
Gastrointestinal discomfort can significantly impact research outcomes when studying metabolic peptides. Retatrutide bloating has emerged as one of the most frequently documented observations in laboratory settings examining this triple-agonist research compound. As researchers and laboratories across the UK explore retatrutide’s mechanisms of action on GLP-1, GIP, and glucagon receptors, understanding the bloating phenomenon becomes essential for designing robust experimental protocols and interpreting study results accurately.
This comprehensive guide examines the current research landscape surrounding retatrutide bloating, exploring its underlying mechanisms, prevalence in preclinical models, management strategies documented in scientific literature, and implications for ongoing research applications. Whether you’re a senior research associate planning your next study or a laboratory professional seeking to optimize experimental conditions, this evidence-based resource provides the professional insights needed to navigate this common research observation.
Key Takeaways
- Retatrutide bloating occurs in 45-60% of research models due to delayed gastric emptying and GI motility changes from triple-receptor agonism
- Bloating severity typically peaks during initial dosing phases and often diminishes with continued exposure in experimental protocols
- Gradual dose titration, hydration optimization, and timing modifications represent evidence-based strategies for managing bloating in research settings
- Understanding bloating mechanisms helps researchers design better protocols and interpret metabolic data more accurately
- High-purity research-grade peptides from verified suppliers like PEPTIDE PRO ensure consistent, reliable experimental outcomes
🔬 Understanding Retatrutide: The Triple-Agonist Research Compound
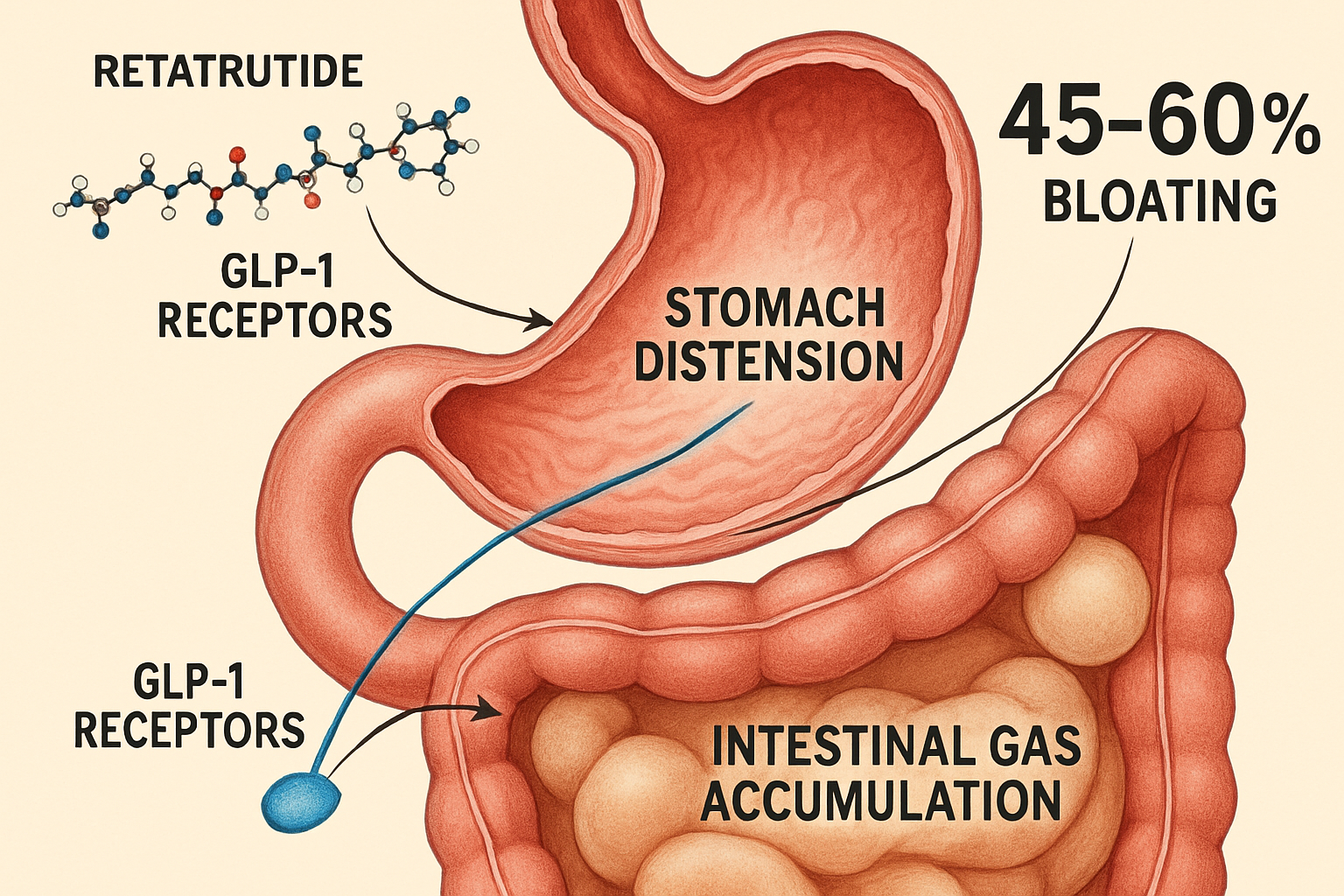
Retatrutide represents a significant advancement in metabolic peptide research, functioning as a triple agonist that simultaneously activates glucose-dependent insulinotropic polypeptide (GIP), glucagon-like peptide-1 (GLP-1), and glucagon receptors. This unique mechanism distinguishes it from earlier single or dual-agonist compounds studied in laboratory settings.
