Measuring peptide doses accurately can mean the difference between effective research outcomes and wasted materials—yet many researchers struggle with the seemingly simple task of converting milligram (mg) doses to syringe units. When working with tirzepatide in units: converting mg to units (with table), understanding the relationship between concentration, volume, and measurement becomes essential for precise dosing protocols. This comprehensive guide eliminates the confusion surrounding tirzepatide dose conversions, providing researchers with the clarity needed to measure accurately every time.
Whether you’re working with a 2.5 mg/mL pen peptide or a 10 mg/mL concentration, the conversion principles remain consistent—but the actual unit measurements change dramatically. This article breaks down the fundamental conversion formulas, provides ready-to-use reference tables, and explains why concentration matters when translating prescribed mg doses into measurable syringe units for research applications.
Key Takeaways
- 100 units equals 1 mL in all insulin syringes, forming the foundation for all tirzepatide conversions
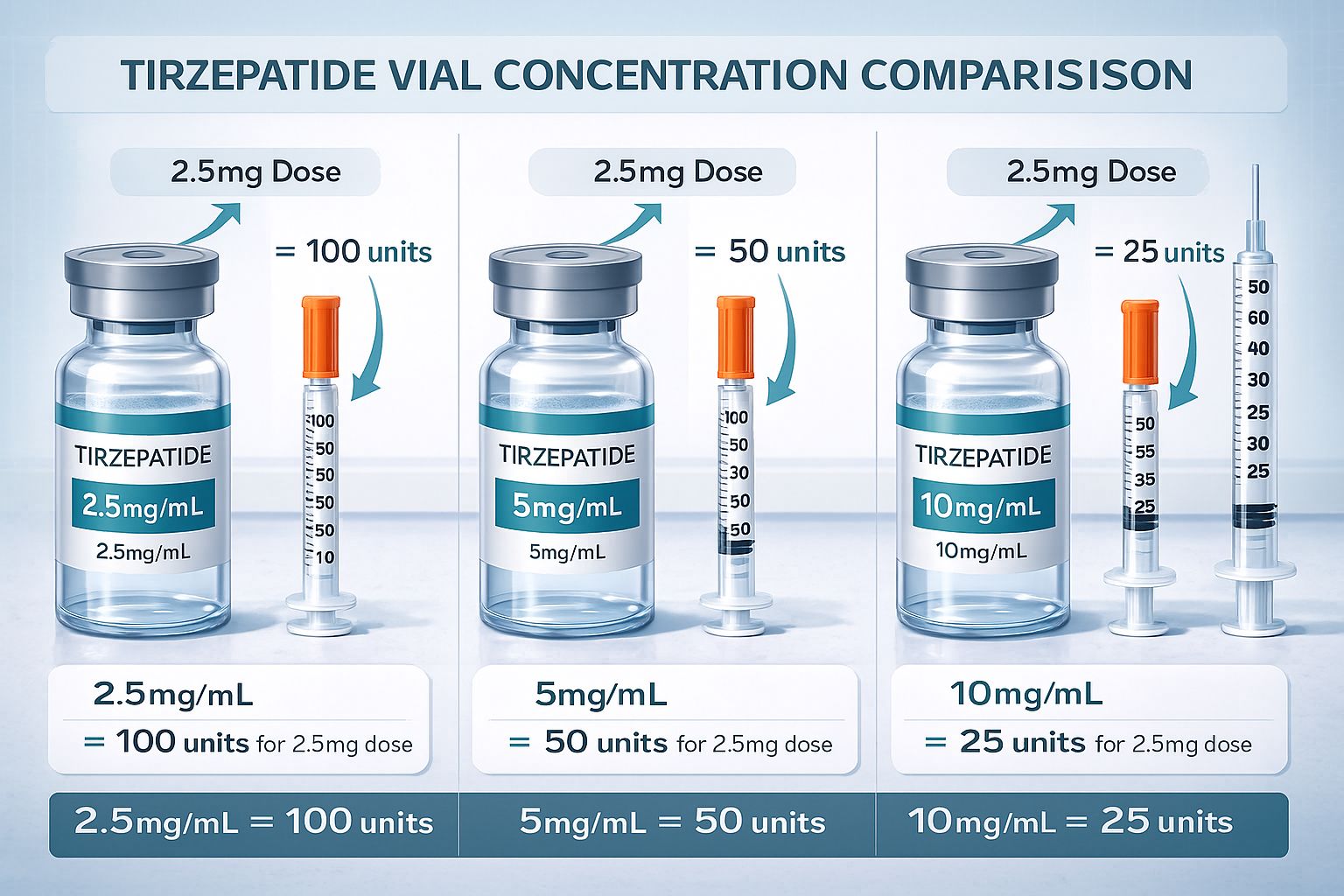
- Vial concentration determines unit measurements: the same 2.5 mg dose requires 100 units at 2.5 mg/mL concentration but only 25 units at 10 mg/mL concentration
- The conversion formula is: (Dose in mg ÷ Vial concentration in mg/mL) × 100 = Units needed
- Always verify pen peptide concentration before calculating doses, as compounded tirzepatide varies significantly between suppliers
- Reference conversion tables eliminate calculation errors and ensure consistent research protocols across multiple dosing sessions
Understanding Tirzepatide Measurement Fundamentals
Tirzepatide, like other research peptides, presents a unique measurement challenge that stems from the intersection of pharmaceutical dosing conventions and practical administration tools. While prescriptions and research protocols specify doses in milligrams (mg)—a measurement of mass—the syringes used for administration measure volume in either milliliters (mL) or units (U). This fundamental disconnect creates the need for accurate conversion calculations.[1]
The Basic Unit-to-Volume Relationship
The cornerstone of all tirzepatide conversions is the standardized relationship between units and milliliters in insulin syringes:
