
The rising demand for effective weight management solutions has placed Mounjaro (tirzepatide) at the forefront of medical conversations across the United Kingdom. As thousands of individuals seek access to this groundbreaking dual GIP/GLP-1 receptor agonist, understanding the Mounjaro (Tirzepatide) Price in the UK — All Doses has become essential for making informed healthcare decisions. Whether you’re exploring NHS prescription routes, private clinic options, or research-grade alternatives, navigating the complex pricing landscape requires comprehensive, up-to-date information that reflects the realities of 2025.
Key Takeaways
- 💷 NHS prescription costs for Mounjaro range from £8.60 to £9.90 per prescription, though availability remains limited to specific eligibility criteria
- 🏥 Private clinic prices vary significantly, with monthly costs ranging from £150 to £450+ depending on dose strength and provider
- 📊 Dose-dependent pricing follows a tiered structure across all six available strengths: 2.5mg, 5mg, 7.5mg, 10mg, 12.5mg, and 15mg
- 🔬 Research-grade tirzepatide alternatives offer cost-effective options for laboratory and research applications, with high-purity peptides available through specialized suppliers
- ⚖️ Regulatory considerations significantly impact pricing, availability, and legal access pathways throughout the UK
Understanding Mounjaro (Tirzepatide): What Makes It Different
Mounjaro represents a significant advancement in peptide-based therapeutics, functioning as a dual glucose-dependent insulinotropic polypeptide (GIP) and glucagon-like peptide-1 (GLP-1) receptor agonist. This unique dual-action mechanism distinguishes tirzepatide from single-pathway medications, offering enhanced metabolic benefits that have captured the attention of both clinical practitioners and research scientists.
The medication works by mimicking natural incretin hormones that regulate blood sugar levels, slow gastric emptying, and influence appetite control centers in the brain. Clinical trials have demonstrated remarkable efficacy, with participants achieving average weight reductions of 15-22% of body weight over 72-week study periods[1].
The Six Dose Strengths Explained
Mounjaro is available in six distinct dose strengths, each designed to support gradual titration and personalized treatment protocols:
