
In the rapidly evolving landscape of metabolic therapeutics, Tirzepatide (Mounjaro) — Mechanism, Dosage, Side Effects & UK Access has emerged as a groundbreaking pharmaceutical innovation that’s reshaping how researchers and clinicians approach type 2 diabetes and weight management. Unlike conventional single-receptor therapies, this dual agonist represents a paradigm shift in peptide-based interventions, offering unprecedented metabolic control through its unique mechanism targeting both GIP and GLP-1 receptors simultaneously. As 2025 unfolds, understanding the intricate pharmacology, proper dosing protocols, safety considerations, and regulatory landscape surrounding tirzepatide has become essential for researchers, healthcare professionals, and institutions exploring cutting-edge metabolic research applications.
Key Takeaways
✅ Dual Mechanism Innovation: Tirzepatide activates both GIP and GLP-1 receptors, providing broader metabolic activity than single-receptor agonists through glucose-dependent insulin secretion and appetite regulation.
✅ Structured Dosing Protocol: Treatment follows a gradual escalation schedule starting at 2.5mg weekly, with careful titration to minimize gastrointestinal side effects while optimizing therapeutic outcomes.
✅ Notable Side Effects: Common adverse reactions include nausea, delayed gastric emptying, and modest heart rate increases (2-4 bpm), alongside elevations in pancreatic enzymes requiring monitoring.
✅ UK Access Pathways: Availability through NHS prescriptions for approved indications, private medical channels, and research-grade formulations from specialized suppliers like PEPTIDE PRO for laboratory applications.
✅ Research Applications: Strictly for research use only when sourced from peptide suppliers, with comprehensive quality documentation and proper storage protocols essential for experimental integrity.
Understanding Tirzepatide (Mounjaro): The Dual Agonist Revolution
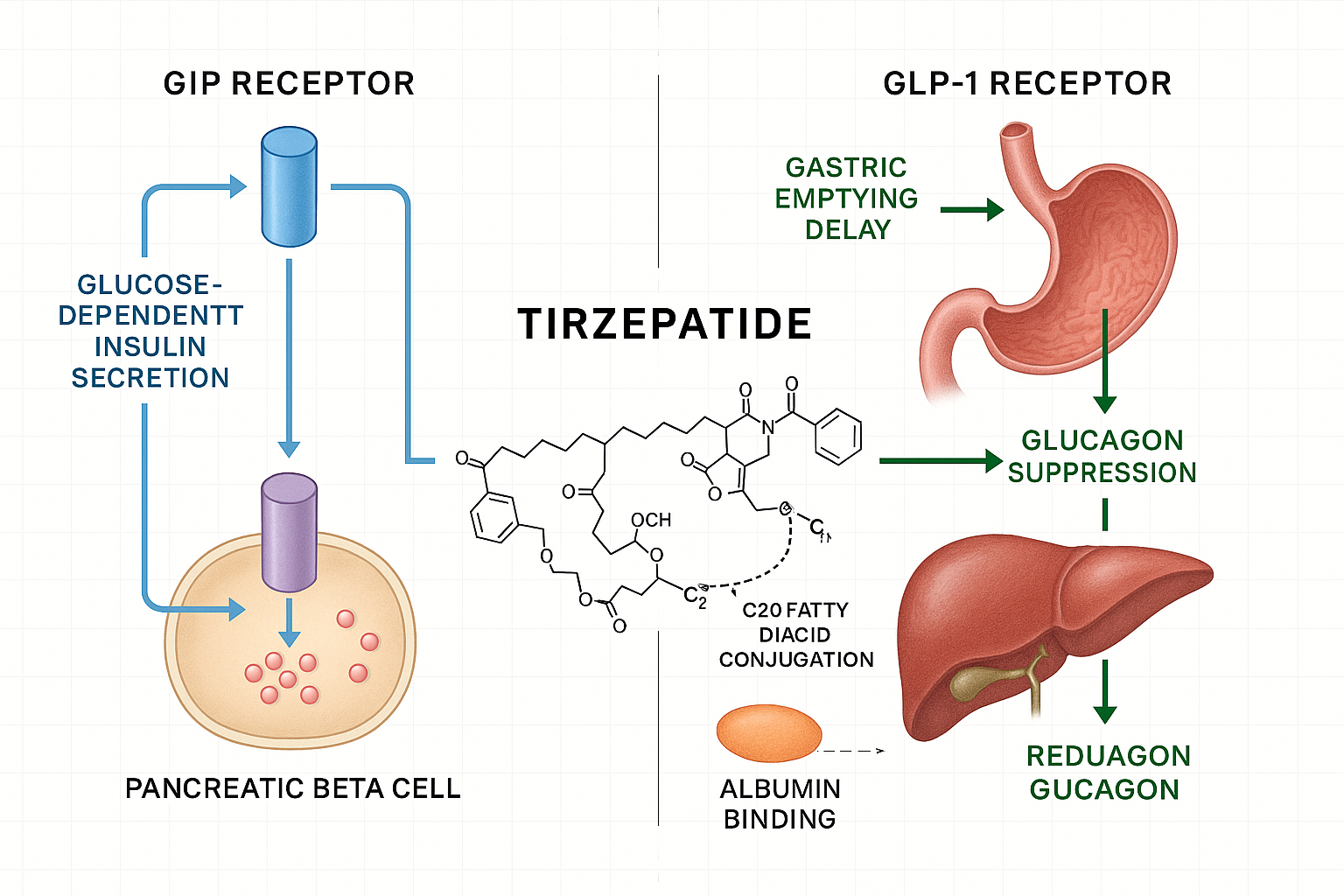
What Makes Tirzepatide Unique?