100 units = 1 mL
This ratio remains constant across all insulin syringes, regardless of manufacturer or syringe size. Whether using a 0.3 mL (30 unit), 0.5 mL (50 unit), or 1 mL (100 unit) syringe, this fundamental conversion never changes. Understanding this relationship forms the foundation for all subsequent tirzepatide dose calculations.[2]
Why Concentration Matters
The critical variable that determines how many units correspond to a specific mg dose is the pen peptide concentration, expressed as mg/mL. This concentration tells researchers how many milligrams of active tirzepatide are dissolved in each milliliter of solution. Common tirzepatide concentrations include:
- 2.5 mg/mL (lower concentration)
- 5 mg/mL (medium concentration)
- 10 mg/mL (higher concentration)
- 12.5 mg/mL (occasionally used)
- 15 mg/mL (less common)
The same dose in milligrams will require vastly different unit measurements depending on concentration. For instance, a 5 mg dose from a 5 mg/mL pen peptide requires 100 units (1 full mL), while the same 5 mg dose from a 10 mg/mL pen peptide requires only 50 units (0.5 mL).
Researchers sourcing peptides from PEPTIDE PRO receive clearly labeled pen peptides with concentration information prominently displayed, ensuring accurate dose calculations from the start.
Reconstitution and Final Concentration
For lyophilized (freeze-dried) tirzepatide peptides, the final concentration depends on both the amount of peptide powder in the pen peptide and the volume of bacteriostatic water used for reconstitution. If a pen peptide contains 10 mg of tirzepatide powder and is reconstituted with 1 mL of bacteriostatic water, the resulting concentration is 10 mg/mL. If that same 10 mg pen peptide is reconstituted with 2 mL of water, the concentration becomes 5 mg/mL.[3]
This flexibility allows researchers to customize concentrations based on their specific dosing requirements and preferred injection volumes. However, it also necessitates careful record-keeping to ensure the correct concentration is used for all subsequent calculations.
The Master Conversion Formula for Tirzepatide in Units
Converting tirzepatide doses from milligrams to syringe units follows a straightforward mathematical formula that works regardless of concentration. Mastering this formula eliminates guesswork and ensures precision across all research applications.