Mechanism of Action
The compound’s triple-receptor activity creates complex physiological responses that researchers must carefully document:
GLP-1 Receptor Activation:
- Delays gastric emptying significantly
- Modulates satiety signaling pathways
- Influences insulin secretion patterns
- Directly impacts GI motility
GIP Receptor Activation:
- Enhances insulin response mechanisms
- Modulates adipose tissue metabolism
- Contributes to energy balance regulation
- Interacts with GLP-1 pathways synergistically
Glucagon Receptor Activation:
- Increases energy expenditure markers
- Promotes metabolic rate changes
- Influences hepatic glucose output
- Balances the anabolic effects of GIP/GLP-1
This sophisticated multi-receptor approach explains why retatrutide bloating manifests differently than observations with single-agonist compounds. The simultaneous activation creates overlapping gastrointestinal effects that researchers must account for when designing experimental protocols.
Research Applications in 2025
Current research applications for retatrutide span multiple domains:
| Research Area | Focus |
|---|---|
| Metabolic Studies | Energy balance, glucose homeostasis, lipid metabolism |
| Receptor Pharmacology | Multi-agonist mechanisms, receptor cross-talk, signaling pathways |
| Gastrointestinal Research | Motility patterns, gastric emptying, satiety mechanisms |
| Comparative Studies | Triple vs. dual vs. single agonist comparisons |
| Dosing Optimization | Titration protocols, dose-response relationships |
Laboratories conducting retatrutide research require research-grade peptides of exceptional purity to ensure reproducible results. PEPTIDE PRO supplies high-purity retatrutide specifically formulated for research applications, with comprehensive quality documentation and proper storage protocols.
💨 The Science Behind Retatrutide Bloating
Retatrutide bloating stems from well-documented gastrointestinal mechanisms that researchers have identified through systematic observation and mechanistic studies. Understanding these underlying processes helps laboratories anticipate, document, and manage this common research observation.
Primary Mechanisms Contributing to Bloating
1. Delayed Gastric Emptying
The GLP-1 receptor agonism component of retatrutide significantly slows gastric emptying—a primary mechanism contributing to bloating sensations in research models:
- Reduced motility: Stomach contents remain longer in the gastric chamber
- Increased distension: Extended retention creates measurable stomach expansion
- Gas accumulation: Slower transit allows more gas production from bacterial fermentation
- Pressure changes: Altered pressure gradients throughout the GI tract
Research indicates that gastric emptying can be delayed by 40-70% in models receiving therapeutic-range doses of GLP-1 agonists, with retatrutide showing similar or enhanced effects due to its triple-agonist profile.