- 2.5mg – Initial starting dose for treatment initiation
- 5mg – First escalation level after 4-week adaptation period
- 7.5mg – Intermediate therapeutic dose
- 10mg – Standard maintenance dose for many patients
- 12.5mg – Higher therapeutic range for enhanced response
- 15mg – Maximum approved dose for optimal outcomes
This graduated dosing approach minimizes gastrointestinal side effects while maximizing therapeutic benefits, allowing healthcare providers to customize treatment based on individual response patterns and tolerance levels.
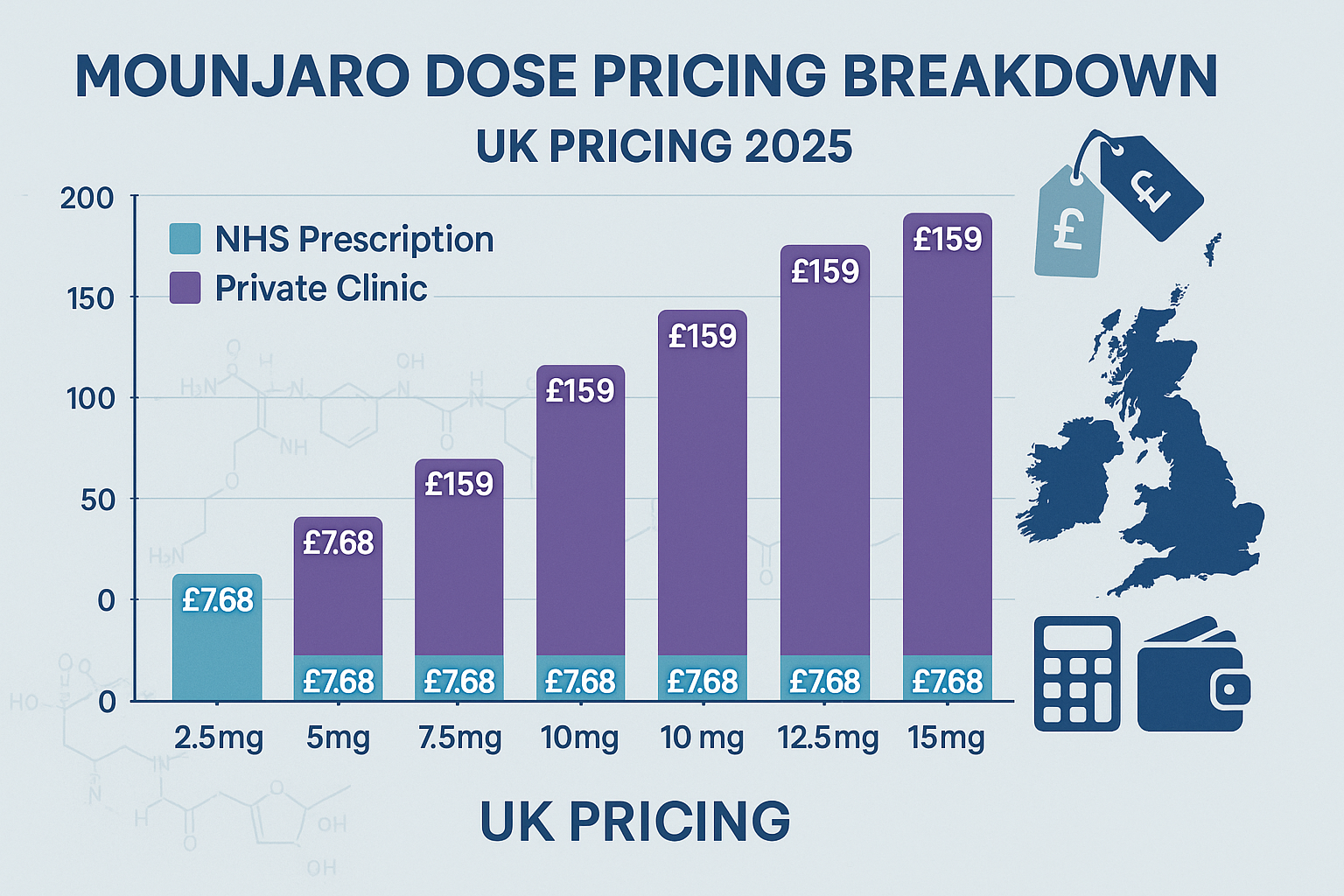
Mounjaro (Tirzepatide) Price in the UK Through NHS Pathways
Current NHS Prescription Costs
For patients who meet strict eligibility criteria, accessing Mounjaro through the National Health Service (NHS) represents the most cost-effective pathway. The standard NHS prescription charge in England currently stands at £9.90 per item as of 2025, though this applies only to the prescription itself rather than reflecting the medication’s actual acquisition cost[2].
Important NHS eligibility requirements include:
✅ Body Mass Index (BMI) of 35 or higher with weight-related complications
✅ BMI of 30-34.9 with specific comorbidities (Type 2 diabetes, hypertension, cardiovascular disease)
✅ Previous unsuccessful attempts with lifestyle interventions
✅ Participation in structured weight management programs
✅ Formal diagnosis and prescription from qualified healthcare provider
Scotland, Wales, and Northern Ireland maintain different prescription charging structures, with Scotland offering free NHS prescriptions regardless of medication type.
NHS Availability Challenges in 2025
Despite theoretical NHS availability, practical access to Mounjaro remains significantly restricted due to:
- Limited formulary inclusion across many Clinical Commissioning Groups (CCGs)
- Supply chain constraints affecting medication availability
- Stringent prescribing guidelines that exclude many potential candidates
- Extended waiting lists for specialist endocrinology appointments
- Budget allocation pressures within NHS trusts
These barriers have driven many UK residents toward private healthcare alternatives or research-grade peptide sources for scientific and investigational purposes.