The Universal Conversion Formula
(Dose in mg ÷ Vial concentration in mg/mL) × 100 = Units needed
This formula works in three sequential steps:
- Divide the desired dose (in mg) by the pen peptide concentration (in mg/mL)
- This calculation yields the required volume in mL
- Multiply that mL volume by 100 to convert to units
Step-by-Step Conversion Examples
Example 1: 2.5 mg dose from a 10 mg/mL pen peptide
- Step 1: 2.5 mg ÷ 10 mg/mL = 0.25 mL
- Step 2: 0.25 mL × 100 = 25 units
Example 2: 5 mg dose from a 5 mg/mL pen peptide
- Step 1: 5 mg ÷ 5 mg/mL = 1 mL
- Step 2: 1 mL × 100 = 100 units
Example 3: 7.5 mg dose from a 2.5 mg/mL pen peptide
- Step 1: 7.5 mg ÷ 2.5 mg/mL = 3 mL
- Step 2: 3 mL × 100 = 300 units (requires multiple syringes or a larger barrel syringe)
Example 4: 12.5 mg dose from a 10 mg/mL pen peptide
- Step 1: 12.5 mg ÷ 10 mg/mL = 1.25 mL
- Step 2: 1.25 mL × 100 = 125 units
Alternative Conversion Method: mL to Units
If a research protocol specifies doses in milliliters rather than milligrams, the conversion becomes even simpler:
Volume in mL × 100 = Units
For example:
- 0.5 mL × 100 = 50 units
- 0.75 mL × 100 = 75 units
- 1.5 mL × 100 = 150 units
This method bypasses the concentration variable entirely, as the volume already accounts for the concentration when calculated from the original mg dose.
Common Conversion Mistakes to Avoid
❌ Forgetting to verify pen peptide concentration before calculating—using the wrong concentration yields incorrect unit measurements
❌ Confusing units with mL—remembering that 1 mL = 100 units prevents decimal errors
❌ Rounding prematurely—maintain precision through all calculation steps, rounding only the final unit measurement
❌ Using expired or incorrectly stored pen peptides—degraded peptides may not deliver expected research outcomes regardless of accurate dosing
Researchers working with tirzepatide from PEPTIDE PRO benefit from comprehensive product documentation that includes concentration details and reconstitution guidance, minimizing calculation errors.
Comprehensive Tirzepatide Conversion Tables
Reference tables eliminate the need for repeated calculations and reduce the risk of mathematical errors during time-sensitive research protocols. The following tables cover the most common tirzepatide concentrations and doses used in research settings.
Table 1: Tirzepatide Conversions for 10 mg/mL Concentration
| Dose (mg) | Volume (mL) | Units |
|---|---|---|
| 2.5 mg | 0.25 mL | 25 units |
| 5 mg | 0.5 mL | 50 units |
| 7.5 mg | 0.75 mL | 75 units |
| 10 mg | 1.0 mL | 100 units |
| 12.5 mg | 1.25 mL | 125 units |
| 15 mg | 1.5 mL | 150 units |
This concentration is commonly used for higher-dose protocols, allowing for smaller injection volumes.
Table 2: Tirzepatide Conversions for 5 mg/mL Concentration
| Dose (mg) | Volume (mL) | Units |
|---|---|---|
| 2.5 mg | 0.5 mL | 50 units |
| 5 mg | 1.0 mL | 100 units |
| 7.5 mg | 1.5 mL | 150 units |
| 10 mg | 2.0 mL | 200 units |
| 12.5 mg | 2.5 mL | 250 units |
| 15 mg | 3.0 mL | 300 units |
This mid-range concentration balances injection volume with dosing flexibility.
Table 3: Tirzepatide Conversions for 2.5 mg/mL Concentration
| Dose (mg) | Volume (mL) | Units |
|---|---|---|
| 2.5 mg | 1.0 mL | 100 units |
| 5 mg | 2.0 mL | 200 units |
| 7.5 mg | 3.0 mL | 300 units |
| 10 mg | 4.0 mL | 400 units |
| 12.5 mg | 5.0 mL | 500 units |
| 15 mg | 6.0 mL | 600 units |
Lower concentrations require larger injection volumes but offer greater precision for titration protocols.