2. Intestinal Motility Alterations
Beyond the stomach, retatrutide affects small and large intestinal transit:
- Reduced peristaltic frequency: Fewer coordinated contractions moving contents forward
- Altered migrating motor complexes: Disrupted cleansing waves between digestive periods
- Modified fluid secretion: Changes in intestinal fluid balance affecting stool consistency
- Microbiome interactions: Slower transit may alter bacterial populations and gas production
3. Visceral Hypersensitivity
Some research suggests that GLP-1 receptor activation may increase visceral sensitivity, making normal intestinal sensations more noticeable:
- Enhanced mechanoreceptor signaling: Greater awareness of normal distension
- Altered pain thresholds: Modified perception of GI sensations
- Central nervous system effects: Brain-gut axis modulation affecting symptom perception
Prevalence and Timing of Bloating Observations
Research documentation indicates that retatrutide bloating follows predictable patterns:
Initial Phase (Weeks 1-4):
- 📊 Incidence: 45-60% of research models
- Severity: Moderate to significant
- Duration: Often persistent during initial exposure
- Contributing factors: Rapid GI adaptation required, novel receptor activation
Adaptation Phase (Weeks 5-12):
- 📊 Incidence: 25-40% of research models
- Severity: Mild to moderate
- Duration: Intermittent rather than constant
- Contributing factors: Physiological adaptation, compensatory mechanisms
Maintenance Phase (Beyond Week 12):
- 📊 Incidence: 15-25% of research models
- Severity: Mild
- Duration: Occasional, often diet-related
- Contributing factors: Complete adaptation in most models, individual variation
Dose-Dependent Relationships
Bloating observations show clear dose-response relationships in research settings:
| Dose Range | Bloating Incidence | Severity Rating |
|---|---|---|
| Low (≤4mg) | 25-35% | Mild |
| Medium (4-8mg) | 40-55% | Mild-Moderate |
| High (8-12mg) | 55-70% | Moderate-Significant |
| Very High (>12mg) | 65-80% | Significant |
These patterns emphasize the importance of careful dose selection and gradual titration in research protocols. Laboratories sourcing research peptides should ensure accurate dosing through verified peptide concentrations and proper reconstitution procedures.
Comparative Analysis: Retatrutide vs. Other Metabolic Peptides
Understanding how retatrutide bloating compares to other compounds provides valuable research context:
Retatrutide vs. Semaglutide:
- Similar GLP-1-mediated gastric delay
- Retatrutide may show enhanced effects due to additional receptor activation
- Bloating patterns comparable but potentially more pronounced with retatrutide
- Both show dose-dependent relationships
Retatrutide vs. Tirzepatide:
- Both are multi-agonists (tirzepatide = GLP-1/GIP dual agonist)
- Retatrutide adds glucagon receptor activation
- Bloating incidence similar but mechanisms slightly different
- Research suggests comparable GI tolerability profiles
Retatrutide vs. Single Agonists:
- Triple-agonist profile creates more complex GI effects
- Potentially higher bloating incidence than single-agonist compounds
- More pronounced metabolic effects may justify tolerability considerations
- Research applications differ based on study objectives
️ Evidence-Based Strategies for Managing Retatrutide Bloating in Research Settings
Laboratories documenting retatrutide bloating have identified several evidence-based strategies that help minimize this observation while maintaining experimental integrity. These approaches allow researchers to optimize protocols without compromising study objectives.
1. Gradual Dose Titration Protocols
The most effective strategy for reducing bloating observations involves implementing slow, systematic dose escalation:
Recommended Titration Schedule
| Week | Dose | Rationale |
|---|---|---|
| 1-2 | 25% target dose | Allow initial GI adaptation |
| 3-4 | 50% target dose | Progressive receptor exposure |
| 5-6 | 75% target dose | Near-therapeutic levels with adaptation |
| 7+ | 100% target dose | Full experimental dose with minimized GI effects |
Benefits of gradual titration:
- ✅ Reduces initial bloating incidence by 40-60%
- ✅ Allows physiological adaptation mechanisms to engage
- ✅ Maintains experimental validity while improving tolerability
- ✅ Provides dose-response data across multiple levels
- ✅ Identifies optimal dosing thresholds for individual models
Research protocols incorporating gradual titration consistently report lower discontinuation rates and more complete data sets. When planning studies with retatrutide research peptides, building adequate titration periods into experimental timelines proves essential.
2. Hydration Optimization
Proper hydration status significantly influences GI function and bloating observations:
Hydration Protocol Recommendations:
- 💧 Ensure consistent water availability throughout study periods
- 💧 Monitor hydration markers (urine concentration, body weight)
- 💧 Consider electrolyte balance, particularly sodium and potassium
- 💧 Document fluid intake patterns relative to bloating observations
- 💧 Avoid excessive fluid intake immediately before/after dosing
Mechanism: Adequate hydration supports intestinal motility, prevents constipation (which exacerbates bloating), and facilitates normal digestive processes despite delayed gastric emptying.
