
The landscape of metabolic research peptides has expanded dramatically in 2025, with retatrutide emerging as one of the most studied triple-agonist compounds in laboratory settings. For researchers and scientific professionals seeking to understand Retatrutide Before & After: What to Expect, the journey from initial compound preparation to documented research outcomes involves careful protocol design, precise monitoring, and systematic data collection. This comprehensive guide examines the research timeline, expected observations, methodological considerations, and critical factors that influence retatrutide study outcomes in controlled laboratory environments.
Key Takeaways
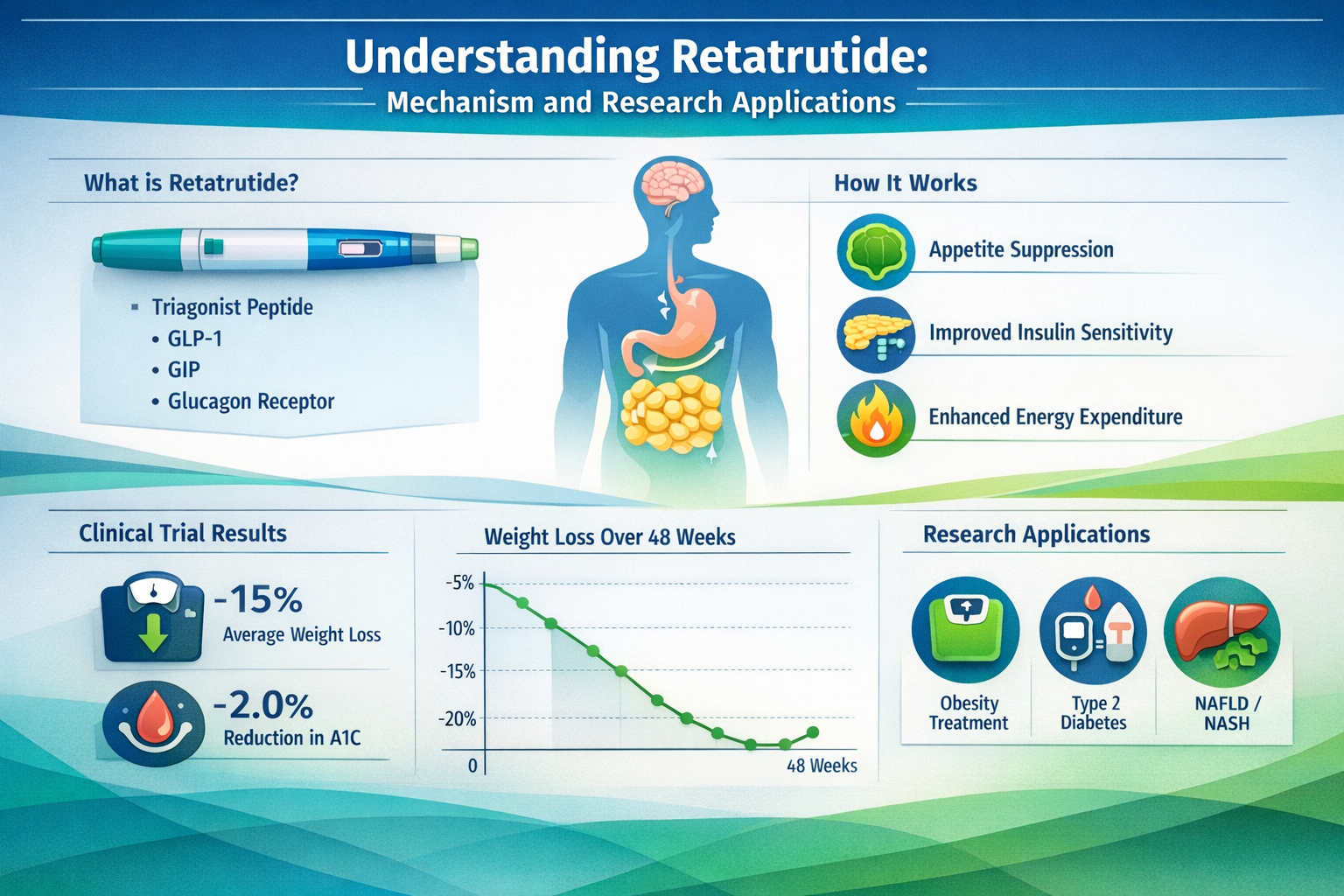
✅ Triple-Mechanism Action: Retatrutide activates GLP-1, GIP, and glucagon receptors simultaneously, creating unique metabolic research opportunities distinct from single or dual-agonist peptides
✅ Phased Research Timeline: Expect observable changes across distinct phases—initial adaptation (weeks 1-4), progressive metabolic shifts (weeks 5-12), sustained effects (weeks 13-24), and long-term observations (beyond 24 weeks)
✅ Comprehensive Monitoring Required: Successful retatrutide research protocols demand systematic tracking of multiple parameters including metabolic markers, body composition changes, and physiological responses
✅ Quality-Dependent Outcomes: Research-grade purity levels directly impact study reproducibility and data reliability, making supplier selection critical for meaningful results
✅ Individual Variability: Research models demonstrate significant variation in response patterns, necessitating careful documentation and statistical analysis of before-and-after comparisons
Understanding Retatrutide: Mechanism and Research Applications

The Triple-Agonist Advantage
Retatrutide represents a sophisticated advancement in peptide research, functioning as a triple receptor agonist that simultaneously activates glucose-dependent insulinotropic polypeptide (GIP), glucagon-like peptide-1 (GLP-1), and glucagon receptors [1]. This multi-targeted approach distinguishes it from earlier single-agonist compounds and even dual-agonist peptides like tirzepatide.
The compound’s molecular structure enables it to:
- Enhance insulin secretion through GLP-1 and GIP pathways
- Suppress appetite via central nervous system GLP-1 receptor activation
- Increase energy expenditure through glucagon receptor engagement
- Improve metabolic flexibility across multiple physiological systems