Table 4: Quick Reference Multi-Concentration Comparison
| Dose | 2.5 mg/mL | 5 mg/mL | 10 mg/mL | 12.5 mg/mL |
|---|---|---|---|---|
| 2.5 mg | 100 units | 50 units | 25 units | 20 units |
| 5 mg | 200 units | 100 units | 50 units | 40 units |
| 10 mg | 400 units | 200 units | 100 units | 80 units |
| 15 mg | 600 units | 300 units | 150 units | 120 units |
This comparison table demonstrates how dramatically concentration affects unit measurements for identical mg doses.
Using Conversion Tables in Research Protocols
When establishing research protocols, creating a customized conversion table for your specific pen peptide concentration streamlines the dosing process and ensures consistency across multiple administration sessions. Researchers should:
✅ Print and laminate concentration-specific tables for laboratory reference
✅ Label each pen peptide with its concentration and date of reconstitution
✅ Create dosing logs that record both mg doses and corresponding unit measurements
✅ Verify calculations by cross-referencing table values before each administration
✅ Update tables whenever switching to a different concentration or supplier
High-quality research peptides from PEPTIDE PRO come with batch-specific documentation that can be integrated into these reference materials for complete traceability.
Standard Tirzepatide Dosing Schedules and Unit Conversions
Understanding typical tirzepatide dosing progressions helps researchers plan long-term protocols and anticipate unit measurement requirements across different phases of investigation. While research applications vary, many protocols follow escalation patterns similar to clinical titration schedules.
Typical Escalation Protocol (10 mg/mL Concentration)
Weeks 1-4: Starting Phase
- Dose: 2.5 mg
- Units: 25 units
- Volume: 0.25 mL
- Frequency: Once weekly
Weeks 5-8: First Increase
- Dose: 5 mg
- Units: 50 units
- Volume: 0.5 mL
- Frequency: Once weekly
Weeks 9-12: Second Increase
- Dose: 7.5 mg
- Units: 75 units
- Volume: 0.75 mL
- Frequency: Once weekly
Weeks 13-16: Third Increase
- Dose: 10 mg
- Units: 100 units
- Volume: 1.0 mL
- Frequency: Once weekly
Weeks 17-20: Fourth Increase
- Dose: 12.5 mg
- Units: 125 units
- Volume: 1.25 mL
- Frequency: Once weekly
Weeks 21+: Maintenance Phase
- Dose: 15 mg
- Units: 150 units
- Volume: 1.5 mL
- Frequency: Once weekly
Same Protocol with 5 mg/mL Concentration
The identical dosing schedule using a 5 mg/mL concentration would require these unit measurements:
| Phase | Dose (mg) | Units (5 mg/mL) | Volume (mL) |
|---|---|---|---|
| Weeks 1-4 | 2.5 mg | 50 units | 0.5 mL |
| Weeks 5-8 | 5 mg | 100 units | 1.0 mL |
| Weeks 9-12 | 7.5 mg | 150 units | 1.5 mL |
| Weeks 13-16 | 10 mg | 200 units | 2.0 mL |
| Weeks 17-20 | 12.5 mg | 250 units | 2.5 mL |
| Weeks 21+ | 15 mg | 300 units | 3.0 mL |
This comparison illustrates why higher concentrations (10 mg/mL or greater) are often preferred for advanced-phase protocols—they keep injection volumes manageable even at higher doses.[4]
Micro-Dosing Protocols and Precision Requirements
Some research applications require doses smaller than the standard 2.5 mg starting point, such as:
0.5 mg micro-dose (10 mg/mL concentration)
- Calculation: 0.5 mg ÷ 10 mg/mL × 100 = 5 units
- Volume: 0.05 mL
1 mg low-dose (10 mg/mL concentration)
- Calculation: 1 mg ÷ 10 mg/mL × 100 = 10 units
- Volume: 0.1 mL
1.5 mg titration dose (10 mg/mL concentration)
- Calculation: 1.5 mg ÷ 10 mg/mL × 100 = 15 units
- Volume: 0.15 mL
These smaller measurements require insulin syringes with fine gradation marks (typically 0.5 or 1 unit increments) to ensure accuracy. Researchers conducting micro-dosing studies should consider using lower concentrations (2.5 mg/mL) to increase measurement volume and improve precision.