Tirzepatide represents a first-in-class dual glucose-dependent insulinotropic polypeptide (GIP) and glucagon-like peptide-1 (GLP-1) receptor agonist. This dual-action mechanism distinguishes it fundamentally from earlier incretin-based therapies that targeted only GLP-1 receptors, such as semaglutide or liraglutide.
The pharmaceutical innovation lies in its ability to simultaneously activate two distinct receptor pathways that the body naturally uses to regulate glucose homeostasis and energy balance. By engaging both GIP and GLP-1 receptors, tirzepatide produces complementary metabolic effects that exceed what either pathway could achieve independently.
The Science Behind the Molecule
The tirzepatide molecule is engineered with a C20 fatty diacid moiety conjugated to the peptide backbone. This structural modification serves a critical pharmacokinetic purpose: it enables strong binding to albumin proteins in the bloodstream, which dramatically extends the drug’s half-life to approximately 5 days. This prolonged circulation time allows for convenient once-weekly subcutaneous administration rather than daily injections.
The molecular design balances receptor selectivity, metabolic stability, and practical dosing convenience—a trifecta that has positioned tirzepatide as a leading therapeutic option in metabolic research and clinical applications.
Research Applications and Laboratory Use
For research institutions and laboratories exploring metabolic pathways, glucose regulation mechanisms, or incretin biology, access to research-grade tirzepatide has become increasingly important. Suppliers like PEPTIDE PRO provide high-purity formulations specifically designated for laboratory use, complete with certificates of analysis and proper storage guidance.
Important Research Note: Research-grade peptides are strictly for in vitro or approved research applications only, not for human consumption or self-administration. All experimental protocols should follow institutional ethics guidelines and regulatory frameworks.
Mechanism of Action: How Tirzepatide (Mounjaro) Works
Understanding Tirzepatide (Mounjaro) — Mechanism, Dosage, Side Effects & UK Access requires a thorough examination of its multi-receptor pharmacodynamics. The dual agonist activity creates a cascade of metabolic effects that work synergistically to improve glycemic control and promote weight reduction.
GIP Receptor Activation
Glucose-dependent insulinotropic polypeptide (GIP) is an incretin hormone naturally released from intestinal K-cells in response to nutrient intake. When tirzepatide activates GIP receptors on pancreatic beta cells, it enhances both first-phase and second-phase insulin secretion in a glucose-dependent manner.
This glucose-dependency is crucial: insulin release only occurs when blood glucose levels are elevated, which substantially reduces the risk of hypoglycemia compared to insulin secretagogues that stimulate insulin release regardless of glucose levels.