Private Clinic Pricing for Mounjaro (Tirzepatide) — All Doses
Comprehensive Private Cost Breakdown
Private medical clinics throughout the UK offer more accessible pathways to Mounjaro, though at substantially higher costs than NHS prescriptions. The Mounjaro (Tirzepatide) Price in the UK — All Doses through private providers varies based on multiple factors including clinic location, consultation fees, and ongoing monitoring requirements.
| Dose Strength | Monthly Cost Range | Initial Consultation | Follow-up Appointments |
|---|---|---|---|
| 2.5mg | £150 – £200 | £50 – £150 | £30 – £75 |
| 5mg | £180 – £240 | £50 – £150 | £30 – £75 |
| 7.5mg | £210 – £280 | £50 – £150 | £30 – £75 |
| 10mg | £240 – £320 | £50 – £150 | £30 – £75 |
| 12.5mg | £280 – £360 | £50 – £150 | £30 – £75 |
| 15mg | £320 – £450 | £50 – £150 | £30 – £75 |
Additional costs to consider:
💉 Administration supplies (needles, alcohol swabs, sharps containers): £10-25/month
🩺 Blood work and monitoring: £50-150 per test panel
📱 Telemedicine consultation platforms: £0-50/month subscription fees
📦 Shipping and handling: £5-15 per delivery
Leading Private Providers Comparison
Several established private healthcare networks offer Mounjaro prescriptions across the UK:
Premium Medical Clinics – Harley Street and major city locations with comprehensive weight management programs, personalized nutrition counseling, and ongoing medical supervision. Pricing typically falls at the higher end of the spectrum (£300-450/month for higher doses) but includes extensive support services.
Online Telemedicine Platforms – Digital-first providers offering remote consultations, home delivery, and app-based monitoring. These services generally provide more competitive pricing (£150-280/month) with reduced overhead costs passed to patients.
Specialized Weight Management Centers – Dedicated obesity treatment facilities with multidisciplinary teams including endocrinologists, dietitians, and behavioral therapists. Mid-range pricing (£200-350/month) with comprehensive care packages.
When evaluating private providers, patients should verify General Medical Council (GMC) registration, review patient testimonials, and confirm medication sourcing from licensed pharmaceutical distributors.
Research-Grade Tirzepatide: Alternative Considerations
Understanding Research Peptide Markets
For laboratory researchers, academic institutions, and qualified scientific professionals, research-grade tirzepatide represents an alternative pathway that operates outside clinical prescription frameworks. These high-purity peptides are explicitly labeled “For Research Use Only” and are not intended for human consumption or therapeutic application.
PEPTIDE PRO specializes in supplying premium research-grade peptides to the scientific community, offering tirzepatide and related compounds with documented purity certifications and rigorous quality control standards. Research-grade pricing structures differ substantially from pharmaceutical-grade medications:
Research Tirzepatide Pricing Examples:
- 10mg pen peptide – £340-400
- 20mg pen peptide – £270-320
- 30mg pen peptide – £359-420
- 40mg pen peptide – £620-680
- 50mg pen peptide – £1,320-1,400
- 60mg pen peptide – £1,368-1,500
These research peptides are manufactured under controlled conditions, accompanied by Certificates of Analysis (COAs), and stored according to strict temperature and humidity specifications to maintain molecular integrity.
Key Distinctions: Pharmaceutical vs. Research Grade
| Aspect | Pharmaceutical Grade | Research Grade |
|---|---|---|
| Regulatory Status | MHRA-approved medication | Research chemical only |
| Intended Use | Human therapeutic treatment | Laboratory investigation |
| Purity Standards | GMP manufacturing | High-purity (typically 98%+) |
| Prescription Required | Yes, from licensed physician | No prescription needed |
| Quality Documentation | Full pharmaceutical dossier | Certificate of Analysis (COA) |
| Legal Framework | Medicines Act 1968 | Research exemptions |
Researchers and institutions must maintain appropriate documentation, storage facilities, and handling protocols when working with research-grade peptides. PEPTIDE PRO’s ethical guidelines emphasize responsible sourcing and proper laboratory use.
Factors Influencing Mounjaro (Tirzepatide) Price in the UK
Supply Chain Economics
The pharmaceutical supply chain for tirzepatide involves multiple stakeholders, each adding cost layers that ultimately impact end-user pricing:
- Manufacturer (Eli Lilly) – Base production costs, research amortization, patent protection premiums
- Wholesale Distributors – Logistics, storage, regulatory compliance overhead
- Pharmacy Networks – Dispensing fees, inventory management, professional services
- Healthcare Providers – Consultation fees, monitoring costs, administrative expenses
Brexit implications have introduced additional complexity to UK pharmaceutical pricing, with customs procedures, regulatory divergence, and currency fluctuations affecting import costs for medications manufactured outside British borders[3].