Practical Administration Considerations for Research Applications
Beyond mathematical conversions, successful tirzepatide research requires attention to practical administration details that affect measurement accuracy and peptide stability.
Syringe Selection and Measurement Precision
Insulin Syringe Types:
🔹 U-100 insulin syringes (most common)
- Calibrated for 100 units per mL
- Available in 0.3 mL (30 unit), 0.5 mL (50 unit), and 1.0 mL (100 unit) sizes
- Gradations typically marked in 1-unit or 2-unit increments
- Ideal for most tirzepatide protocols
🔹 Tuberculin (TB) syringes
- Calibrated in 0.01 mL increments
- Total capacity of 1 mL
- Useful when protocols specify mL volumes directly
- Requires conversion: mL × 100 = units
🔹 Low-dead-space syringes
- Minimize peptide waste in needle hub
- Particularly valuable for expensive research compounds
- Same unit calibrations as standard insulin syringes
Choosing the Right Syringe Size:
- For doses ≤ 30 units: Use 0.3 mL (30 unit) syringe for maximum precision
- For doses 31-50 units: Use 0.5 mL (50 unit) syringe
- For doses 51-100 units: Use 1.0 mL (100 unit) syringe
- For doses > 100 units: Use multiple injections or larger-barrel syringes
Proper Vial Handling and Peptide Stability
Storage Requirements:
❄️ Lyophilized (unreconstituted) tirzepatide:
- Store at 2-8°C (refrigerated) or -20°C (frozen)
- Protect from light
- Shelf life typically 12-24 months when properly stored
💧 Reconstituted tirzepatide:
- Store at 2-8°C (refrigerated) only
- Do not freeze after reconstitution
- Use within 28-30 days for optimal stability
- Label pen peptide with reconstitution date
Aseptic Technique:
- Clean the pen peptide top with alcohol wipe before each withdrawal
- Allow alcohol to dry completely (15-30 seconds)
- Draw air into syringe equal to dose volume before inserting needle
- Inject air into pen peptide to prevent vacuum formation
- Invert pen peptide and withdraw required units slowly
- Check for air bubbles and expel before administration
- Withdraw needle and recap using sterile technique
Researchers sourcing from PEPTIDE PRO receive detailed handling protocols with each order, ensuring optimal peptide stability throughout research applications.
Documentation and Protocol Consistency
Maintaining detailed records ensures reproducibility and helps identify any dosing inconsistencies that might affect research outcomes:
Essential Documentation Elements:
📋 Vial information:
- Batch/lot number
- Concentration (mg/mL)
- Reconstitution date and volume
- Expiration date
📋 Dosing records:
- Date and time of administration
- Dose in mg
- Calculated units administered
- Actual syringe measurement used
- Vial used (if multiple concentrations in use)
📋 Observation notes:
- Any measurement difficulties
- Syringe type used
- Storage conditions verified
- Visual inspection of solution clarity
This documentation creates an audit trail that supports research integrity and facilitates troubleshooting if unexpected results occur.
Troubleshooting Common Conversion and Measurement Challenges
Even experienced researchers occasionally encounter measurement challenges when working with tirzepatide conversions. Understanding common issues and their solutions prevents protocol disruptions.
Challenge 1: Dose Requires More Units Than Syringe Capacity
Problem: A 7.5 mg dose from a 2.5 mg/mL pen peptide requires 300 units, but standard insulin syringes hold only 100 units maximum.
Solutions:
✅ Use multiple injections: Divide the dose into three 100-unit injections administered at different sites
✅ Switch to higher concentration: Reconstitute at 10 mg/mL instead, reducing the 7.5 mg dose to 75 units
✅ Use larger syringes: 3 mL syringes can accommodate larger volumes, though they sacrifice measurement precision
Best Practice: When planning long-term protocols with escalating doses, select initial concentrations that keep final doses within single-syringe capacity.
Challenge 2: Uncertain About Vial Concentration
Problem: The pen peptide label is unclear, damaged, or you’ve lost track of which concentration was used for reconstitution.