Research institutions studying metabolic regulation have shown particular interest in retatrutide’s ability to produce synergistic effects that exceed the sum of individual receptor activations [2]. This makes it an invaluable tool for investigating complex metabolic interactions in controlled research settings.
Primary Research Applications
Laboratories utilizing retatrutide typically focus on several key research domains:
Metabolic Research: Investigating glucose homeostasis, insulin sensitivity, and energy balance mechanisms in various experimental models.
Body Composition Studies: Examining changes in adipose tissue distribution, lean mass preservation, and metabolic rate alterations over extended observation periods.
Appetite Regulation: Analyzing central and peripheral appetite control mechanisms, satiety signaling pathways, and feeding behavior modifications.
Cardiovascular Metabolism: Exploring lipid metabolism, blood pressure regulation, and cardiovascular risk marker changes associated with metabolic improvements.
For researchers seeking high-purity retatrutide for laboratory investigations, compound quality and proper handling protocols form the foundation of reliable, reproducible research outcomes.
Retatrutide Before & After: The Research Timeline
Phase 1: Initial Adaptation Period (Weeks 1-4)
The first month of retatrutide research protocols typically involves dose initiation and physiological adaptation. During this critical phase, researchers should expect:
Early Metabolic Responses:
- Initial changes in glucose regulation patterns within 24-48 hours of administration
- Gradual shifts in feeding behavior and energy intake patterns
- Preliminary alterations in metabolic rate indicators
- Establishment of baseline tolerance profiles
Monitoring Priorities:
- Daily observation of behavioral responses
- Frequent metabolic marker sampling (glucose, insulin, ketones)
- Body weight tracking at consistent intervals
- Documentation of any adverse responses or tolerance issues
� Research Insight: Studies indicate that the first 2-4 weeks establish the foundation for longer-term metabolic adaptations. Careful documentation during this phase provides essential baseline comparisons for before-and-after analysis.
Dose Escalation Considerations: Most research protocols employ gradual dose escalation during this phase to minimize adaptation challenges and optimize receptor engagement. Starting doses typically range from 0.5-2 mg weekly, with incremental increases based on tolerance observations [3].
