
The landscape of metabolic research is evolving rapidly, and among the most promising compounds under investigation in 2025 is retatrutide—a novel triple-agonist peptide that has captured the attention of researchers worldwide. Understanding the Retatrutide Pen: How It Works, Doses & Needle Types is essential for laboratories conducting metabolic and obesity-related studies. This comprehensive guide explores the delivery mechanisms, dosing protocols, and needle specifications that make retatrutide administration precise and reproducible in controlled research settings.
Key Takeaways
- 🔬 Retatrutide is a triple-receptor agonist targeting GIP, GLP-1, and glucagon receptors, making it a unique research compound for metabolic studies
- 💉 Pen delivery systems provide precise, reproducible dosing essential for consistent research protocols and data integrity
- 📊 Dosing typically escalates from 2mg to 12mg in research settings, with protocols varying based on study design and objectives
�
� Needle selection matters: gauge (thickness) and length affect delivery precision, with 30G-32G and 4-8mm being common specifications
- ⚗️ Research-grade purity and proper reconstitution are critical for reliable experimental outcomes
What Is Retatrutide and Why Does It Matter in Research?
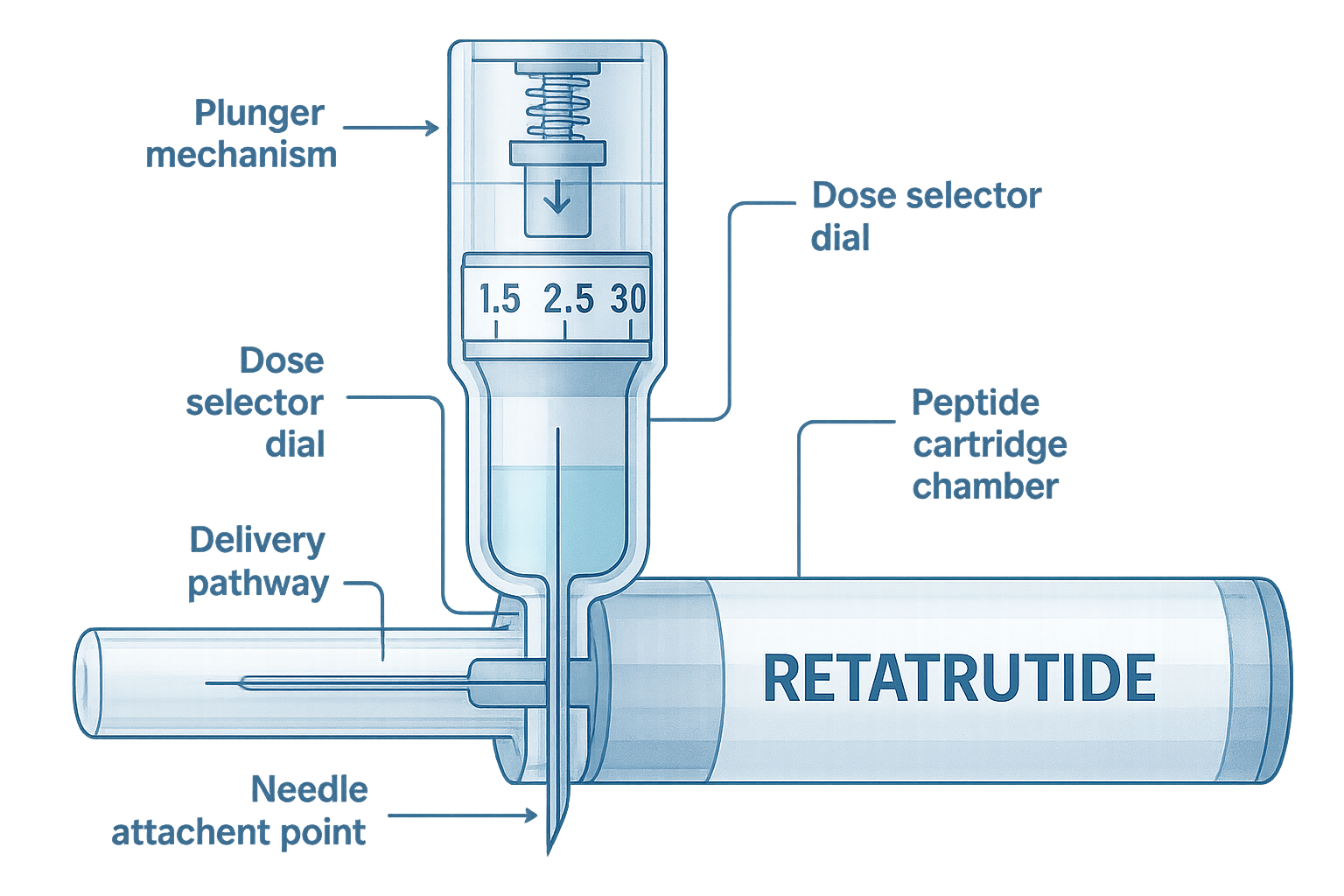
Retatrutide represents a significant advancement in peptide research, particularly in the field of metabolic science. As a triple-receptor agonist, this compound simultaneously activates glucose-dependent insulinotropic polypeptide (GIP), glucagon-like peptide-1 (GLP-1), and glucagon receptors[1]. This unique mechanism of action distinguishes it from single or dual-agonist peptides currently available for research purposes.