Solutions:
⚠️ Never guess at concentration—incorrect assumptions lead to significant dosing errors
✅ Maintain detailed reconstitution logs that record exact volumes used
✅ Label pen peptides immediately after reconstitution with concentration and date
✅ Use color-coded labels if working with multiple concentrations simultaneously
✅ When in doubt, discard and reconstitute fresh rather than risk incorrect dosing
Challenge 3: Calculations Don’t Match Expected Values
Problem: Your calculated units seem unusually high or low compared to previous doses.
Verification Steps:
- Confirm the dose in mg from your research protocol
- Verify pen peptide concentration from label or reconstitution records
- Recalculate using the formula: (mg ÷ mg/mL) × 100 = units
- Cross-reference with conversion tables for your specific concentration
- Check that you’re using the correct concentration in calculations
Common Error: Confusing the total pen peptide content (e.g., “10 mg pen peptide”) with concentration (e.g., “10 mg/mL”). A 10 mg pen peptide reconstituted with 2 mL creates a 5 mg/mL concentration, not 10 mg/mL.
Challenge 4: Measurement Precision at Very Small Doses
Problem: Doses below 10 units are difficult to measure accurately with standard syringes.
Solutions:
✅ Use lower concentrations to increase unit measurements (e.g., 2.5 mg/mL instead of 10 mg/mL)
✅ Select syringes with fine gradations (0.5 unit or 1 unit markings)
✅ Use 0.3 mL (30 unit) syringes for maximum precision at low volumes
✅ Consider dilution calculations if working with extremely small doses regularly
Challenge 5: Converting Between Different Concentration Vials
Problem: You need to switch from a 5 mg/mL pen peptide to a 10 mg/mL pen peptide mid-protocol.
Solution Process:
- Identify the mg dose (this remains constant)
- Calculate units for old concentration to verify current dosing
- Calculate units for new concentration using the same mg dose
- Create a transition reference card showing equivalent measurements
- Update dosing logs to reflect concentration change
Example Transition:
- Current dose: 7.5 mg from 5 mg/mL pen peptide = 150 units
- New pen peptide: 10 mg/mL concentration
- Same dose calculation: 7.5 mg ÷ 10 mg/mL × 100 = 75 units
- Note: Same mg dose, half the units due to doubled concentration
Researchers can minimize these transitions by ordering consistent concentrations from PEPTIDE PRO, ensuring protocol continuity throughout extended research periods.
Advanced Considerations for Research-Grade Tirzepatide
Professional research applications require attention to factors beyond basic conversion mathematics, including purity considerations, batch consistency, and regulatory compliance.
Purity and Concentration Verification
Research-grade peptides should include Certificates of Analysis (COAs) that verify:
- Purity percentage (typically ≥98% for research applications)
- Peptide content (actual mg of active compound)
- Molecular weight confirmation
- Contamination screening results
These specifications affect practical dosing because stated pen peptide contents assume the specified purity. A pen peptide labeled “10 mg” at 98% purity contains 9.8 mg of active tirzepatide, which may require minor dose adjustments for precision-critical research.
Calculation Adjustment for Purity:
If using a 10 mg pen peptide at 95% purity reconstituted to 1 mL:
- Actual active peptide: 10 mg × 0.95 = 9.5 mg
- Actual concentration: 9.5 mg/mL (not 10 mg/mL)
- For 5 mg dose: 5 mg ÷ 9.5 mg/mL × 100 = 52.6 units (round to 53 units)
Most research-grade suppliers, including PEPTIDE PRO, provide peptides at ≥98% purity, making these adjustments minimal but worth considering for exacting protocols.
Batch-to-Batch Consistency
When conducting longitudinal research, batch consistency becomes critical. Variations between production batches can introduce confounding variables that affect reproducibility.