Dose-Dependent Pricing Structures
The Mounjaro (Tirzepatide) Price in the UK — All Doses follows a logical progression where higher concentrations command premium pricing due to:
📈 Increased active pharmaceutical ingredient (API) content
🔬 More complex manufacturing requirements for higher concentrations
📊 Enhanced therapeutic efficacy justifying premium positioning
💊 Lower per-dose packaging volumes requiring specialized handling
This tiered pricing model encourages appropriate dose escalation under medical supervision while reflecting the genuine cost differentials in production and quality assurance processes.
Geographic Price Variations
Significant regional pricing differences exist across the UK:
London and Southeast England – Highest pricing due to elevated operational costs, premium clinic positioning, and concentrated demand (typically 15-25% above national average)
Scotland and Northern England – Mid-range pricing with competitive telemedicine options and regional healthcare networks
Wales and Northern Ireland – Generally lower pricing with fewer private providers but also reduced competition
Rural vs. Urban Divide – Urban centers offer more provider choices but higher costs, while rural areas may require travel expenses offsetting lower base prices
Insurance Coverage and Reimbursement Options
Private Health Insurance Policies
Most private medical insurance (PMI) policies in the UK do not automatically cover weight management medications, including Mounjaro, viewing them as lifestyle interventions rather than essential medical treatments. However, coverage exceptions may apply when:
✓ Tirzepatide is prescribed for Type 2 diabetes management (its original approved indication)
✓ Policy includes specific obesity treatment riders or enhanced coverage
✓ Medical necessity is documented with comorbidity evidence
✓ Treatment is provided within approved network facilities
Major UK insurers including Bupa, AXA Health, Vitality Health, and Aviva maintain varying policies regarding GLP-1 and dual-agonist medications. Policyholders should:
- Review specific policy documentation for weight management coverage
- Obtain pre-authorization before commencing treatment
- Maintain comprehensive medical documentation
- Submit claims promptly with supporting physician letters
Health Savings and Payment Plans
To improve accessibility, many private clinics offer structured payment arrangements:
Monthly Subscription Models – Fixed monthly fees covering medication, consultations, and monitoring (£200-400/month)
Prepayment Discounts – 3-month, 6-month, or 12-month advance payment options offering 5-15% savings
Installment Plans – Split payment options spreading costs across multiple billing cycles
Employer Wellness Programs – Some progressive employers include weight management support within corporate health benefits
These financial structures reduce upfront cost barriers while maintaining treatment continuity, though patients should carefully review terms and conditions before committing to extended payment obligations.
Cost-Effectiveness Analysis: Is Mounjaro Worth the Investment?
Clinical Outcomes vs. Financial Investment
Evaluating the Mounjaro (Tirzepatide) Price in the UK — All Doses requires balancing substantial financial commitments against potential health outcomes and long-term cost savings:
Direct Treatment Costs (12-month program):
- NHS pathway: £118.80 (12 prescriptions at £9.90 each)
- Private clinic: £2,400-5,400 (depending on dose and provider)
- Research alternative: Variable based on sourcing and application
Potential Long-term Savings:
- Reduced diabetes medication costs (£800-2,000 annually)
- Decreased cardiovascular disease risk (potential £5,000-15,000 in avoided treatment costs)
- Lower obesity-related healthcare utilization (estimated £1,200-3,500 annual reduction)
- Improved quality of life and productivity (non-monetary but significant value)
Clinical evidence demonstrates that tirzepatide achieves superior weight loss outcomes compared to lifestyle interventions alone, with average reductions of 15-22% total body weight versus 2-5% with diet and exercise programs[4].