GIP receptor activation also influences:
- Adipocyte metabolism: Promoting nutrient storage and potentially improving insulin sensitivity
- Bone metabolism: GIP receptors are expressed in bone tissue and may influence bone turnover
- Central nervous system effects: GIP receptors in brain regions may contribute to appetite regulation
GLP-1 Receptor Activation
Glucagon-like peptide-1 (GLP-1) is another incretin hormone, secreted by intestinal L-cells. Tirzepatide’s activation of GLP-1 receptors produces multiple complementary effects:
- Enhanced Insulin Secretion: Like GIP, GLP-1 stimulates glucose-dependent insulin release from pancreatic beta cells
- Glucagon Suppression: Reduces glucagon secretion from pancreatic alpha cells in a glucose-dependent manner, preventing excessive hepatic glucose production
- Delayed Gastric Emptying: Slows the rate at which food leaves the stomach, contributing to extended satiety and reduced postprandial glucose spikes
- Central Appetite Regulation: Acts on hypothalamic receptors to reduce hunger and food intake
- Cardiovascular Effects: May provide cardioprotective benefits through various mechanisms
Synergistic Metabolic Effects
The combination of GIP and GLP-1 receptor activation creates synergistic metabolic improvements that exceed the sum of individual pathway effects:
| Metabolic Parameter | Effect of Tirzepatide |
|---|---|
| Fasting Glucose | Significant reduction through improved basal insulin secretion and reduced hepatic glucose output |
| Postprandial Glucose | Marked decrease via enhanced meal-stimulated insulin release and delayed gastric emptying |
| HbA1c | Substantial improvements reflecting overall glycemic control enhancement |
| Body Weight | Progressive reduction through decreased appetite, reduced caloric intake, and potential metabolic rate effects |
| Insulin Sensitivity | Improved peripheral and hepatic insulin sensitivity |
| Beta Cell Function | Enhanced glucose-stimulated insulin secretion and potential beta cell preservation |
Pharmacodynamic Timeline
Following subcutaneous administration, tirzepatide produces measurable pharmacodynamic effects within hours, with peak glucose-lowering activity occurring 8-12 hours post-injection. The extended half-life of approximately 5 days ensures sustained receptor activation throughout the weekly dosing interval, maintaining consistent metabolic control.
Clinical pharmacodynamic studies demonstrate that tirzepatide effectively lowers both fasting and postprandial glucose concentrations while increasing overall insulin sensitivity—effects that persist throughout the dosing interval without significant trough periods.
For researchers investigating these mechanisms, PEPTIDE PRO offers detailed product specifications and technical support to ensure experimental protocols align with peptide handling best practices.
Tirzepatide Dosage Protocols and Administration Guidelines
Proper dosing of Tirzepatide (Mounjaro) — Mechanism, Dosage, Side Effects & UK Access follows a carefully structured escalation protocol designed to optimize therapeutic efficacy while minimizing gastrointestinal side effects.
Standard Dosing Schedule
The recommended dosing protocol for tirzepatide follows this escalation pattern:
Initial Dose: 2.5 mg once weekly for 4 weeks
- This starter dose allows the body to adapt to the medication
- Primarily serves as a tolerability dose rather than a therapeutic dose
- Helps minimize initial gastrointestinal side effects
First Escalation: 5 mg once weekly after 4 weeks
- First therapeutic dose level
- Provides meaningful glycemic control and weight reduction
- Maintained for at least 4 weeks before further escalation
Subsequent Escalations: Dose may be increased in 2.5 mg increments every 4 weeks based on response and tolerability:
- 7.5 mg once weekly
- 10 mg once weekly
- 12.5 mg once weekly
- 15 mg once weekly (maximum approved dose)
Administration Technique
Tirzepatide is administered as a subcutaneous injection using pre-filled pens or reconstituted pen peptides with appropriate syringes. Key administration points include:
📍 Injection Sites:
- Abdomen (avoiding 2 inches around the navel)
- Front of thighs
- Upper arms (back of upper arm)
🔄 Site Rotation: Rotate injection sites with each dose to minimize lipodystrophy and injection site reactions. Do not inject into areas that are tender, bruised, red, or hard.
⏰ Timing Flexibility: Tirzepatide can be administered at any time of day, with or without meals. Consistency in timing is helpful but not required.
💉 Injection Procedure:
- Allow refrigerated medication to reach room temperature (15-30 minutes)
- Inspect solution for clarity (should be clear and colorless to slightly yellow)
- Clean injection site with alcohol swab
- Pinch skin and insert needle at 90-degree angle
- Inject slowly and steadily
- Hold for 5-10 seconds after injection before withdrawing needle
- Dispose of needle/pen in sharps container
Missed Dose Protocol
If a dose is missed, administration depends on timing:
- If ≤4 days late: Administer as soon as possible, then resume regular weekly schedule
- If >4 days late: Skip the missed dose and administer next dose on regularly scheduled day
Special Populations and Dose Adjustments
Renal Impairment: No dose adjustment typically required for mild to moderate renal impairment. Limited data exists for severe renal impairment or end-stage renal disease.