The Science Behind Triple Agonism
The multi-receptor targeting approach offers researchers a sophisticated model for studying metabolic regulation. Each receptor pathway contributes distinct physiological effects:
- GIP receptor activation: Influences insulin secretion and lipid metabolism
- GLP-1 receptor activation: Affects satiety signaling and glucose homeostasis
- Glucagon receptor activation: Impacts energy expenditure and metabolic rate
Research institutions studying obesity, diabetes, and metabolic syndrome have shown particular interest in retatrutide’s potential applications. The compound’s ability to engage multiple pathways simultaneously provides a comprehensive research model that more closely mimics complex metabolic interactions[2].
Research Applications and Study Design
Laboratories investigating retatrutide typically focus on:
✅ Metabolic pathway interactions and receptor cross-talk mechanisms
✅ Dose-response relationships across different receptor systems
✅ Pharmacokinetic and pharmacodynamic profiling
✅ Comparative studies with single or dual-agonist compounds
✅ Long-term stability and formulation research
For researchers seeking high-purity research-grade peptides for these investigations, quality and consistency remain paramount considerations.
Understanding the Retatrutide Pen Delivery System
The Retatrutide Pen: How It Works, Doses & Needle Types encompasses sophisticated delivery technology designed for precision and reproducibility—critical factors in research protocols. Pen-based delivery systems have become the gold standard for peptide administration in controlled studies due to their accuracy and ease of standardization.
Components of a Research-Grade Peptide Pen
A typical retatrutide pen system consists of several integrated components:
| Component | Function | Research Importance |
|---|---|---|
| Peptide Cartridge | Contains reconstituted retatrutide solution | Ensures sterility and stability of compound |
| Dose Selector Dial | Allows precise dose adjustment | Critical for dose-escalation protocols |
| Plunger Mechanism | Delivers exact volume when activated | Ensures reproducible dosing across trials |
| Needle Attachment Point | Secures disposable needle | Maintains sterile delivery pathway |
| Dose Window | Displays selected dose | Prevents administration errors |
How the Injection Mechanism Works
The pen delivery system operates through a spring-loaded mechanism that provides consistent pressure during administration. When the dose button is depressed:
- Plunger advances through the cartridge at controlled rate
- Precise volume of solution is expelled through the needle
- Audible/tactile click confirms complete dose delivery
- Mechanism resets for next administration
This standardized delivery method eliminates variables associated with manual syringe filling, reducing protocol variability and enhancing data reliability across research sites.
Advantages for Research Protocols
Research teams utilizing pen systems report several methodological benefits:
🔹 Dosing precision: ±3% accuracy typical for quality pen systems
🔹 Reduced waste: Pre-filled cartridges minimize compound loss
🔹 Protocol standardization: Identical delivery across multiple researchers
🔹 Temperature stability: Cartridge design protects peptide integrity
🔹 Documentation ease: Clear dose windows facilitate accurate record-keeping
For laboratories establishing new retatrutide research protocols, understanding proper handling and storage of these delivery systems is essential for maintaining compound integrity throughout study duration.
Retatrutide Dosing Protocols in Research Settings
When examining Retatrutide Pen: How It Works, Doses & Needle Types, understanding dosing strategies represents a critical component of experimental design. Research protocols typically employ dose-escalation approaches to establish tolerance profiles and dose-response relationships.