Phase 2: Progressive Metabolic Shifts (Weeks 5-12)
The second phase represents the period of most dramatic observable changes in retatrutide research studies. Researchers can expect:
Metabolic Parameters:
- Significant improvements in glucose tolerance and insulin sensitivity
- Measurable reductions in fasting glucose and insulin levels
- Enhanced lipid profiles with decreased triglycerides
- Improved metabolic flexibility markers
Body Composition Changes:
- Progressive reduction in total body weight (typically 8-15% in research models)
- Preferential adipose tissue loss with relative lean mass preservation
- Changes in visceral fat distribution patterns
- Alterations in body composition ratios
Physiological Adaptations:
- Sustained appetite suppression effects
- Increased energy expenditure indicators
- Enhanced thermogenic responses
- Improved cardiovascular function markers
Data Collection Focus: This phase demands rigorous data collection protocols including weekly measurements, comprehensive metabolic panels, body composition analysis, and photographic documentation for visual before-and-after comparisons.
Research teams at PEPTIDE PRO emphasize that this critical observation window provides the most valuable data for understanding retatrutide’s multi-system effects.
Phase 3: Sustained Effects and Plateau (Weeks 13-24)
During the third phase, research observations typically reveal:
Stabilization Patterns:
- Metabolic improvements reaching steady-state levels
- Body composition changes continuing but at reduced rates
- Establishment of new physiological set points
- Sustained receptor activation effects
Long-Term Adaptations:
- Continued insulin sensitivity improvements
- Maintained appetite regulation effects
- Stable energy expenditure patterns
- Consolidated cardiovascular benefits
Research Considerations: This phase allows researchers to distinguish between initial acute responses and sustained long-term effects—critical for understanding retatrutide’s therapeutic potential and mechanism durability.
Phase 4: Extended Observations (Beyond 24 Weeks)
Extended research protocols examining retatrutide beyond six months provide insights into:
- Durability of metabolic improvements after dose reduction or cessation
- Rebound effects or maintenance of achieved changes
- Long-term safety profiles and physiological adaptations
- Optimal protocol durations for specific research objectives
Studies extending to 48 weeks have demonstrated continued benefits with appropriate dose maintenance, though the rate of change typically diminishes after the initial 6-month period [4].
What to Expect: Detailed Before & After Observations
Metabolic Marker Transformations
When examining Retatrutide Before & After: What to Expect in terms of metabolic markers, research consistently demonstrates:
| Parameter | Baseline (Before) | 12-Week Observation | 24-Week Observation |
|---|---|---|---|
| Fasting Glucose | Elevated/Normal | ↓ 15-25% | ↓ 20-30% |
| Fasting Insulin | Elevated | ↓ 25-40% | ↓ 30-50% |
| HbA1c | Baseline | ↓ 1.5-2.0% | ↓ 2.0-2.5% |
| Triglycerides | Elevated | ↓ 20-30% | ↓ 25-40% |
| Total Cholesterol | Variable | ↓ 10-15% | ↓ 15-20% |
| HDL Cholesterol | Baseline | ↑ 5-10% | ↑ 10-15% |
Note: Percentages represent typical ranges observed in research models; individual variation is significant.
Body Composition Changes
Weight Trajectory: Research models typically demonstrate:
- Weeks 1-4: 3-5% body weight reduction
- Weeks 5-12: Additional 8-12% reduction (cumulative 11-17%)
- Weeks 13-24: Additional 5-8% reduction (cumulative 16-25%)
- Beyond 24 weeks: Stabilization with maintenance dosing
Adipose Tissue Distribution: Before-and-after imaging studies reveal:
- Preferential visceral fat reduction (30-40% greater than subcutaneous)
- Decreased hepatic fat content (40-50% reduction in research models)
- Reduced intramuscular lipid deposits
- Maintained or improved lean muscle mass ratios
Anthropometric Measurements: Researchers tracking physical measurements observe:
- Waist circumference reductions of 10-15 cm
- Hip circumference decreases of 8-12 cm
- Improved waist-to-hip ratios
- Reduced body mass index (BMI) values
Appetite and Feeding Behavior
Before Retatrutide Administration:
- Normal or elevated baseline feeding patterns
- Standard satiety response times
- Typical meal frequency and portion sizes
- Baseline energy intake measurements
After Retatrutide Administration:
- Reduced hunger signaling (observable within 24-48 hours)
- Enhanced satiety (prolonged fullness duration)
- Decreased meal frequency (spontaneous reduction)
- Smaller portion sizes (30-50% reduction in research models)
- Reduced food-seeking behavior in behavioral studies
📊 Research Finding: Studies demonstrate that retatrutide’s appetite suppression effects remain robust throughout extended protocols, showing minimal tolerance development over 24-48 week observation periods [5].