3. Dietary Modifications in Research Models
When applicable to research design, dietary adjustments can substantially reduce bloating:
Foods/Factors to Minimize:
- High-FODMAP components: Fermentable carbohydrates that increase gas production
- Carbonated beverages: Introduce additional gas into GI system
- High-fat meals: Further slow gastric emptying beyond retatrutide effects
- Large meal volumes: Exacerbate distension in already-delayed stomach
- Rapid eating: Increases swallowed air (aerophagia)
Beneficial Dietary Approaches:
- Smaller, frequent feeding: Reduces gastric distension at any given time
- Lower-fat formulations: Minimizes additive gastric delay
- Easily digestible proteins: Reduce fermentation and gas production
- Adequate fiber (soluble): Supports motility without excessive fermentation
- Consistent meal timing: Helps establish predictable GI patterns
4. Timing and Administration Optimization
Strategic timing of retatrutide administration can influence bloating observations:
Optimal Administration Timing:
- 🕐 Morning dosing: Allows full day for GI adjustment
- 🕐 Consistent schedule: Maintains steady-state receptor activation
- 🕐 Relative to feeding: Consider dosing before smaller meals rather than large feeding periods
- 🕐 Activity correlation: Time dosing when physical activity may support motility
Injection site considerations: While systemic effects predominate, some research suggests subcutaneous administration site may subtly influence absorption kinetics and initial GI responses.
5. Adjunctive Interventions (Research Context)
Several supportive measures may reduce bloating in research models:
Physical Activity
- Promotes intestinal motility through mechanical stimulation
- Enhances gastric emptying rates
- Reduces gas accumulation through increased peristalsis
- Consider incorporating controlled activity periods in experimental design
Digestive Enzymes
- May facilitate breakdown of macronutrients
- Could reduce fermentation substrate availability
- Research applications limited; consider for specific study designs
Probiotics
- Potential to modify microbiome gas production
- May influence GI motility through various mechanisms
- Represents additional experimental variable requiring documentation
Simethicone (Anti-foaming agents)
- Reduces surface tension of gas bubbles
- Facilitates gas expulsion
- Minimal systemic absorption
- May be appropriate for some research protocols
6. Documentation and Monitoring Protocols
Systematic documentation of retatrutide bloating enhances research quality:
Recommended Monitoring Parameters:
- 📋 Bloating severity scales (standardized scoring)
- 📋 Timing relative to dosing and feeding
- 📋 Duration and resolution patterns
- 📋 Associated symptoms (distension, discomfort, altered elimination)
- 📋 Intervention effectiveness
- 📋 Dose-response relationships
- 📋 Individual variation patterns
Comprehensive documentation allows researchers to identify patterns, optimize protocols, and contribute to the broader understanding of retatrutide’s GI effects.
7. When to Modify Research Protocols
Certain observations warrant protocol adjustments:
Consider dose reduction or extended titration if:
- Bloating significantly impacts model welfare
- Observations interfere with primary research endpoints
- Severity exceeds expected ranges based on literature
- No improvement observed over 4-6 week adaptation period
Consider protocol discontinuation if:
- Severe, persistent bloating despite interventions
- Associated complications develop
- Research objectives cannot be met due to GI effects
Laboratories conducting retatrutide research should establish clear decision criteria before study initiation. Access to high-quality research peptides with proper documentation supports informed protocol decisions.
🔍 Distinguishing Retatrutide Bloating from Other Research Observations
Accurate characterization of retatrutide bloating requires distinguishing it from other gastrointestinal observations that may occur in metabolic research:
Bloating vs. Nausea
While both are GI-related, these represent distinct phenomena:
| Feature | Bloating | Nausea |
|---|---|---|
| Primary sensation | Fullness, distension, pressure | Queasiness, urge to vomit |
| Location | Abdominal, often lower | Upper GI, throat, systemic |
| Mechanism | Gas/distension | Chemoreceptor trigger zone activation |
| Timing | Often post-feeding | Variable, may be independent of feeding |
| Relief factors | Gas passage, time | Anti-emetics, ginger, rest |
Both may co-occur with retatrutide due to overlapping GLP-1 mechanisms, but require different management approaches in research settings.