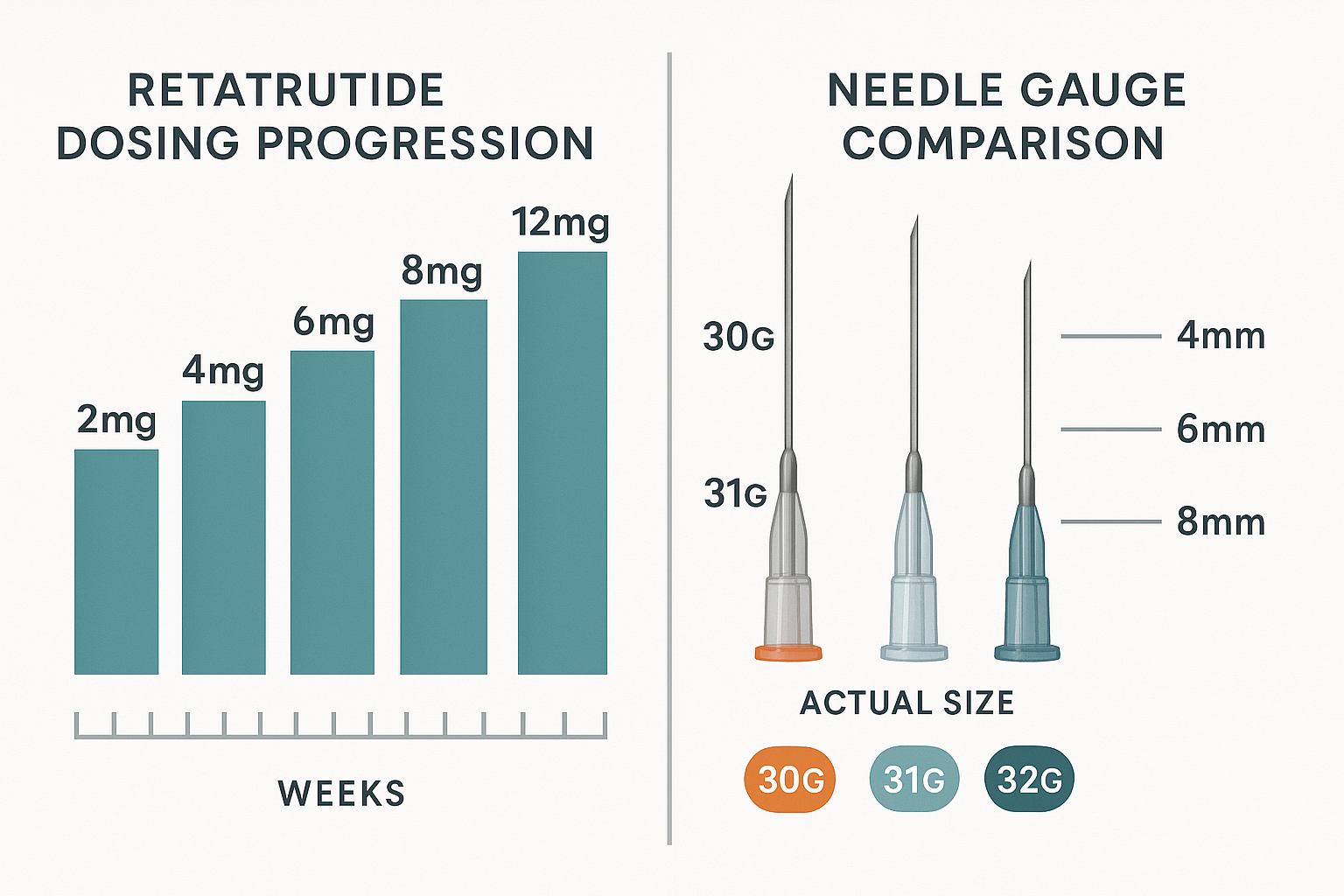
Standard Dose Escalation Framework
Clinical research investigating retatrutide has established a progressive dosing framework that many laboratory studies adapt for their specific objectives[3]:
Initial Phase (Weeks 1-4)
- Starting dose: 2mg administered weekly
- Purpose: Establish baseline tolerance and initial response
Escalation Phase (Weeks 5-20)
- Progressive increases: 4mg → 6mg → 8mg → 12mg
- Timing: Typically 4-week intervals between escalations
- Rationale: Gradual receptor adaptation and response optimization
Maintenance Phase (Week 20+)
- Target dose: 8-12mg depending on study parameters
- Frequency: Weekly administration standard
- Duration: Variable based on research objectives
Dose-Response Considerations
Research teams must carefully consider several factors when establishing dosing protocols:
| Factor | Consideration | Impact on Protocol |
|---|---|---|
| Study Duration | Short-term vs. long-term investigation | Influences escalation speed |
| Research Objectives | Mechanistic vs. outcome-focused | Determines dose range selection |
| Comparative Analysis | Single vs. multiple dose groups | Requires parallel protocol design |
| Sample Characteristics | Baseline metabolic parameters | May necessitate dose adjustment |
Volume and Concentration Calculations
Proper reconstitution is essential for achieving target doses. Research-grade retatrutide typically comes in lyophilized form requiring reconstitution with bacteriostatic water:
Example Calculation for 10mg Vial:
- Reconstitute with 2ml bacteriostatic water = 5mg/ml concentration
- For 4mg dose: 0.8ml injection volume
- For 8mg dose: 1.6ml injection volume
- For 12mg dose: 2.4ml injection volume (requires fresh pen peptide)
Research Note: “Accurate reconstitution and dose calculation are fundamental to reproducible research outcomes. Even minor variations in concentration can significantly impact receptor activation profiles and downstream experimental results.” — Laboratory Protocol Guidelines
Documentation and Protocol Adherence
Maintaining detailed records of dosing protocols ensures research integrity:
✔️ Batch numbers and reconstitution dates
✔️ Exact volumes administered per session
✔️ Time of administration relative to other protocol elements
✔️ Storage conditions between administrations
✔️ Any deviations from standard protocol
Researchers can access comprehensive product information and certificates of analysis to support their documentation requirements and ensure batch-to-batch consistency.
Needle Types and Specifications for Retatrutide Administration
The needle component of Retatrutide Pen: How It Works, Doses & Needle Types significantly influences delivery precision, tissue penetration, and protocol standardization. Selecting appropriate needle specifications is a critical methodological decision in peptide research.