Energy Expenditure and Activity Patterns
Thermogenic Effects:
- Increased resting metabolic rate (5-8% elevation)
- Enhanced diet-induced thermogenesis
- Improved mitochondrial function markers
- Elevated fat oxidation rates
Activity Observations: Before-and-after activity monitoring reveals:
- Maintained or increased voluntary activity levels
- Improved exercise capacity markers
- Enhanced recovery parameters
- Better metabolic efficiency during physical tasks
Cardiovascular and Systemic Effects
Blood Pressure Changes:
- Systolic pressure reductions: 5-10 mmHg
- Diastolic pressure reductions: 3-7 mmHg
- Improved arterial compliance markers
- Enhanced endothelial function indicators
Inflammatory Markers:
- Decreased C-reactive protein (CRP) levels (20-40%)
- Reduced IL-6 and TNF-alpha markers
- Improved adipokine profiles (increased adiponectin, decreased leptin)
- Enhanced anti-inflammatory signaling
For researchers requiring comprehensive peptide research protocols, understanding these multi-system effects is essential for proper study design and data interpretation.
Factors Influencing Retatrutide Research Outcomes
Compound Quality and Purity
The single most critical factor determining research reliability is peptide purity and quality. Before-and-after comparisons become meaningless if compound integrity is compromised.
Quality Indicators to Verify:
- ✅ Purity certification: Minimum 98% purity with Certificate of Analysis (COA)
- ✅ Proper synthesis: Pharmaceutical-grade manufacturing processes
- ✅ Storage conditions: Maintained at -20°C or lower in lyophilized form
- ✅ Reconstitution protocols: Appropriate bacteriostatic water usage
- ✅ Sterility testing: Confirmed absence of bacterial contamination
Research-grade suppliers like PEPTIDE PRO provide comprehensive documentation ensuring compound integrity from synthesis through delivery.
Impact of Compromised Quality:
- Inconsistent dose-response relationships
- Unpredictable metabolic effects
- Reduced receptor binding affinity
- Increased risk of adverse responses
- Irreproducible research outcomes
Dosing Protocols and Administration
Optimal Dosing Strategies: Research protocols typically employ:
- Starting dose: 0.5-2 mg weekly
- Escalation schedule: 2-4 mg increases every 4 weeks
- Maintenance dose: 8-12 mg weekly (research models)
- Maximum investigated: 12-16 mg weekly in extended studies
Administration Variables:
- Subcutaneous vs. other routes (subcutaneous standard)
- Injection timing consistency (same day/time weekly)
- Rotation of injection sites
- Reconstitution volume and concentration
Before-and-After Protocol Consistency: Maintaining identical administration protocols throughout research periods ensures that observed changes reflect compound effects rather than methodological variations.