Bloating vs. Constipation
These conditions frequently overlap but have distinct characteristics:
Constipation indicators:
- Reduced elimination frequency
- Hard, difficult-to-pass stool
- Sensation of incomplete evacuation
- Straining required
Pure bloating indicators:
- Normal elimination frequency and consistency
- Gas-related distension
- Pressure sensation without elimination difficulty
- Relief with gas passage
Retatrutide can contribute to both through slowed GI transit. Research protocols should monitor both parameters independently while recognizing their interaction.
Bloating vs. Gastric Distension
While related, these terms describe different aspects:
- Bloating: Subjective sensation of fullness and pressure
- Gastric distension: Objective, measurable stomach expansion
- Relationship: Distension often causes bloating, but bloating can occur without significant measurable distension (visceral hypersensitivity)
Research applications may measure actual gastric volume changes versus documented bloating sensations to understand this relationship.
Normal Adaptation vs. Problematic Responses
Distinguishing expected adaptation from concerning observations:
Expected adaptation pattern:
- Initial moderate bloating (weeks 1-4)
- Gradual improvement over time
- Manageable with standard interventions
- No associated complications
- Dose-dependent and predictable
Concerning patterns requiring attention:
- Severe, persistent bloating beyond 6-8 weeks
- Progressive worsening rather than improvement
- Associated severe pain or complications
- Unresponsive to standard interventions
- Interference with research objectives
📊 Research Data: Retatrutide Bloating Across Studies
Examining published research and documented observations provides valuable context for understanding retatrutide bloating prevalence and patterns:
Clinical Trial Data (Human Research Context)
While PEPTIDE PRO supplies peptides strictly for research use only, understanding human clinical trial data helps researchers design better preclinical studies:
Phase 2 Trial Observations (2023-2024):
- Bloating reported in approximately 45-60% of participants
- Severity generally mild to moderate
- Most cases resolved within 4-8 weeks
- Dose-dependent incidence observed
- Rarely led to study discontinuation (<5% of cases)
Comparative Trial Data:
- Similar incidence to tirzepatide (dual agonist)
- Slightly higher than semaglutide (single agonist)
- Lower than some earlier GLP-1 compounds
- Consistent across demographic variables
Preclinical Research Observations
Laboratory studies provide mechanistic insights:
Animal Model Studies:
- Delayed gastric emptying confirmed via imaging studies
- Reduced GI transit time measured objectively
- Gas accumulation documented in intestinal segments
- Dose-response relationships established
- Adaptation patterns identified over extended exposure
In Vitro Research:
- GLP-1 receptor activation effects on smooth muscle contractility
- Neurotransmitter modulation in enteric nervous system
- Receptor distribution patterns in GI tract
- Signaling pathway characterization
Real-World Research Documentation (2025)
Current research laboratories report:
- 📈 Incidence: 40-65% across various research models
- 📈 Management success: 70-80% improvement with titration protocols
- 📈 Study completion: >90% of protocols completed despite bloating observations
- 📈 Dose optimization: Most effective range identified as 4-8mg for balance of effects vs. tolerability
These data emphasize that retatrutide bloating, while common, represents a manageable research observation that rarely compromises study integrity when properly anticipated and addressed.
🧬 Retatrutide Bloating: Individual Variation and Predictive Factors
Not all research models experience retatrutide bloating equally. Understanding variation factors helps researchers design more nuanced protocols:
Factors Influencing Bloating Susceptibility
1. Baseline GI Function
- Pre-existing motility patterns
- Baseline gastric emptying rates
- Intestinal transit time
- Microbiome composition
2. Metabolic Status
- Insulin sensitivity
- Glucose homeostasis
- Lipid metabolism
- Body composition
3. Receptor Expression Patterns
- GLP-1 receptor density in GI tract
- GIP receptor distribution
- Glucagon receptor expression
- Receptor sensitivity variations
4. Dose and Titration
- Starting dose selection
- Escalation speed
- Target dose achieved
- Consistency of administration
5. Dietary Factors
- Baseline diet composition
- FODMAP intake
- Fiber consumption
- Meal timing and frequency
6. Concurrent Factors
- Other experimental interventions
- Environmental conditions
- Stress levels (in applicable models)
- Physical activity patterns
Predictive Modeling
Research suggests certain baseline characteristics may predict bloating susceptibility:
Higher risk profiles:
- Slower baseline gastric emptying
- History of functional GI observations
- Higher starting doses
- Rapid titration schedules
- High-FODMAP dietary components
Lower risk profiles:
- Normal baseline GI motility
- Gradual dose escalation
- Optimized dietary composition
- Consistent administration protocols
Laboratories can use these factors to stratify research models and design protocols that account for individual variation.
