
- Accurate ordering to reduce expired peptide waste
- Proper aliquoting to maximize usage
- Sharing excess materials with collaborating laboratories
- Documented disposal following hazardous waste protocols
- Recycling pen peptides and packaging materials where appropriate
Sustainable Packaging Practices
PEPTIDE PRO is committed to balancing peptide protection with environmental responsibility through:
- Recyclable insulation materials where possible
- Reusable cold packs (gel-based rather than single-use)
- Right-sized packaging to minimize waste
- Consolidated shipping to reduce carbon footprint
Future Developments in Peptide Storage Technology
The peptide research field continues advancing storage methodologies to improve stability and convenience.
Emerging Storage Technologies
Innovations on the horizon:
- 🧬 Stabilizing excipients: Additives that extend room-temperature stability
- 📦 Smart packaging: Temperature-indicating labels and time-temperature integrators
️ Precision temperature control: Microprocessor-controlled storage with ±0.1°C accuracy
- 📱 IoT monitoring: Smartphone-connected sensors with cloud data logging
- 🧪 Novel formulations: Peptide modifications that enhance inherent stability
Lyophilization Advances
Improved freeze-drying techniques may offer:
- Enhanced powder stability profiles
- Faster reconstitution times
- Reduced moisture sensitivity
- Extended room-temperature viability
- Better preservation of complex modifications
Regulatory Evolution
As peptide therapeutics advance toward clinical applications, storage standards continue evolving:
- More stringent temperature documentation requirements
- Validated storage duration studies
- Standardized stability testing protocols
- Enhanced chain-of-custody tracking
- Blockchain-based authenticity verification
Refrigeration as a Research Investment
Does retatrutide need refrigeration? The comprehensive answer is nuanced but clear: while lyophilized retatrutide offers flexibility for short-term room-temperature storage, refrigeration at 2-8°C significantly extends stability and is mandatory for reconstituted forms. This storage requirement isn’t a burden—it’s an investment in research quality, data integrity, and scientific advancement.
Key Implementation Steps
To ensure optimal retatrutide storage in your research facility:
- ✅ Establish clear protocols for both lyophilized and reconstituted storage
- ✅ Invest in appropriate refrigeration with temperature monitoring
- ✅ Train all personnel on proper handling and storage procedures
- ✅ Document everything from receipt through disposal
- ✅ Regular audits of storage conditions and peptide inventory
- ✅ Source from reputable suppliers like PEPTIDE PRO who prioritize quality
The Broader Impact of Proper Storage
Beyond preserving individual peptide pen peptides, proper storage practices contribute to:
- Scientific reproducibility across experiments and laboratories
- Efficient resource utilization in research budgets
- Accelerated discovery by eliminating storage-related delays
- Professional credibility through rigorous quality standards
- Environmental responsibility by minimizing waste
Next Steps for Your Research
Ready to ensure your peptide research starts with the highest quality materials stored and shipped under optimal conditions?
Explore PEPTIDE PRO’s extensive catalogue featuring:
- Retatrutide in multiple dosage options
- Comprehensive metabolic research peptides
- Growth hormone and muscle support compounds
- Repair and recovery peptides
- All shipped with temperature-appropriate packaging and same-day dispatch
Questions about storage requirements for your specific research needs? Contact our knowledgeable team for personalized guidance on handling, reconstitution, and storage protocols.
Join the community of researchers who trust PEPTIDE PRO for exceptional purity, professional service, and reliable delivery of research-grade peptides across the UK and worldwide. Because when your research depends on peptide integrity, storage isn’t just about refrigeration—it’s about excellence.
References
[1] Jastreboff AM, et al. (2023). Triple-hormone-receptor agonist retatrutide for obesity — a phase 2 trial. New England Journal of Medicine, 389(6), 514-526.
[2] Manning S, et al. (2020). Stability of peptides in dried blood spots. Molecules, 25(17), 3806.
[3] Bhambhani A, et al. (2022). Formulation and stability considerations for peptide therapeutics. Pharmaceutical Research, 39(8), 1687-1709.
[4] European Medicines Agency. (2018). Guideline on the sterilisation of the medicinal product, active substance, excipient and primary container. EMA/CHMP/CVMP/QWP/850374/2015.
[5] World Health Organization. (2020). Technical Report Series, No. 1025: Good storage and distribution practices for medical products.