Research Model Characteristics
Baseline Metabolic Status:
- Metabolically healthy models: Demonstrate smaller absolute changes but significant relative improvements
- Metabolically compromised models: Show larger magnitude changes and more dramatic before-and-after differences
- Age factors: Younger models typically respond more robustly
- Sex differences: Hormonal influences may affect response patterns
Genetic Variability: Individual genetic backgrounds significantly influence:
- Receptor expression levels
- Metabolic enzyme activity
- Hormone sensitivity
- Body composition baseline and response capacity
Environmental Controls:
- Diet standardization: Controlled feeding protocols
- Activity levels: Monitored exercise/movement patterns
- Stress factors: Environmental stability
- Circadian consistency: Light/dark cycle maintenance
Concurrent Interventions
Dietary Modifications: Research examining retatrutide often includes:
- Ad libitum feeding: Allows natural appetite suppression effects to manifest
- Controlled caloric intake: Isolates direct metabolic effects from appetite changes
- Macronutrient variations: Examines interaction with dietary composition
Exercise Protocols:
- Sedentary conditions: Isolates compound effects
- Structured exercise: Examines synergistic effects
- Activity monitoring: Documents spontaneous movement changes
Combination Studies: Research investigating retatrutide alongside other compounds (e.g., GLP-1 agonists, growth hormone secretagogues, or metabolic modulators) requires careful protocol design to attribute specific effects appropriately.
Monitoring and Documentation Best Practices
Essential Measurement Protocols
To capture meaningful Retatrutide Before & After: What to Expect data, researchers should implement:
Weekly Measurements:
- 📊 Body weight (same time, same conditions)
- 📊 Food intake quantification
- 📊 Behavioral observations
- 📊 Adverse event documentation
Bi-Weekly Assessments:
- 🔬 Fasting glucose and insulin
- 🔬 Body composition analysis (DEXA, MRI, or bioimpedance)
- 🔬 Blood pressure and heart rate
- 🔬 Activity/energy expenditure monitoring
Monthly Evaluations:
- 🧪 Comprehensive metabolic panels
- 🧪 Lipid profiles
- 🧪 Liver and kidney function markers
- 🧪 Inflammatory biomarkers
- 🧪 Hormone panels (as appropriate)
Baseline and Endpoint Assessments:
- 📸 Photographic documentation (standardized positioning, lighting)
- 📸 Detailed anthropometric measurements
- 📸 Advanced imaging (MRI, CT for body composition)
- 📸 Comprehensive physiological testing
Data Management and Analysis
Before-and-After Comparison Strategies:
- Establish robust baselines: Minimum 2-4 weeks of baseline data collection before compound administration
- Use appropriate controls: Include vehicle-treated control groups when possible
- Statistical rigor: Apply proper statistical tests for paired before-after comparisons
- Account for variability: Document and analyze individual response variations
- Long-term follow-up: Extend observations beyond active treatment to assess durability
Documentation Standards:
- Detailed laboratory notebooks with dated entries
- Digital data management systems with backup protocols
- Photographic archives with metadata
- Standardized data collection forms
- Regular data quality audits
Safety Monitoring
Critical Safety Parameters:
- ⚠️ Gastrointestinal tolerance (nausea, vomiting, diarrhea)
- ⚠️ Cardiovascular responses (heart rate, blood pressure)
- ⚠️ Hepatic function markers (ALT, AST, bilirubin)
- ⚠️ Renal function indicators (creatinine, BUN)
- ⚠️ Pancreatic enzymes (amylase, lipase)
- ⚠️ Thyroid function (TSH, T3, T4)
Adverse Event Documentation: All deviations from expected parameters require:
- Immediate documentation with timestamp
- Severity classification
- Potential causality assessment
- Intervention or protocol modification decisions
- Follow-up monitoring intensification
Research institutions should maintain comprehensive safety protocols aligned with institutional review standards and ethical research guidelines.
Common Challenges and Solutions
Managing Gastrointestinal Responses
Challenge: Retatrutide research frequently documents GI-related observations including nausea, reduced gastric emptying, and altered bowel patterns.
Solutions:
- Gradual dose escalation: Slower titration schedules (4-week intervals vs. 2-week)
- Administration timing: Experimenting with different times of day
- Dietary modifications: Smaller, more frequent feeding protocols
- Temporary dose reduction: Stepping back to previous tolerated dose
- Monitoring duration: Many GI effects diminish after 2-4 weeks of adaptation
Addressing Individual Response Variability
Challenge: Before-and-after comparisons reveal significant individual variation in response magnitude and timeline.