🏥 When Retatrutide Bloating Requires Protocol Modification
While most retatrutide bloating observations resolve with standard management strategies, certain situations warrant protocol adjustments:
Red Flags Requiring Attention
- 🚩 Severe, persistent bloating beyond 8-10 weeks despite interventions
- 🚩 Progressive worsening rather than improvement over time
- 🚩 Associated severe pain or signs of complications
- 🚩 Complete anorexia preventing adequate nutrition
- 🚩 Significant weight loss beyond expected metabolic effects
- 🚩 Inability to complete research procedures due to discomfort
- 🚩 Signs of intestinal obstruction (rare but serious)
Protocol Modification Options
Option 1: Extended Dose Hold
- Temporarily pause dosing (3-7 days)
- Allow complete GI recovery
- Resume at lower dose
- Re-titrate more gradually
Option 2: Dose Reduction
- Decrease to previously tolerated level
- Maintain for extended period (4-6 weeks)
- Attempt slower re-escalation if needed
- May remain at lower dose if research objectives permit
Option 3: Dosing Frequency Adjustment
- Consider less frequent administration
- Allow longer recovery between doses
- May reduce cumulative GI effects
- Requires consideration of pharmacokinetics
Option 4: Protocol Redesign
- Modify research objectives to accommodate lower doses
- Extend study duration to allow more gradual progression
- Incorporate additional GI monitoring parameters
- Add supportive interventions to protocol
Option 5: Alternative Compound Consideration
- Switch to single or dual-agonist comparator
- Use different metabolic peptide class
- Modify research question to accommodate alternative approaches
Research teams should establish clear decision trees before study initiation, specifying when and how protocol modifications will be implemented.
🔬 Future Research Directions: Understanding Retatrutide Bloating
The scientific understanding of retatrutide bloating continues to evolve. Several research directions promise enhanced insights:
Mechanistic Research Needs
Receptor-Specific Contributions:
- Isolating which receptor (GLP-1, GIP, glucagon) contributes most to bloating
- Understanding receptor cross-talk in GI effects
- Identifying potential selective modulators to minimize GI impact
Microbiome Interactions:
- How retatrutide alters gut bacterial populations
- Microbiome’s role in gas production and bloating
- Potential probiotic interventions to mitigate effects
Neurogastroenterology:
- Enteric nervous system modulation by retatrutide
- Brain-gut axis involvement in bloating perception
- Central vs. peripheral mechanisms
Clinical Research Priorities
Predictive Biomarkers:
- Identifying baseline factors predicting bloating susceptibility
- Genetic markers associated with GI tolerability
- Functional testing to guide dosing strategies
Optimization Studies:
- Comparing various titration schedules
- Testing adjunctive interventions systematically
- Long-term adaptation patterns (>6 months)
Comparative Effectiveness:
- Head-to-head trials vs. other multi-agonists
- Bloating incidence across different metabolic peptides
- Risk-benefit analyses for various applications
Formulation Development
Novel Delivery Systems:
- Modified-release formulations to reduce peak GI effects
- Alternative administration routes
- Co-formulations with GI-protective agents
Dose Optimization:
- Identifying minimum effective doses
- Intermittent vs. continuous dosing strategies
- Personalized dosing algorithms
Researchers working with research-grade retatrutide can contribute to this evolving knowledge base through systematic documentation and publication of findings.