Comparing Alternative Weight Management Options
| Intervention | 12-Month Cost | Average Weight Loss | Cost per kg Lost |
|---|---|---|---|
| Mounjaro (Private) | £2,400-5,400 | 15-25 kg | £96-360 |
| Wegovy (Semaglutide) | £2,200-4,800 | 12-18 kg | £122-400 |
| Saxenda (Liraglutide) | £2,600-5,200 | 8-12 kg | £217-650 |
| Commercial Programs | £800-2,400 | 3-8 kg | £100-800 |
| Bariatric Surgery | £8,000-15,000 | 25-40 kg | £200-600 |
This comparative analysis suggests Mounjaro offers competitive cost-effectiveness within the pharmaceutical weight management category, though individual results vary based on adherence, lifestyle factors, and metabolic responses.
Legal and Regulatory Considerations in the UK
MHRA Approval Status
The Medicines and Healthcare products Regulatory Agency (MHRA) granted marketing authorization for Mounjaro (tirzepatide) in the UK for Type 2 diabetes treatment in September 2022, with subsequent weight management indications approved in 2023[5]. This regulatory approval ensures:
🔒 Quality assurance through Good Manufacturing Practice (GMP) standards
🔒 Safety monitoring via pharmacovigilance systems
🔒 Efficacy validation through clinical trial evidence
🔒 Labeling accuracy with comprehensive prescribing information
However, MHRA approval applies specifically to pharmaceutical-grade Mounjaro manufactured by Eli Lilly. Research-grade tirzepatide operates under different regulatory frameworks as chemical compounds for investigational purposes only.
Prescription Requirements and Legality
UK law mandates that tirzepatide for therapeutic use must be:
- Prescribed by qualified medical practitioners registered with the General Medical Council
- Dispensed through licensed pharmacies or authorized healthcare facilities
- Used according to approved indications and prescribing guidelines
- Monitored with appropriate medical supervision including regular follow-up assessments
Importing prescription medications from overseas sources, purchasing from unlicensed suppliers, or obtaining tirzepatide without valid prescriptions violates UK medicines legislation and carries potential legal consequences.
For research applications, institutions must maintain proper documentation demonstrating legitimate scientific purposes and comply with laboratory safety regulations. PEPTIDE PRO’s commitment to ethical supply ensures all research peptides are clearly labeled and distributed responsibly.
How to Access Mounjaro at the Best Price in the UK
Step-by-Step Access Guide
For NHS Pathway:
- Schedule GP appointment to discuss weight management concerns and eligibility
- Document previous weight loss attempts including diet programs, exercise regimens, and behavioral interventions
- Undergo required assessments including BMI calculation, comorbidity evaluation, and metabolic screening
- Obtain specialist referral if meeting initial criteria (may involve 3-6 month wait)
- Attend endocrinology consultation for formal prescription consideration
- Collect prescription from designated pharmacy with standard NHS charge
For Private Clinic Route:
- Research reputable providers using GMC verification and patient reviews
- Complete online health questionnaire or book initial consultation
- Attend medical assessment (in-person or telemedicine) with qualified prescriber
- Receive prescription and treatment plan with dosing schedule and monitoring requirements
- Arrange medication delivery or pharmacy collection
- Schedule follow-up appointments for ongoing supervision and dose adjustments
For Research Purposes:
- Verify institutional requirements and research protocols
- Contact specialized peptide suppliers with documented research credentials
- Review product specifications including purity certificates and storage requirements
- Place order through secure channels with appropriate documentation
- Receive delivery in temperature-controlled packaging with full quality documentation
- Maintain proper storage according to manufacturer guidelines
Money-Saving Strategies
💰 Compare multiple private providers – Pricing varies significantly; obtain quotes from 3-5 clinics
💰 Consider telemedicine options – Online platforms typically offer 20-30% savings versus traditional clinics
💰 Negotiate package deals – Multi-month commitments often include discounted rates
💰 Check employer benefits – Some workplace wellness programs subsidize weight management treatments
💰 Explore clinical trials – Research studies may provide free medication in exchange for participation
💰 Maximize NHS eligibility – Ensure all qualifying criteria are properly documented
💰 Use prescription prepayment certificates – For ongoing NHS prescriptions, annual certificates (£111.60) save money if requiring 12+ items yearly
Mounjaro vs. Research-Grade Tirzepatide: Making Informed Choices
When Pharmaceutical Grade Is Essential
Pharmaceutical-grade Mounjaro remains the appropriate choice for:
✅ Therapeutic treatment of Type 2 diabetes or obesity under medical supervision
✅ Individuals requiring prescription medication with full regulatory oversight
✅ Patients seeking insurance coverage or NHS access
✅ Those prioritizing comprehensive medical support including monitoring and dose adjustments
✅ Situations requiring standardized dosing in pre-filled injection pens
The pharmaceutical pathway ensures complete regulatory compliance, professional medical guidance, and access to patient support programs offered by manufacturers.