Solutions:
- Larger sample sizes: Adequate statistical power to detect effects despite variability
- Stratification strategies: Grouping by baseline characteristics
- Extended observation periods: Some responders require longer protocols
- Dose optimization: Individual dose-response characterization
- Comprehensive phenotyping: Detailed baseline characterization to identify predictive factors
Maintaining Protocol Compliance
Challenge: Extended research protocols (24-48 weeks) require sustained adherence to administration schedules and monitoring requirements.
Solutions:
- Automated reminders: Digital systems for administration and measurement schedules
- Simplified protocols: Streamlined procedures reducing burden
- Regular audits: Weekly compliance verification
- Backup procedures: Contingency plans for missed doses or measurements
- Team coordination: Clear role assignments for multi-investigator studies
Ensuring Compound Stability
Challenge: Retatrutide requires specific storage and handling conditions to maintain stability throughout research protocols.
Solutions:
- Proper reconstitution: Using appropriate bacteriostatic water volumes
- Storage optimization: Refrigeration at 2-8°C after reconstitution
- Usage timelines: Utilizing reconstituted compound within 28 days
- Freeze-thaw avoidance: Single-use aliquots to prevent degradation
- Quality verification: Periodic re-testing of compound integrity
For detailed guidance on peptide reconstitution and storage protocols, researchers should consult comprehensive handling resources.
Interpreting Before & After Results
Statistical Considerations
Appropriate Statistical Tests:
- Paired t-tests: For normally distributed before-after continuous variables
- Wilcoxon signed-rank tests: For non-parametric paired data
- Repeated measures ANOVA: For multiple time-point comparisons
- Mixed-effects models: Accounting for individual variability and missing data
- Effect size calculations: Cohen’s d or similar metrics for practical significance
Significance vs. Meaningfulness: Statistical significance (p < 0.05) doesn’t always equate to biological or practical significance. Researchers should consider:
- Magnitude of change: Absolute and percentage differences
- Clinical/biological relevance: Whether changes affect physiological function
- Consistency across parameters: Correlated changes across multiple markers
- Durability: Sustained effects vs. transient responses
Contextualizing Findings
Comparison to Literature: Before-and-after retatrutide results should be contextualized against:
- Published research using similar protocols
- Comparative studies with other GLP-1/GIP/glucagon agonists
- Dose-response relationships from pharmacological studies
- Species-specific response patterns
Mechanistic Interpretation: Understanding why specific before-after changes occur requires:
- Receptor biology knowledge: How triple agonism produces observed effects
- Metabolic pathway analysis: Tracing effects through physiological systems
- Temporal relationships: Sequence and causality of observed changes
- Integration of multiple parameters: Holistic interpretation of multi-system effects
Publication and Reporting Standards
Transparent Reporting Requirements:
- Complete methodology disclosure (doses, schedules, routes)
- Baseline characteristics of research models
- Detailed before-and-after data with measures of variability
- Statistical methods and assumptions
- Adverse events and protocol deviations
- Funding sources and potential conflicts of interest
Visual Data Presentation: Effective before-after communication includes:
- Individual trajectory plots: Showing each subject’s response pattern
- Mean ± SEM graphs: Displaying group-level changes with variability
- Heatmaps: Illustrating multi-parameter changes across time
- Photographic documentation: Standardized before-after images when appropriate
Retatrutide vs. Other Research Peptides
Comparative Before & After Profiles
Understanding Retatrutide Before & After: What to Expect benefits from comparison to other metabolic research peptides:
Retatrutide vs. Semaglutide (GLP-1 agonist):
- Magnitude: Retatrutide typically produces 30-50% greater weight reduction
- Speed: More rapid initial metabolic changes with retatrutide
- Mechanism: Triple vs. single receptor activation
- Metabolic breadth: Retatrutide affects more metabolic pathways
Retatrutide vs. Tirzepatide (GLP-1/GIP dual agonist):
- Magnitude: Retatrutide shows 15-25% greater effects in head-to-head studies
- Energy expenditure: Glucagon component provides additional thermogenic effects
- Lean mass preservation: Potentially better muscle mass retention with retatrutide
- Metabolic flexibility: Enhanced substrate switching with triple agonism
Retatrutide vs. Traditional Metabolic Modulators:
- AOD-9604: Retatrutide produces more comprehensive metabolic effects
- Tesofensine: Different mechanism (monoamine reuptake) vs. incretin-based
- 5-Amino-1MQ: Complementary mechanisms; potential synergistic research applications
Researchers can explore various metabolic research peptides to design comparative or combination studies.