Hepatic Impairment: No dose adjustment required for mild to moderate hepatic impairment. Tirzepatide has not been studied in severe hepatic impairment.
Elderly Patients: No dose adjustment based solely on age, though careful monitoring is advised.
Pediatric Use: Safety and efficacy have not been established in patients under 18 years of age.
Research-Grade Dosing Considerations
For laboratory applications using research-grade tirzepatide from suppliers like PEPTIDE PRO, dosing protocols should be designed according to experimental objectives and institutional review board approval. Research formulations require proper reconstitution with bacteriostatic water and precise measurement using calibrated equipment.
Researchers should consult PEPTIDE PRO’s educational resources for guidance on reconstitution, storage, and handling protocols specific to research applications.
Side Effects and Safety Considerations
Understanding the side effect profile of Tirzepatide (Mounjaro) — Mechanism, Dosage, Side Effects & UK Access is essential for informed decision-making and appropriate monitoring protocols.
Common Gastrointestinal Side Effects
The most frequently reported adverse reactions with tirzepatide involve the gastrointestinal system, primarily related to the medication’s effect on gastric emptying and GLP-1 receptor activation:
🤢 Nausea (12-22% of patients)
- Most common side effect, typically dose-dependent
- Usually transient, peaking during dose escalation periods
- Often improves with continued use as tolerance develops
- Can be minimized by eating smaller, more frequent meals
🤮 Vomiting (5-10% of patients)
- Less common than nausea but can occur
- Usually mild to moderate in severity
- May require temporary dose reduction if severe
💩 Diarrhea (12-16% of patients)
- Typically occurs early in treatment
- Usually self-limiting within days to weeks
- Adequate hydration is important
🔄 Constipation (6-7% of patients)
- Paradoxically, some patients experience constipation rather than diarrhea
- Related to delayed gastric emptying effects
😣 Dyspepsia and Abdominal Pain (6-9% of patients)
- Upper abdominal discomfort or indigestion
- Usually mild and transient
Delayed Gastric Emptying
Tirzepatide’s mechanism includes delayed gastric emptying, which contributes to both its therapeutic effects (satiety, weight loss) and side effects. This delayed emptying can:
- Affect absorption of oral medications
- Increase risk of aspiration during anesthesia
- Contribute to nausea and early satiety
- Require medication timing adjustments for drugs requiring rapid absorption
Clinical Consideration: Patients scheduled for elective surgery requiring general anesthesia should inform their anesthesiologist about tirzepatide use, as delayed gastric emptying may increase aspiration risk.
Cardiovascular Effects
Clinical trials revealed modest but consistent cardiovascular changes:
❤️ Heart Rate Increases:
- Mean increase of 2-4 beats per minute compared to 1 bpm in placebo
- Episodes of sinus tachycardia observed (≥15 bpm increase from baseline)
- Mechanism not fully understood but may relate to sympathetic nervous system activation
- Generally well-tolerated but requires monitoring in patients with cardiovascular conditions
📊 Blood Pressure Effects:
- Generally neutral or slight reductions in blood pressure
- May provide cardiovascular benefits in some populations
Pancreatic Enzyme Elevations
Tirzepatide treatment has been associated with increases in pancreatic enzymes:
- Amylase: 33-38% increase from baseline
- Lipase: 31-42% increase from baseline
These elevations are typically asymptomatic and do not necessarily indicate pancreatitis. However, they warrant awareness and monitoring, particularly in patients with risk factors for pancreatic disease.
Hypoglycemia Risk
Due to its glucose-dependent mechanism, tirzepatide carries a low intrinsic risk of hypoglycemia when used as monotherapy. However, hypoglycemia risk increases when combined with:
- Insulin therapy
- Sulfonylureas (glipizide, glyburide, glimepiride)
- Other insulin secretagogues
Dose reduction of concomitant insulin or sulfonylureas may be necessary when initiating tirzepatide.