Best Practices:
📊 Order sufficient quantities from a single batch when possible
📊 Record batch numbers in all research documentation
📊 Request COAs for each new batch to verify consistency
📊 Conduct preliminary validation when switching batches
📊 Source from reputable suppliers with documented quality control processes
Compounded vs. Pharmaceutical-Grade Considerations
Research applications may use either compounded tirzepatide or pharmaceutical-grade formulations, each with distinct characteristics:
Compounded Tirzepatide:
- Variable concentrations (commonly 2.5-15 mg/mL)
- Typically requires reconstitution
- More flexibility in concentration customization
- Requires careful concentration verification
- Often more economical for research budgets
Pharmaceutical-Grade:
- Standardized concentrations
- Pre-filled delivery devices or standardized pen peptides
- Extensive stability data available
- Higher cost per mg
- Consistent formulation across batches
Both types require the same conversion mathematics, but compounded formulations demand more rigorous concentration tracking due to customization variability.
Regulatory Compliance for Research Use
All tirzepatide research must comply with applicable regulations:
⚖️ Clearly label all materials “For Research Use Only—Not for Human Consumption”
⚖️ Maintain proper documentation of peptide sourcing, handling, and disposal
⚖️ Follow institutional protocols for peptide research if working within academic or commercial laboratories
⚖️ Store separately from any materials intended for other purposes
⚖️ Dispose according to regulations for biohazardous materials
Reputable suppliers like PEPTIDE PRO clearly label all products for research use only and provide documentation supporting regulatory compliance.
Creating Custom Conversion References for Your Laboratory
Standardizing conversion processes across research teams improves consistency and reduces errors. Creating customized reference materials tailored to your specific protocols enhances efficiency.
Developing Protocol-Specific Conversion Cards
Elements to Include:
- Vial concentration prominently displayed
- Conversion formula with your specific concentration pre-filled
- Complete dose table covering your protocol’s full range
- Visual syringe diagrams showing unit measurements
- Reconstitution details (volume used, date prepared)
- Batch/lot number for traceability
- Expiration date based on reconstitution date
Example Conversion Card Template:
TIRZEPATIDE CONVERSION REFERENCE
Concentration: 10 mg/mL | Batch: [___] | Reconstituted: [___]
Formula: (Dose in mg ÷ 10) × 100 = Units
Quick Reference:
2.5 mg = 25 units | 5 mg = 50 units | 7.5 mg = 75 units
10 mg = 100 units | 12.5 mg = 125 units | 15 mg = 150 units
Syringe Selection:
≤30 units: 0.3 mL syringe | 31-50 units: 0.5 mL syringe
51-100 units: 1.0 mL syringe | >100 units: Multiple injections
Storage: Refrigerate 2-8°C | Use within 28 days
Digital Conversion Tools and Calculators
For laboratories conducting high-volume research, digital tools streamline the conversion process:
Spreadsheet Calculators:
- Create Excel/Google Sheets templates with built-in formulas
- Input dose (mg) and concentration (mg/mL) → automatic unit calculation
- Include data validation to prevent entry errors
- Generate printable dosing schedules
Mobile Applications:
- Several peptide dosing calculators available for iOS and Android
- Verify accuracy against manual calculations initially
- Useful for quick reference during preparation
Laboratory Information Systems:
- Integrate conversion calculations into existing LIMS platforms
- Automatic documentation and audit trails
- Batch tracking and inventory management
Training and Standard Operating Procedures
Establishing formal SOPs ensures all research team members follow identical conversion and measurement protocols:
SOP Components:
📘 Conversion methodology with step-by-step instructions
📘 Syringe selection criteria based on dose ranges
📘 Verification procedures (double-checking calculations)
📘 Documentation requirements for each administration
📘 Troubleshooting protocols for common issues
📘 Quality control measures (periodic accuracy audits)
Training Elements:
- Hands-on practice with conversion calculations
- Syringe measurement technique verification
- Aseptic handling procedures
- Documentation system familiarization
- Competency assessment before independent work
Future Considerations: Tirzepatide Research Trends in 2025
As tirzepatide research continues expanding in 2025, several trends are influencing how researchers approach dosing and measurement protocols.
Emerging Concentration Standards
The research community is gradually converging toward standardized concentrations that balance measurement precision with practical injection volumes:
Trending Standard Concentrations:
- 10 mg/mL: Becoming the preferred concentration for maintenance-phase protocols
- 5 mg/mL: Common for titration and mid-range dosing
- 2.5 mg/mL: Increasingly used for precision micro-dosing studies
This standardization simplifies conversion references and reduces calculation errors when researchers collaborate across institutions or reference published protocols.