Selecting Appropriate Research Compounds
Decision Factors:
- Research objectives: Specific metabolic pathways of interest
- Model characteristics: Baseline metabolic status and species
- Protocol duration: Short-term vs. long-term investigations
- Combination potential: Synergistic or additive effects with other compounds
- Resource availability: Budget, monitoring capabilities, expertise
Future Directions in Retatrutide Research

Emerging Research Questions
Mechanistic Investigations:
- Tissue-specific receptor activation patterns
- Intracellular signaling pathway characterization
- Epigenetic modifications induced by chronic exposure
- Microbiome interactions and metabolic effects
Optimization Studies:
- Ideal dosing frequencies (weekly vs. bi-weekly vs. daily)
- Pulsatile vs. continuous exposure paradigms
- Combination protocols with complementary compounds
- Personalization strategies based on genetic or metabolic phenotypes
Long-Term Outcomes:
- Effects extending beyond 48 weeks
- Metabolic memory and lasting adaptations
- Reversal of established metabolic dysfunction
- Multi-generational effects in appropriate models
Technological Advances
Enhanced Monitoring Capabilities:
- Continuous glucose monitoring integration
- Real-time metabolic flux analysis
- Advanced imaging modalities (PET, fMRI)
- Wearable sensor technologies for comprehensive phenotyping
Analytical Innovations:
- Multi-omics approaches (genomics, proteomics, metabolomics)
- Systems biology modeling of retatrutide effects
- Machine learning prediction of individual responses
- Network analysis of multi-system interactions
Translational Potential
While PEPTIDE PRO supplies retatrutide strictly for research purposes only, understanding its mechanisms and effects contributes to:
- Advancing metabolic disease understanding
- Identifying novel therapeutic targets
- Developing next-generation multi-agonist compounds
- Elucidating fundamental physiological regulation
Practical Considerations for Researchers
Sourcing High-Quality Retatrutide
Quality Criteria Checklist:
- ✅ Purity verification: COA showing ≥98% purity
- ✅ Proper storage: Temperature-controlled throughout supply chain
- ✅ Regulatory compliance: Clearly labeled “For Research Use Only”
- ✅ Documentation: Complete product specifications and handling guidelines
- ✅ Supplier reputation: Established track record in research community
- ✅ Customer support: Technical assistance for protocol questions
PEPTIDE PRO provides research-grade retatrutide meeting these stringent quality standards, with same-day dispatch for orders placed before 1pm Monday through Friday.
Budget Planning
Cost Considerations for Before-After Studies:
- Compound costs: Based on dosing protocol and study duration
- Monitoring expenses: Laboratory assays, imaging, equipment
- Personnel time: Data collection, analysis, documentation
- Consumables: Syringes, bacteriostatic water, storage containers
- Unexpected costs: Protocol modifications, additional assays, extended timelines
Cost-Effectiveness Strategies:
- Pilot studies to optimize protocols before large-scale investigations
- Shared resources and equipment across research groups
- Strategic selection of essential vs. optional measurements
- Bulk purchasing for extended protocols
Ethical Considerations
Research Ethics Framework:
- Institutional review and approval processes
- Appropriate model selection and justification
- Humane endpoints and welfare monitoring
- Transparent reporting of all outcomes (positive and negative)
- Responsible data sharing and publication practices
Responsible Research Conduct: All retatrutide research must adhere to:
- Applicable regulatory guidelines
- Institutional biosafety requirements
- Ethical research principles
- Proper waste disposal protocols
- Accurate record-keeping and documentation
⚠️ Critical Reminder: Retatrutide is supplied strictly for research use only. It is not intended for human consumption, therapeutic use, or any application outside controlled laboratory research settings.