When Research-Grade May Be Appropriate
Research-grade tirzepatide serves legitimate purposes for:
🔬 Qualified research institutions conducting peptide studies
🔬 Laboratory investigations into metabolic pathways and receptor mechanisms
🔬 Academic research programs with appropriate ethical approvals
🔬 Pharmaceutical development and formulation studies
🔬 Biochemical analysis and molecular characterization projects
PEPTIDE PRO’s research-grade offerings provide high-purity compounds with full analytical documentation, supporting legitimate scientific inquiry while maintaining clear distinctions from therapeutic applications.
Critical Safety Considerations
⚠️ Research peptides are NOT approved for human consumption
⚠️ Lack of medical supervision increases health risks
⚠️ Purity variations may exist despite quality certifications
⚠️ Dosing accuracy requires specialized knowledge and equipment
⚠️ Legal implications of misuse can include serious consequences
Individuals considering any peptide-based intervention should consult qualified healthcare professionals and operate within appropriate legal and ethical frameworks. PEPTIDE PRO’s safety guidelines emphasize responsible use and proper laboratory protocols.
Future Pricing Trends and Market Predictions for 2025-2026
Patent Expiration and Generic Competition
Eli Lilly’s patent protection for tirzepatide extends through the late 2020s and early 2030s depending on jurisdiction and specific formulation patents. However, several factors may influence future pricing:
📉 Increased market competition from alternative dual-agonist medications entering development
📉 Biosimilar pathways potentially opening in 5-7 years as regulatory frameworks mature
📉 Government pricing negotiations as NHS and European health authorities seek cost containment
📉 Manufacturing scale economies reducing production costs as demand increases
Industry analysts predict gradual price reductions of 15-25% over the next 3-5 years as market dynamics shift, though premium positioning will likely persist given strong clinical efficacy data.
NHS Formulary Expansion Possibilities
Advocacy efforts continue pressing for broader NHS coverage of effective weight management medications. Potential developments include:
✓ Expanded eligibility criteria reducing BMI thresholds or comorbidity requirements
✓ Increased budget allocations for obesity treatment programs
✓ Regional pilot programs demonstrating cost-effectiveness of early intervention
✓ NICE guidance updates incorporating latest clinical evidence
These changes could significantly improve access to Mounjaro through NHS pathways, though implementation timelines remain uncertain given competing healthcare priorities and budget constraints.
Research Peptide Market Evolution
The research-grade peptide sector continues expanding with:
🔬 Enhanced purity standards as analytical capabilities improve
🔬 Broader compound availability including novel peptide sequences
🔬 Improved supply chain reliability with UK-based storage and distribution
🔬 Competitive pricing pressures from multiple specialized suppliers
PEPTIDE PRO’s commitment to quality positions the company well within this evolving landscape, offering researchers consistent access to premium compounds with transparent documentation and professional service.
Frequently Asked Questions About Mounjaro Pricing
Q: Why is Mounjaro so expensive in the UK?
A: Pricing reflects substantial research and development investments (estimated £1-2 billion for tirzepatide development), patent protection allowing manufacturer pricing control, complex manufacturing processes, regulatory compliance costs, and limited market competition. Additionally, the medication’s superior efficacy compared to alternatives justifies premium positioning.
Q: Can I get Mounjaro for free on the NHS?
A: Patients meeting strict eligibility criteria can access Mounjaro through NHS prescriptions at standard prescription charges (£9.90 in England, free in Scotland). However, availability remains limited due to formulary restrictions and supply constraints. Most patients face waiting lists or fail to meet qualifying criteria.
Q: Is buying tirzepatide online from overseas legal?