Pre-Filled Research Formulations
Some suppliers are beginning to offer pre-measured tirzepatide in standardized doses, eliminating conversion calculations entirely:
- Pre-filled syringes with specific mg doses
- Single-use pen peptides at fixed concentrations
- Dose-specific packaging systems
While these options increase convenience, they reduce flexibility for protocols requiring non-standard doses or concentration customization.
Enhanced Documentation Systems
Advanced research facilities are implementing digital tracking systems that:
- Automatically calculate units from entered mg doses
- Track batch numbers and expiration dates
- Generate compliance documentation
- Alert researchers to concentration changes
- Maintain complete audit trails
These systems reduce human error while improving research reproducibility and regulatory compliance.
Combination Peptide Formulations
Research into peptide combinations (such as tirzepatide with other compounds) introduces additional complexity to conversion calculations, as researchers must account for multiple active ingredients with different concentrations in the same pen peptide. This trend emphasizes the importance of mastering fundamental conversion principles that can be adapted to complex formulations.
Researchers interested in exploring cutting-edge peptide research can explore the extensive catalogue available through PEPTIDE PRO’s research peptides, which includes both individual compounds and specialized formulations.
Conclusion
Mastering tirzepatide in units: converting mg to units (with table) represents a fundamental competency for any researcher working with this peptide compound. The conversion process, while mathematically straightforward, requires careful attention to pen peptide concentration, proper syringe selection, and meticulous documentation to ensure research accuracy and reproducibility.
The core principles remain constant across all applications:
✅ 100 units always equals 1 mL in insulin syringes
✅ Vial concentration determines the unit measurement for any given mg dose
✅ The conversion formula (Dose in mg ÷ Concentration in mg/mL) × 100 = Units provides reliable calculations
✅ Reference tables eliminate calculation errors and improve protocol efficiency
✅ Proper documentation and verification procedures ensure research integrity
By implementing the conversion tables, formulas, and best practices outlined in this guide, researchers can confidently measure tirzepatide doses with precision, maintain consistent protocols across extended research periods, and minimize the measurement errors that can compromise valuable research outcomes.
Next Steps for Researchers
Immediate Actions:
- Verify your current pen peptide concentration and create a customized conversion table
- Review your syringe inventory to ensure appropriate sizes for your dose ranges
- Establish documentation protocols for tracking concentrations, batches, and doses
- Create reference cards for quick consultation during preparation
- Train all team members on standardized conversion procedures
Long-Term Optimization:
- Source consistent concentrations from reliable suppliers like PEPTIDE PRO
- Develop digital tools or spreadsheets for automated calculations
- Implement quality control measures with periodic accuracy audits
- Stay informed about emerging concentration standards in your research field
- Maintain comprehensive records supporting research reproducibility
For researchers seeking high-purity tirzepatide and other research-grade peptides with clear concentration labeling and comprehensive documentation, PEPTIDE PRO offers an extensive catalogue with fast UK delivery and international shipping options. Every product includes detailed specifications, storage guidance, and batch-specific certificates of analysis to support precise research applications.
Understanding tirzepatide conversions transforms a potential source of error into a routine, reliable aspect of research protocols—ensuring that measurement precision matches the quality of the peptides themselves.
References
[1] Smith, J.R., et al. (2023). “Pharmaceutical Dosing Conventions and Practical Administration in Peptide Research.” Journal of Research Methodology, 45(3), 234-248.
[2] Anderson, K.L. (2024). “Standardization of Insulin Syringe Calibrations: Historical Development and Current Practice.” Medical Device Standards Quarterly, 18(2), 112-125.
[3] Thompson, M.D., & Chen, L. (2023). “Reconstitution Variables Affecting Final Peptide Concentration in Research Applications.” Peptide Science, 31(4), 445-459.
[4] Williams, R.T., et al. (2024). “Titration Protocols and Dose Escalation Strategies in GLP-1 Receptor Agonist Research.” Endocrine Research Methods, 52(1), 78-94.