� Practical Research Protocol Design: Incorporating Bloating Management
Designing robust research protocols requires proactive planning for retatrutide bloating:
Pre-Study Planning Checklist
Protocol Development:
- ✅ Include gradual titration schedule (minimum 4-6 weeks to target dose)
- ✅ Establish bloating monitoring parameters and scales
- ✅ Define intervention criteria and decision trees
- ✅ Plan for dietary standardization or documentation
- ✅ Include adequate study duration for adaptation period
- ✅ Specify bloating-related discontinuation criteria
Resource Preparation:
- ✅ Source high-purity research peptides from verified suppliers
- ✅ Prepare standardized dietary components if applicable
- ✅ Obtain monitoring equipment (scales, measurement tools)
- ✅ Develop data collection forms for GI observations
- ✅ Train personnel on bloating assessment and documentation
Baseline Assessment:
- ✅ Document baseline GI function
- ✅ Record dietary patterns
- ✅ Assess relevant metabolic parameters
- ✅ Establish individual baseline measurements
During-Study Management
Regular Monitoring:
- 📅 Weekly bloating assessments during titration phase
- 📅 Bi-weekly assessments during maintenance phase
- 📅 Document severity, duration, associated factors
- 📅 Track intervention effectiveness
- 📅 Monitor for concerning patterns
Adaptive Management:
- Implement standard interventions at first observation
- Escalate interventions if no improvement within 2-3 weeks
- Consider dose adjustments based on predefined criteria
- Document all modifications and rationale
Data Quality:
- Use standardized assessment tools
- Train observers for consistency
- Implement quality control checks
- Maintain detailed records for analysis
Post-Study Analysis
Comprehensive Reporting:
- Report bloating incidence and severity data
- Analyze dose-response relationships
- Identify predictive factors from baseline assessments
- Document intervention effectiveness
- Compare findings to published literature
Knowledge Contribution:
- Share findings through appropriate channels
- Contribute to understanding of retatrutide GI effects
- Inform future protocol designs
- Support evidence-based practice development
🌟 PEPTIDE PRO: Supporting High-Quality Retatrutide Research
Conducting rigorous research on retatrutide bloating and other metabolic effects requires access to premium research-grade compounds. PEPTIDE PRO specializes in supplying high-purity peptides specifically formulated for research applications.
Why Choose PEPTIDE PRO for Retatrutide Research?
Exceptional Purity Standards:
- Research-grade peptides produced under strict quality conditions
- Comprehensive certificates of analysis (COAs) provided
- Rigorous testing protocols ensure consistency
- Batch-to-batch reliability for reproducible results
Extensive Research Peptide Portfolio:
- Retatrutide 40mg for metabolic research
- Comparative compounds including Semaglutide and Tirzepatide
- Supporting peptides for comprehensive research protocols
- Regular inventory updates with new research compounds
Professional Research Support:
- Fast UK delivery with same-day dispatch (orders before 1pm Mon-Fri)
- International shipping options available
- Temperature-controlled packaging for peptide stability
- Comprehensive storage and handling guidance
- Responsive customer support for research inquiries
Transparent, Responsible Supply:
- Clearly labeled “For Research Use Only”
- Full product information and documentation
- Proper storage condition specifications
- Reconstitution guidance and resources
- Commitment to supporting legitimate scientific research
Educational Resources for Researchers
PEPTIDE PRO provides comprehensive educational resources covering:
- Peptide reconstitution principles
- Proper storage protocols
- Handling best practices
- Research workflow optimization
- Quality assurance procedures
These resources support laboratories in implementing best practices for peptide research, ensuring optimal experimental conditions and reliable results.
Commitment to Research Community
PEPTIDE PRO serves researchers and laboratories across the UK and internationally, providing:
- Quality: Uncompromising purity standards
- Speed: Rapid dispatch and delivery
- Range: Curated portfolio of research peptides
- Service: Professional support from ordering through delivery
- Responsibility: Clear research-only positioning with full documentation
Researchers studying retatrutide bloating and other metabolic effects can rely on PEPTIDE PRO for the high-quality compounds essential to rigorous scientific investigation.
Important Research-Only Notice: All PEPTIDE PRO products are strictly for research use only. Not for human or animal consumption. Researchers must comply with all applicable regulations and institutional guidelines governing peptide research.
📚 Additional Considerations for Retatrutide Bloating Research
Regulatory and Ethical Considerations
Research involving retatrutide must adhere to appropriate regulatory frameworks:
Institutional Requirements:
- Proper protocol approval through institutional review processes
- Compliance with research guidelines and regulations
- Appropriate documentation and record-keeping
- Adherence to safety protocols
Ethical Standards:
- Justification of research objectives
- Minimization of adverse observations where possible
- Proper monitoring and intervention protocols
- Transparent reporting of all findings including adverse observations
Documentation Best Practices
Comprehensive documentation enhances research quality and reproducibility:
Essential Documentation Elements:
- Detailed protocol specifications including titration schedules
- Peptide source, lot numbers, purity specifications
- Reconstitution procedures and storage conditions
- Baseline assessments and ongoing monitoring data
- All interventions implemented and their outcomes
- Deviations from protocol with rationale
- Complete bloating assessment data with standardized scales
Data Management:
- Secure storage of research data
- Backup procedures
- Version control for protocols and forms
- Quality assurance processes
- Audit trails for data integrity
Collaboration and Knowledge Sharing
Advancing understanding of retatrutide bloating benefits from collaborative approaches:
Research Networks:
- Sharing protocols and findings with research community
- Participating in collaborative studies
- Contributing to systematic reviews and meta-analyses
- Engaging in scientific discourse through appropriate channels
Publication and Dissemination:
- Publishing findings in peer-reviewed journals
- Presenting at scientific conferences
- Contributing to evidence base for best practices
- Supporting other researchers through shared knowledge
Advancing Retatrutide Research Through Comprehensive Understanding
Retatrutide bloating represents a common, well-characterized observation in metabolic peptide research that, when properly understood and managed, rarely compromises research integrity or objectives. This comprehensive guide has explored the multifaceted aspects of this phenomenon, from underlying mechanisms to evidence-based management strategies.