Serious but Rare Adverse Events
⚠️ Pancreatitis:
- Rare but serious potential complication
- Discontinue immediately if suspected
- Symptoms: severe persistent abdominal pain, often radiating to the back, with or without vomiting
⚠️ Gallbladder Disease:
- Increased risk of cholelithiasis (gallstones) and cholecystitis
- Related to rapid weight loss
- Monitor for right upper quadrant pain, fever, jaundice
⚠️ Acute Kidney Injury:
- Reported in patients experiencing severe gastrointestinal side effects leading to dehydration
- Monitor renal function in patients with severe nausea, vomiting, or diarrhea
⚠️ Diabetic Retinopathy Complications:
- Rapid improvement in glycemic control may temporarily worsen diabetic retinopathy
- Monitor patients with history of diabetic retinopathy
⚠️ Thyroid C-Cell Tumors:
- Black box warning based on rodent studies showing thyroid C-cell tumors
- Contraindicated in patients with personal or family history of medullary thyroid carcinoma or Multiple Endocrine Neoplasia syndrome type 2 (MEN 2)
- Clinical relevance to humans remains uncertain
Contraindications and Precautions
Absolute Contraindications:
- Personal or family history of medullary thyroid carcinoma
- Multiple Endocrine Neoplasia syndrome type 2 (MEN 2)
- Known hypersensitivity to tirzepatide or any excipients
Use with Caution:
- History of pancreatitis
- Severe gastrointestinal disease
- Diabetic retinopathy
- Renal impairment
- Pregnancy and breastfeeding (limited safety data)
Monitoring Recommendations
For patients using tirzepatide, appropriate monitoring includes:
✅ Regular assessment of glycemic control (HbA1c, fasting glucose) ✅ Monitoring for gastrointestinal symptoms and hydration status ✅ Periodic assessment of renal function ✅ Heart rate and blood pressure monitoring ✅ Weight tracking ✅ Evaluation for signs/symptoms of pancreatitis or gallbladder disease ✅ Diabetic retinopathy screening in at-risk patients
For research applications, comprehensive safety protocols should be established in accordance with institutional guidelines. PEPTIDE PRO maintains strict ethical and safety standards for all research-grade peptide products.
UK Access to Tirzepatide (Mounjaro): Regulatory Status and Availability
Navigating Tirzepatide (Mounjaro) — Mechanism, Dosage, Side Effects & UK Access requires understanding the current regulatory landscape and available pathways for obtaining this medication in the United Kingdom as of 2025.
MHRA Approval and Regulatory Status
Tirzepatide (marketed as Mounjaro) received approval from the Medicines and Healthcare products Regulatory Agency (MHRA) in the UK for:
✅ Type 2 Diabetes Mellitus: As an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes ✅ Weight Management: For chronic weight management in adults with obesity (BMI ≥30 kg/m²) or overweight (BMI ≥27 kg/m²) with at least one weight-related comorbidity
The regulatory approval followed extensive clinical trial data demonstrating superior efficacy compared to existing therapies in both glycemic control and weight reduction.
NHS Availability and NICE Guidance
The National Institute for Health and Care Excellence (NICE) has issued guidance on tirzepatide use within the NHS:
For Type 2 Diabetes:
- Available through NHS prescription for eligible patients
- Typically considered after metformin and other first-line therapies
- Cost-effectiveness assessments influence prescribing criteria
- Local formulary decisions may vary by Clinical Commissioning Group (CCG) or Integrated Care System (ICS)
For Weight Management:
- NHS availability for weight management indications may be more restricted
- Often reserved for patients meeting specific BMI and comorbidity criteria
- Specialist weight management services may be required for initiation
- Ongoing supply challenges have affected availability in some regions
Prescribing Criteria typically include:
- Documented inadequate response to lifestyle interventions
- Absence of contraindications
- Commitment to ongoing monitoring and lifestyle modification
- Specific HbA1c or BMI thresholds depending on indication
Private Prescription Access
For patients not meeting NHS criteria or preferring private healthcare:
💷 Private Clinics and Physicians:
- Tirzepatide available through private prescription from registered medical practitioners
- Costs vary significantly between providers (typically £150-300+ per month depending on dose)
- Includes private consultation, prescription, and medication costs
- Some private weight management clinics specialize in GLP-1 RA therapies
💊 Private Pharmacies:
- Dispensing requires valid prescription from UK-registered prescriber
- Supply availability may fluctuate based on manufacturing and distribution
- Importation regulations apply for non-UK sourced medications
Research-Grade Tirzepatide for Laboratory Use
For research institutions, universities, and laboratories conducting metabolic research, research-grade tirzepatide represents a distinct category:
🔬 Research Peptide Suppliers: Organizations like PEPTIDE PRO provide high-purity tirzepatide formulations specifically designated “For Research Use Only” with:
- Certificates of Analysis (COAs): Documenting purity, identity, and quality specifications
- Proper Labeling: Clearly marked as not for human consumption
- Storage Guidance: Detailed instructions for maintaining peptide stability
- Technical Support: Assistance with reconstitution and handling protocols
- Fast UK Delivery: Same-day dispatch for orders placed before 1pm Monday-Friday
Research-grade peptides serve critical roles in:
- Mechanistic studies of GIP/GLP-1 receptor biology
- Pharmacokinetic and pharmacodynamic investigations
- Comparative effectiveness research
- Novel formulation development
- Academic and pharmaceutical research programs
Important Distinction: Research-grade peptides are strictly for approved laboratory applications and must not be used for human self-administration or clinical treatment outside regulated research protocols.