Understanding Needle Gauge
Needle gauge refers to the diameter or thickness of the needle, with higher numbers indicating thinner needles. For retatrutide research administration, three gauges are most commonly employed:
30-Gauge (30G)
- Outer diameter: 0.31mm
- Inner diameter: 0.16mm
- Characteristics: Moderate thickness, reliable flow
- Best for: Higher viscosity solutions or larger volumes
31-Gauge (31G)
- Outer diameter: 0.26mm
- Inner diameter: 0.13mm
- Characteristics: Balanced precision and flow
- Best for: Standard retatrutide protocols, most versatile option
32-Gauge (32G)
- Outer diameter: 0.23mm
- Inner diameter: 0.11mm
- Characteristics: Ultra-fine, minimal tissue disruption
- Best for: Precision protocols requiring minimal trauma
Needle Length Specifications
Needle length determines depth of delivery and tissue targeting. Research protocols typically utilize three standard lengths:
| Length | Penetration Depth | Target Tissue | Research Application |
|---|---|---|---|
| 4mm | Shallow subcutaneous | Superficial adipose | Minimal depth studies |
| 6mm | Mid subcutaneous | Standard adipose | Most common research protocols |
| 8mm | Deep subcutaneous | Deeper adipose layers | Specialized depth studies |
Matching Needle to Protocol Requirements
Selecting the optimal needle specification depends on several research variables:
�
� Solution viscosity: Thicker solutions require larger gauge (lower number) �
� Injection volume: Larger volumes benefit from shorter, wider needles �
� Tissue characteristics: Variable adipose thickness may require length adjustment �
� Frequency of administration: Weekly protocols may prioritize comfort considerations �
� Comparative studies: Standardization across all subjects essential
Needle Compatibility and Pen Systems
Most research-grade pen systems utilize universal thread standards compatible with major needle manufacturers. Key compatibility considerations include:
- Thread type: Typically 8mm outer thread diameter
- Safety features: Some needles include safety shields
- Sterility: Single-use, individually packaged
- Material: Medical-grade stainless steel standard
Technical Insight: “In controlled research settings, needle selection can influence subcutaneous depot formation and subsequent absorption kinetics. Maintaining consistent needle specifications across all study subjects is essential for reducing inter-subject variability in pharmacokinetic parameters.”
Best Practices for Needle Handling
Research protocols should incorporate standardized needle handling procedures:
✅ New needle for each administration to maintain sterility
✅ Proper attachment technique to prevent leakage
✅ Perpendicular insertion angle for consistent depth
✅ Appropriate disposal in sharps containers
✅ Documentation of needle specifications in research records
Laboratories establishing retatrutide research programs can contact specialized suppliers for guidance on optimal needle selection for their specific protocol requirements.
Reconstitution and Preparation Protocols
Proper preparation is fundamental to understanding Retatrutide Pen: How It Works, Doses & Needle Types effectively. Research-grade retatrutide typically arrives in lyophilized (freeze-dried) form, requiring careful reconstitution to maintain peptide integrity and ensure accurate dosing.
Step-by-Step Reconstitution Process
Materials Required:
- Lyophilized retatrutide pen peptide
- Bacteriostatic water (0.9% benzyl alcohol)
- Sterile syringes and needles
- Alcohol swabs
- Clean workspace
Procedure:
- Prepare workspace: Clean surface with 70% isopropanol, allow to dry
- Remove caps: Carefully remove protective caps from both pen peptides
- Sanitize stoppers: Swab rubber stoppers with alcohol, allow to air dry
- Draw bacteriostatic water: Using sterile syringe, draw calculated volume
- Add to peptide pen peptide: Inject water slowly down the side of the pen peptide (never directly onto powder)
- Gentle mixing: Swirl gently in circular motion—never shake vigorously
- Visual inspection: Solution should be clear and colorless
- Transfer to pen cartridge: If using pen system, transfer using sterile technique
Concentration Calculations for Research
Establishing the correct concentration is critical for dose accuracy:
Formula: Concentration (mg/ml) = Total peptide mass (mg) ÷ Reconstitution volume (ml)
Common Research Concentrations:
| Vial Size | Reconstitution Volume | Final Concentration | Dose Range Suitability |
|---|---|---|---|
| 10mg | 2ml | 5mg/ml | 2-8mg doses |
| 20mg | 2ml | 10mg/ml | 8-12mg doses |
| 30mg | 3ml | 10mg/ml | Full dose range |
| 40mg | 4ml | 10mg/ml | Extended protocols |
Storage Requirements Post-Reconstitution
Once reconstituted, retatrutide requires specific storage conditions to maintain stability:
Short-term storage (up to 28 days):
- Temperature: 2-8°C (refrigerated)
- Protection: Keep away from light
- Container: Original pen peptide or sterile pen cartridge
- Monitoring: Regular visual inspection for clarity
Long-term storage (lyophilized):
- Temperature: -20°C or below (frozen)
- Protection: Desiccated environment
- Duration: Typically 12-24 months (check COA)
- Thawing: Gradual at refrigerated temperature
Critical Research Consideration: “Peptide degradation can occur rapidly if storage protocols are not strictly followed. Temperature excursions, even brief ones, can compromise peptide integrity and introduce significant variability into research data. Continuous temperature monitoring is recommended for high-value research programs.”