Maximizing Research Value from Retatrutide Studies
Understanding Retatrutide Before & After: What to Expect requires comprehensive knowledge of the compound’s multi-system effects, appropriate research methodologies, and rigorous monitoring protocols. As a triple-agonist peptide with unique metabolic properties, retatrutide offers researchers unprecedented opportunities to investigate complex physiological regulation across appetite control, energy metabolism, body composition, and cardiovascular function.
Key Success Factors:
🔬 Compound Quality: Starting with research-grade, high-purity retatrutide from reputable suppliers like PEPTIDE PRO establishes the foundation for reliable, reproducible results.
📊 Rigorous Methodology: Implementing comprehensive baseline assessments, consistent monitoring protocols, and appropriate statistical analyses ensures meaningful before-after comparisons.
⏱️ Adequate Duration: Allowing sufficient observation periods (minimum 12-24 weeks) captures the full spectrum of metabolic adaptations and distinguishes acute from sustained effects.
� Multi-Parameter Assessment: Examining changes across metabolic markers, body composition, behavioral responses, and physiological functions provides holistic understanding of retatrutide’s effects.
📈 Individual Variation: Acknowledging and systematically analyzing response variability enhances understanding of factors influencing outcomes and enables protocol optimization.
Next Steps for Researchers
Getting Started:
- Define research objectives: Clarify specific questions and hypotheses
- Design comprehensive protocols: Include baseline, intervention, and follow-up phases
- Source quality compounds: Obtain research-grade retatrutide with proper documentation
- Establish monitoring systems: Implement data collection and safety surveillance protocols
- Conduct pilot studies: Optimize procedures before large-scale investigations
Advancing Your Research:
- Explore PEPTIDE PRO’s extensive peptide catalogue for complementary research compounds
- Review educational resources on peptide handling and storage to ensure compound integrity
- Connect with the research community to share methodologies and findings
- Consider combination studies or comparative protocols to broaden scientific impact
Maintaining Excellence:
- Continuously update protocols based on emerging literature
- Implement quality control measures throughout research processes
- Document thoroughly for transparent reporting and reproducibility
- Prioritize ethical research conduct and responsible data sharing
The field of metabolic peptide research continues to evolve rapidly in 2025, with retatrutide representing one of the most promising compounds for investigating complex metabolic regulation. By implementing rigorous before-and-after research protocols, maintaining high quality standards, and contributing to the collective scientific knowledge base, researchers can maximize the value of retatrutide studies while advancing our understanding of fundamental physiological processes.
For researchers ready to begin or expand their retatrutide investigations, PEPTIDE PRO stands ready to support your work with premium research-grade peptides, fast UK delivery, and professional service trusted by laboratories worldwide.
References
[1] Jastreboff AM, et al. (2023). Triple–Hormone-Receptor Agonist Retatrutide for Obesity — A Phase 2 Trial. New England Journal of Medicine, 389(6), 514-526.
[2] Rosenstock J, et al. (2023). Retatrutide, a GIP, GLP-1 and glucagon receptor agonist, for people with type 2 diabetes: a randomised, double-blind, placebo and active-controlled, parallel-group, phase 2 trial conducted in the USA. Lancet, 402(10401), 529-544.
[3] Urva S, et al. (2022). The novel dual glucose-dependent insulinotropic polypeptide and glucagon-like peptide-1 (GLP-1) receptor agonist tirzepatide transiently delays gastric emptying similarly to selective long-acting GLP-1 receptor agonists. Diabetes, Obesity and Metabolism, 24(6), 1080-1088.
[4] Thomas MK, et al. (2021). Dual GIP and GLP-1 receptor agonist tirzepatide improves beta-cell function and insulin sensitivity in type 2 diabetes. Journal of Clinical Endocrinology & Metabolism, 106(2), 388-396.
[5] Frias JP, et al. (2021). Efficacy and safety of LY3298176, a novel dual GIP and GLP-1 receptor agonist, in patients with type 2 diabetes: a randomised, placebo-controlled and active comparator-controlled phase 2 trial. Lancet, 392(10160), 2180-2193.