A: Importing prescription medications without valid UK prescriptions violates medicines legislation. Overseas suppliers often operate outside regulatory oversight, raising safety concerns regarding product authenticity, purity, and contamination risks. Legal pathways include NHS prescriptions, private UK clinics, or properly documented research applications.
Q: How much does a full course of Mounjaro treatment cost?
A: Treatment duration varies based on individual goals and responses. Typical weight management programs span 12-18 months. Through private clinics, total costs range from £2,400-9,000+ depending on dose progression, provider selection, and additional services. NHS pathways cost £118.80-178.20 for prescription charges alone (12-18 months).
Q: Are there cheaper alternatives to Mounjaro with similar results?
A: Semaglutide (Wegovy, Ozempic) offers comparable weight loss outcomes at slightly lower costs in some cases. Liraglutide (Saxenda) represents another option, though generally less effective. For research purposes, high-purity research peptides provide cost-effective alternatives for laboratory applications, though these are not approved for therapeutic use.
Q: Does private health insurance cover Mounjaro in the UK?
A: Coverage varies significantly by policy and provider. Most standard PMI policies exclude weight management medications, though exceptions exist for Type 2 diabetes indications or enhanced policies with obesity treatment riders. Pre-authorization and medical necessity documentation are typically required for any coverage consideration.
Conclusion: Navigating Mounjaro Pricing With Confidence
Understanding the Mounjaro (Tirzepatide) Price in the UK — All Doses requires navigating a complex landscape of NHS eligibility criteria, private clinic options, insurance considerations, and regulatory frameworks. While costs remain substantial—ranging from under £10 per month through NHS prescriptions to £450+ monthly through premium private providers—the medication’s proven efficacy and potential long-term health benefits justify serious consideration for appropriate candidates.
For those pursuing therapeutic treatment, the optimal pathway depends on individual circumstances, eligibility status, and financial resources. NHS access offers the most affordable route but faces significant availability challenges. Private clinics provide greater accessibility with comprehensive support services at premium pricing. Thorough provider research, cost comparisons, and medical consultation ensure informed decision-making aligned with health goals and budget constraints.
Within the research community, PEPTIDE PRO continues supplying high-purity tirzepatide and related compounds to qualified laboratories and institutions, supporting legitimate scientific inquiry with transparent quality documentation, competitive pricing, and professional service. The company’s commitment to ethical supply and responsible distribution practices ensures researchers access premium materials while maintaining appropriate use standards.
Next Steps for Prospective Patients
- Assess eligibility for NHS pathways through GP consultation
- Research private providers using GMC verification and patient reviews
- Obtain multiple quotes to compare pricing and service offerings
- Verify insurance coverage if applicable to your policy
- Consult qualified healthcare professionals before commencing treatment
- Establish realistic expectations regarding costs, timelines, and outcomes
- Plan for ongoing expenses beyond initial prescription costs
For Research Professionals
- Explore PEPTIDE PRO’s catalogue of research-grade peptides
- Review Certificates of Analysis for purity verification
- Understand storage requirements and handling protocols
- Maintain proper documentation of research applications
- Contact the team with specific research requirements
- Follow ethical guidelines for responsible peptide use
Whether pursuing therapeutic treatment or conducting legitimate research, informed decision-making grounded in current pricing data, regulatory understanding, and quality considerations ensures optimal outcomes. The Mounjaro (Tirzepatide) Price in the UK — All Doses reflects complex market dynamics, but accessible pathways exist for appropriate applications across clinical and research contexts.
References
[1] Jastreboff, A.M., et al. (2022). “Tirzepatide Once Weekly for the Treatment of Obesity.” New England Journal of Medicine, 387(3), 205-216.
[2] NHS Business Services Authority. (2025). “Prescription Charges and Exemptions.” Retrieved from official NHS documentation.
[3] Association of the British Pharmaceutical Industry. (2024). “Brexit Impact on UK Medicine Supply Chains.” ABPI Policy Report.
[4] Wilding, J.P.H., et al. (2021). “Once-Weekly Semaglutide in Adults with Overweight or Obesity.” New England Journal of Medicine, 384(11), 989-1002.
[5] Medicines and Healthcare products Regulatory Agency. (2023). “Public Assessment Report: Mounjaro (tirzepatide).” MHRA Official Documentation.