Quality Control Checkpoints
Implementing quality control measures ensures consistent peptide quality throughout research protocols:
🔬 Visual inspection: Clear, colorless solution without particulates
🔬 pH verification: Should remain within 5.5-7.5 range
🔬 Sterility maintenance: Aseptic technique throughout handling
🔬 Batch documentation: Record all reconstitution details
🔬 Stability testing: Periodic verification of peptide integrity
Research teams can obtain detailed handling and storage guidance specific to their retatrutide batches, including certificates of analysis confirming purity and providing batch-specific recommendations.
Safety Considerations and Research Compliance
When working with Retatrutide Pen: How It Works, Doses & Needle Types, maintaining rigorous safety standards and regulatory compliance is non-negotiable. Research institutions must establish comprehensive safety protocols that protect personnel and ensure ethical research practices.
Laboratory Safety Protocols
Research-grade peptides require handling within controlled environments:
Personal Protective Equipment (PPE):
- Laboratory coat or gown
- Nitrile gloves (latex-free recommended)
- Safety glasses or face shield
- Closed-toe shoes
Environmental Controls:
- Dedicated clean workspace
- Appropriate ventilation
- Temperature-controlled storage
- Restricted access areas
Waste Disposal:
- Sharps containers for needles
- Biohazard bags for contaminated materials
- Chemical waste protocols for unused solutions
- Documentation of all disposal activities
Regulatory Framework for Research Use
Retatrutide and similar research peptides operate under strict regulatory guidelines:
⚖️ Research-only designation: Not approved for human consumption
⚖️ Institutional oversight: IRB/ethics committee approval required
⚖️ Documentation requirements: Comprehensive record-keeping mandatory
⚖️ Import/export regulations: Compliance with international controls
⚖️ Storage security: Controlled substance protocols may apply
Research institutions should review terms and conditions and ethical safety guidelines before initiating retatrutide research programs.
Quality Assurance and Batch Verification
Ensuring peptide quality throughout the research lifecycle requires systematic verification:
Upon Receipt:
- Verify batch number against order
- Inspect packaging for damage
- Confirm temperature during transit
- Review Certificate of Analysis (COA)
- Document storage immediately
During Use:
- Regular visual inspection
- Adherence to storage protocols
- Sterile technique maintenance
- Deviation documentation
- Batch consistency monitoring
Data Integrity:
- Traceability from batch to result
- Complete chain of custody
- Protocol deviation reporting
- Regular audits and reviews
Incident Response Protocols
Research facilities should establish clear procedures for handling incidents:
| Incident Type | Immediate Action | Documentation Required |
|---|---|---|
| Spill | Contain, neutralize, clean | Incident report, disposal record |
| Exposure | First aid, medical evaluation | Exposure report, medical record |
| Temperature excursion | Isolate batch, stability assessment | Temperature log, disposition decision |
| Protocol deviation | Halt procedure, supervisor notification | Deviation report, corrective action |
Ethical Research Practices
Maintaining the highest ethical standards ensures research integrity:
✔️ Transparency: Clear labeling as “Research Use Only”
✔️ Accountability: Comprehensive documentation trails
✔️ Responsibility: Proper training for all personnel
✔️ Compliance: Adherence to institutional and regulatory requirements
✔️ Integrity: Honest reporting of all results and deviations
Researchers can access additional privacy and compliance information to ensure their programs meet all applicable standards.
Comparing Retatrutide to Other Research Peptides
Understanding where retatrutide fits within the broader landscape of metabolic research peptides provides valuable context for Retatrutide Pen: How It Works, Doses & Needle Types and helps researchers make informed decisions about study design and compound selection.