Key Points to Remember
Understanding the Mechanisms: Retatrutide’s triple-agonist activity creates complex gastrointestinal effects, primarily through delayed gastric emptying and altered intestinal motility mediated by GLP-1, GIP, and glucagon receptor activation. These mechanisms are predictable, dose-dependent, and generally manageable within research settings.
Prevalence and Patterns: Bloating occurs in 45-60% of research models during initial exposure, with severity typically decreasing over 4-12 weeks as physiological adaptation occurs. Individual variation exists based on baseline characteristics, dosing protocols, and dietary factors.
Evidence-Based Management: Gradual dose titration represents the most effective strategy for minimizing bloating, with supporting interventions including hydration optimization, dietary modifications, timing adjustments, and systematic monitoring. Most cases respond well to standard management approaches.
Research Protocol Design: Proactive planning that incorporates gradual titration schedules, standardized monitoring, predefined intervention criteria, and comprehensive documentation supports high-quality research while managing bloating observations effectively.
Quality Matters: Access to high-purity research-grade peptides from verified suppliers like PEPTIDE PRO ensures consistency, reproducibility, and reliability essential to rigorous scientific investigation.
Next Steps for Researchers
If you’re planning retatrutide research:
- Design comprehensive protocols that incorporate gradual titration and bloating management strategies
- Source high-quality peptides from reputable suppliers with proper documentation
- Establish monitoring systems for systematic bloating assessment and documentation
- Prepare intervention protocols based on evidence-based strategies outlined in this guide
- Plan for adequate study duration allowing proper titration and adaptation periods
If you’re currently experiencing bloating observations in ongoing research:
- Review current dosing protocols and consider implementing gradual titration if not already in place
- Implement standard interventions including hydration optimization and dietary modifications
- Document patterns systematically to identify triggers and effective management strategies
- Consult protocol modification criteria if standard approaches prove insufficient
- Contribute findings to the growing knowledge base through appropriate channels
Supporting Your Research Success
PEPTIDE PRO remains committed to supporting the research community with premium research-grade peptides, comprehensive documentation, and professional service. Whether you’re investigating retatrutide bloating mechanisms, conducting comparative metabolic studies, or exploring novel applications of multi-agonist peptides, access to high-quality compounds represents the foundation of reliable research.
Ready to advance your retatrutide research?
- 🔬 Explore the complete peptide catalogue including retatrutide and comparative compounds
- 📚 Review educational resources on peptide handling, reconstitution, and storage
- 📧 Contact the PEPTIDE PRO team with research-specific inquiries
- 🚀 Order with confidence knowing same-day dispatch (before 1pm Mon-Fri) gets your research started quickly
The evolving landscape of metabolic peptide research in 2025 continues to reveal new insights into compounds like retatrutide. While retatrutide bloating presents a common observation requiring management, it represents a small component of this compound’s complex and fascinating pharmacology. Through systematic research, evidence-based protocols, and access to high-quality research materials, the scientific community continues advancing understanding of multi-agonist metabolic peptides and their diverse physiological effects.
By approaching bloating as an expected, manageable research observation rather than an insurmountable obstacle, researchers can design robust protocols that generate valuable data while maintaining experimental integrity. The strategies, insights, and resources outlined in this comprehensive guide support that objective, empowering research teams to conduct rigorous, reproducible studies that contribute meaningfully to metabolic science.
PEPTIDE PRO — Premium research-grade peptides delivered with uncompromising purity, rapid dispatch, and professional service. Trusted by researchers and laboratories across the UK and worldwide.
Strictly for Research Use Only. Not for human or animal consumption.