Mechanism Comparison: Single vs. Dual vs. Triple Agonists
The evolution of metabolic peptide research has progressed from single-receptor targeting to increasingly sophisticated multi-agonist approaches:
Single Agonists (GLP-1 Only):
- Examples: Liraglutide, semaglutide
- Mechanism: GLP-1 receptor activation exclusively
- Research applications: Satiety signaling, glucose regulation
- Dosing frequency: Daily to weekly
Dual Agonists (GLP-1/GIP):
- Examples: Tirzepatide
- Mechanism: Combined GLP-1 and GIP receptor activation
- Research applications: Enhanced metabolic effects vs. single agonists
- Dosing frequency: Weekly
Triple Agonist (GLP-1/GIP/Glucagon):
- Example: Retatrutide
- Mechanism: Simultaneous activation of all three receptors
- Research applications: Comprehensive metabolic pathway investigation
- Dosing frequency: Weekly
Comparative Research Outcomes
Studies comparing these different classes reveal distinct profiles:
| Compound Class | Metabolic Impact | Research Complexity | Protocol Considerations |
|---|---|---|---|
| Single agonist | Moderate, focused pathway | Lower, well-characterized | Established protocols available |
| Dual agonist | Enhanced, two-pathway | Moderate, growing literature | Emerging standardization |
| Triple agonist | Maximal, three-pathway | Higher, novel mechanisms | Protocol development ongoing |
Delivery System Considerations
Pen-based delivery systems have become standard across peptide classes, but specifications vary:
Retatrutide advantages:
- Higher potency allows lower volumes
- Weekly dosing simplifies protocols
- Triple mechanism provides comprehensive data
Comparative peptides:
- Some require daily administration
- Volume requirements may differ
- Mechanism specificity varies
Research Portfolio Integration
Many laboratories maintain diverse peptide portfolios for comparative studies. Researchers investigating metabolic pathways often combine:
🔬 Retatrutide: Triple-agonist comprehensive effects
🔬 Tirzepatide: Dual-agonist comparison
🔬 Semaglutide: Single-agonist baseline
🔬 Supporting peptides: BPC-157, TB-500 for tissue effects
Access to a comprehensive research peptide catalogue enables researchers to design robust comparative protocols with consistent quality across all compounds.
Troubleshooting Common Research Challenges
Even with careful attention to Retatrutide Pen: How It Works, Doses & Needle Types, researchers may encounter challenges during protocol implementation. Understanding common issues and their solutions enhances research reliability and data quality.
Reconstitution Issues
Problem: Cloudy solution after reconstitution
Possible causes:
- Vigorous shaking causing protein aggregation
- Contaminated bacteriostatic water
- Peptide degradation due to storage issues
Solutions:
- Always swirl gently, never shake
- Use fresh, sterile bacteriostatic water
- Verify lyophilized peptide storage conditions
- Inspect pen peptide before reconstitution
Problem: Difficulty dissolving powder
Possible causes:
- Insufficient reconstitution volume
- Water added too quickly
- Extremely cold peptide pen peptide
Solutions:
- Verify correct water volume per protocol
- Add water slowly down pen peptide side
- Allow pen peptide to reach room temperature before reconstitution
- Extend gentle swirling time to 2-3 minutes
Pen System Challenges
Problem: Dose selector difficult to turn
Possible causes:
- Cartridge not properly seated
- Mechanism jamming
- Temperature-related viscosity changes
Solutions:
- Remove and reseat cartridge
- Ensure pen is at room temperature before use
- Check for visible damage to mechanism
- Replace pen if persistent
Problem: Solution leaking from needle attachment
Possible causes:
- Needle not fully secured
- Damaged thread on pen or needle
- Overfilled cartridge
Solutions:
- Ensure needle is screwed completely tight
- Inspect threads for damage
- Verify cartridge fill volume
- Replace damaged components
Dosing Accuracy Concerns
Problem: Inconsistent volumes delivered
Possible causes:
- Air bubbles in cartridge
- Incomplete dose button depression
- Needle gauge too small for solution viscosity
Solutions:
- Prime pen before first use (expel air)
- Ensure complete button depression until click
- Consider larger gauge needle if viscosity high
- Verify dose window reading before administration
Problem: Dose window not advancing
Possible causes:
- Mechanical failure
- Empty cartridge
- Frozen mechanism
Solutions:
- Check remaining solution in cartridge
- Verify pen has not been frozen
- Replace pen if mechanism failure suspected
Storage and Stability Issues
Problem: Crystals forming in solution
Possible causes:
- Temperature fluctuations
- Exceeding recommended storage duration
- pH changes in solution
Solutions:
- Maintain consistent refrigeration (2-8°C)
- Adhere to 28-day post-reconstitution limit
- Do not freeze reconstituted solution
- Discard if crystals persist after gentle warming
Problem: Color change in solution
Possible causes:
- Peptide degradation
- Contamination
- Light exposure
Solutions:
- Discard immediately—do not use
- Review storage conditions
- Ensure light protection
- Verify sterile technique throughout handling
Documentation and Protocol Deviations
Problem: Inconsistent results across research subjects
Possible causes:
- Variable needle specifications
- Inconsistent reconstitution procedures
- Different batch sources
- Storage condition variations
Solutions:
- Standardize all needle specifications
- Implement detailed reconstitution SOPs
- Use single batch for entire study when possible
- Implement temperature monitoring systems
Research Best Practice: “Maintaining a detailed troubleshooting log throughout research protocols allows identification of systematic issues and continuous protocol refinement. Many apparent ‘failures’ provide valuable insights into peptide handling requirements and optimal research conditions.”
For persistent technical challenges, researchers should contact specialized support teams who can provide batch-specific guidance and troubleshooting assistance.
Future Directions in Retatrutide Research
The field of triple-agonist peptide research continues to evolve rapidly, with retatrutide representing the cutting edge of metabolic science in 2025. Understanding emerging trends helps researchers position their work within the broader scientific landscape and anticipate future developments in Retatrutide Pen: How It Works, Doses & Needle Types.
Emerging Research Applications
Current research trajectories suggest expanding applications for retatrutide beyond traditional metabolic studies:
Cardiovascular Research:
- Investigating effects on lipid profiles
- Exploring vascular function impacts
- Examining cardiac metabolic remodeling
- Assessing inflammatory marker modulation
Hepatic Research:
- Non-alcoholic fatty liver disease models
- Hepatic glucose metabolism studies
- Lipid accumulation investigations
- Fibrosis progression research
Neurometabolic Research:
- Brain-gut axis investigations
- Cognitive function correlates
- Neuroprotective mechanism studies
- Appetite regulation pathway mapping
Technology Advancement in Delivery Systems
Pen technology continues to advance, with several innovations on the horizon:
Smart Pen Systems:
- Digital dose tracking and logging
- Bluetooth connectivity for data capture
- Temperature monitoring integration
- Automated protocol compliance verification
Microneedle Arrays:
- Painless delivery alternatives
- Enhanced absorption profiles
- Reduced injection frequency potential
- Improved protocol adherence
Controlled-Release Formulations:
- Extended-duration delivery
- Steady-state concentration maintenance
- Reduced administration frequency
- Enhanced pharmacokinetic profiles
Combination Research Strategies
Researchers are increasingly investigating retatrutide in combination protocols:
🧬 With tissue repair peptides: BPC-157, TB-500 for comprehensive metabolic-repair studies
🧬 With cognitive peptides: Exploring metabolic-cognitive connections
🧬 With longevity compounds: Investigating healthspan extension mechanisms
🧬 With exercise mimetics: Understanding synergistic metabolic activation
Precision Research Approaches
The future of retatrutide research lies in increasingly personalized and precise methodologies:
Biomarker-Driven Protocols:
- Baseline metabolic profiling
- Response prediction algorithms
- Individualized dose optimization
- Real-time protocol adjustment
Omics Integration:
- Genomic response profiling
- Proteomic pathway mapping
- Metabolomic signature identification
- Multi-omics data integration
Computational Modeling:
- In silico dose prediction
- Receptor interaction modeling
- Pharmacokinetic simulation
- Outcome prediction algorithms
Regulatory Evolution
The regulatory landscape for research peptides continues to develop:
📋 Enhanced documentation requirements
📋 Standardized purity specifications
📋 International harmonization efforts
📋 Expanded safety reporting systems
📋 Digital compliance platforms
Researchers should stay informed about evolving standards by regularly reviewing updated terms and conditions and regulatory guidance from suppliers.
Collaborative Research Networks
The complexity of triple-agonist research increasingly favors collaborative approaches:
Multi-Center Studies:
- Larger sample sizes
- Diverse population representation
- Protocol standardization across sites
- Enhanced statistical power
Data Sharing Initiatives:
- Open-access databases
- Protocol repositories
- Negative result publication
- Meta-analytic opportunities
Cross-Disciplinary Integration:
- Metabolic + cardiovascular teams
- Basic science + computational modeling
- Pharmaceutical + bioengineering collaboration
- Academic + industry partnerships
Selecting Quality Research-Grade Retatrutide
The foundation of reliable research lies in peptide quality. When considering Retatrutide Pen: How It Works, Doses & Needle Types, the source and quality of the compound itself cannot be overlooked. Selecting appropriate suppliers and verifying peptide specifications are critical research decisions.
Quality Indicators for Research Peptides
High-quality research-grade retatrutide should meet stringent specifications:
Purity Standards:
- Minimum ≥98% purity by HPLC
- Certificate of Analysis (COA) provided with each batch
- Third-party verification available
- Batch-to-batch consistency documented
Physical Characteristics:
- White to off-white lyophilized powder
- Uniform appearance without discoloration
- Appropriate fill weight (±5% tolerance)
- Intact vacuum seal on pen peptides
Documentation Requirements:
- Complete product specifications
- Storage and handling guidelines
- Reconstitution instructions
- Batch-specific COA
- Material Safety Data Sheet (MSDS)
Supplier Evaluation Criteria
Selecting a reliable peptide supplier requires careful evaluation:
| Criterion | What to Look For | Red Flags |
|---|---|---|
| Transparency | Complete product information, accessible support | Vague descriptions, limited contact options |
| Quality Control | COAs provided, testing protocols disclosed | No verification documentation |
| Shipping | Temperature-controlled, tracked delivery | Uncontrolled shipping, no tracking |
| Reputation | Established presence, researcher testimonials | New entity, no verifiable reviews |
| Compliance | Clear “Research Use Only” labeling | Ambiguous marketing, health claims |
Storage and Handling from Receipt
Proper handling begins the moment peptides arrive:
Immediate Actions:
- Verify package temperature (should arrive cold)
- Inspect packaging for damage
- Check batch number against order
- Store immediately at appropriate temperature
- Document receipt date and conditions
Long-Term Management:
- Dedicated peptide storage area
- Temperature monitoring systems
- Inventory tracking system
- First-in, first-out rotation
- Regular quality checks
Cost vs. Quality Considerations
While research budgets matter, quality should never be compromised:
False Economy Indicators:
- Significantly below-market pricing
- No COA or verification available
- Unclear sourcing or manufacturing
- Limited or no customer support
- Inconsistent product availability
Value Indicators:
- Competitive pricing with quality documentation
- Transparent sourcing and testing
- Responsive technical support
- Consistent product availability
- Researcher-focused service model
Research Investment Principle: “The cost of low-quality peptides extends far beyond the initial purchase price. Unreliable compounds lead to failed experiments, wasted time, questionable data, and potentially retracted publications. Quality peptides are research investments, not expenses.”
Building Supplier Relationships
Long-term research success benefits from stable supplier partnerships:
✅ Consistent quality: Same source reduces batch variability
✅ Technical support: Access to expertise for troubleshooting
✅ Priority service: Established customers receive better support
✅ Custom solutions: Tailored products for specific research needs
✅ Regulatory assistance: Help navigating compliance requirements
Researchers can explore comprehensive peptide catalogues from established suppliers committed to research-grade quality and professional service.
Conclusion: Advancing Research with Retatrutide
Understanding Retatrutide Pen: How It Works, Doses & Needle Types represents far more than technical knowledge—it forms the foundation for rigorous, reproducible metabolic research that advances scientific understanding. As we’ve explored throughout this comprehensive guide, successful retatrutide research requires attention to multiple critical factors:
Key Research Success Factors
Compound Quality: Starting with research-grade retatrutide of verified purity (≥98%) ensures data reliability and experimental reproducibility.
Delivery Precision: Pen-based systems provide the accuracy and consistency essential for dose-response studies and comparative protocols.
Dosing Protocols: Following established escalation frameworks (2mg → 12mg) while adapting to specific research objectives creates robust experimental designs.
Needle Selection: Matching gauge (30G-32G) and length (4-8mm) to protocol requirements optimizes delivery consistency and reduces variables.
Handling Excellence: Proper reconstitution, storage, and quality control procedures protect peptide integrity throughout research duration.
Safety Compliance: Maintaining rigorous safety protocols and regulatory compliance ensures ethical research practices and personnel protection.
Actionable Next Steps for Researchers
For laboratories considering or currently conducting retatrutide research:
- Evaluate current peptide sources against quality criteria outlined in this guide
- Review and standardize reconstitution and handling protocols
- Document all specifications including needle types, doses, and storage conditions
- Implement quality control checkpoints at receipt, reconstitution, and throughout use
- Establish supplier relationships with transparent, research-focused providers
- Stay informed about emerging research applications and technological advances
The Future of Triple-Agonist Research
Retatrutide represents the current frontier in multi-receptor metabolic research, but the field continues to evolve rapidly. Researchers who master the technical aspects of Retatrutide Pen: How It Works, Doses & Needle Types position themselves at the forefront of this exciting scientific domain.
The integration of advanced delivery systems, precision dosing protocols, and high-quality research compounds creates unprecedented opportunities for discovery. Whether investigating fundamental metabolic mechanisms, exploring therapeutic potential, or developing novel research models, retatrutide offers a sophisticated tool for scientific advancement.
Partner with Quality for Research Excellence
Success in peptide research requires more than knowledge—it demands reliable access to research-grade compounds, expert support, and commitment to quality at every step.
PEPTIDE PRO delivers the quality, consistency, and professional service that researchers depend on:
🔬 Research-grade purity with comprehensive documentation
🚀 Fast UK delivery with same-day dispatch for orders before 1pm
📋 Complete transparency with COAs and detailed product information
�
� Extensive catalogue including retatrutide and complementary research peptides 💬 Expert support for technical questions and protocol optimization
Ready to Advance Your Retatrutide Research?
Explore PEPTIDE PRO’s comprehensive research peptide catalogue, including high-purity retatrutide and supporting compounds for metabolic research.
For questions about specific research applications, batch availability, or technical guidance, contact the PEPTIDE PRO team for professional, researcher-focused support.
Remember: All products are strictly for research use only. Not for human or animal consumption.
References
[1] Jastreboff AM, et al. “Triple-Hormone-Receptor Agonist Retatrutide for Obesity — A Phase 2 Trial.” New England Journal of Medicine. 2023;389(6):514-526.
[2] Rosenstock J, et al. “Retatrutide, a GIP, GLP-1 and glucagon receptor agonist, for people with type 2 diabetes: a randomised, double-blind, placebo and active-controlled, parallel-group, phase 2 trial conducted in the USA.” Lancet. 2023;402(10401):529-544.
[3] Urva S, et al. “LY3437943, a novel triple GIP, GLP-1, and glucagon receptor agonist in people with type 2 diabetes: A phase 1b, multicentre, double-blind, placebo-controlled, randomised, multiple-ascending dose trial.” Lancet. 2022;400(10366):1869-1881.