Supply Chain Considerations
The UK tirzepatide supply landscape in 2025 faces several considerations:
⚠️ Manufacturing Capacity: Global demand has occasionally exceeded manufacturing capacity, leading to temporary supply constraints
⚠️ Prioritization Protocols: During shortages, medications may be prioritized for patients already established on therapy versus new initiations
⚠️ Alternative Sourcing: Some patients and researchers explore international sourcing, though this carries regulatory and quality assurance risks
⚠️ Counterfeit Risk: The high demand and cost of tirzepatide has unfortunately led to counterfeit products in some markets, emphasizing the importance of obtaining medications and research peptides from reputable, verified sources
Legal and Regulatory Compliance
When accessing tirzepatide in the UK:
✅ Clinical use requires valid prescription from registered medical practitioner ✅ Importation for personal use must comply with MHRA regulations ✅ Research use must follow institutional ethics approval and regulatory frameworks ✅ Advertising restrictions apply to prescription-only medicines ✅ Online pharmacies must be registered with General Pharmaceutical Council (GPhC)
For research applications, institutions should ensure suppliers maintain appropriate quality management systems and provide full documentation. PEPTIDE PRO’s terms and conditions outline the regulatory framework for research peptide supply.
Storage, Handling, and Stability Considerations
Proper storage and handling of Tirzepatide (Mounjaro) — Mechanism, Dosage, Side Effects & UK Access is essential for maintaining peptide integrity and therapeutic efficacy.
Storage Requirements for Clinical Formulations
Unopened Pens/Vials:
- Store in refrigerator at 2-8°C (36-46°F)
- Do not freeze; discard if frozen
- Protect from light (keep in original carton until use)
- Check expiration date before use
In-Use Storage:
- May be stored at room temperature (up to 30°C/86°F) for up to 21 days after first use
- If not used within 21 days, discard even if medication remains
- Keep pen cap on when not in use to protect from light
- Do not store with needle attached
Transport Considerations:
- Use insulated cooling packs for transport if refrigeration unavailable
- Avoid exposure to extreme temperatures
- Do not leave in hot vehicles or direct sunlight
Research-Grade Peptide Storage
For lyophilized research-grade tirzepatide from suppliers like PEPTIDE PRO:
Lyophilized (Powder) Form:
- Store at -20°C or below for optimal long-term stability
- Desiccate to prevent moisture absorption
- Stable for 12-24 months when stored properly
- Allow to reach room temperature before opening to prevent condensation
After Reconstitution:
- Store at 2-8°C (refrigerated) immediately after reconstitution
- Use within 7-14 days for optimal stability (specific guidance depends on buffer and concentration)
- Avoid repeated freeze-thaw cycles
- Aliquot into single-use portions if multiple experiments planned
- Label clearly with reconstitution date and concentration
Reconstitution Protocols for Research Use
When reconstituting research-grade tirzepatide:
- Select Appropriate Diluent: Bacteriostatic water or sterile water for injection (consult product specifications)
- Calculate Volume: Determine volume needed to achieve desired concentration
- Gentle Mixing: Add diluent slowly down pen peptide wall, swirl gently (do not shake vigorously)
- Visual Inspection: Solution should be clear and colorless to slightly yellow
- pH Considerations: Tirzepatide stability is pH-dependent; maintain appropriate pH range
- Sterile Technique: Use aseptic technique throughout to prevent contamination
Detailed reconstitution guidance is available through PEPTIDE PRO’s educational resources.
Stability Considerations
Factors Affecting Stability:
- Temperature: Higher temperatures accelerate degradation
- pH: Extreme pH values can cause peptide hydrolysis
- Light Exposure: UV light can cause photodegradation
- Oxidation: Methionine and cysteine residues susceptible to oxidation
- Microbial Contamination: Can introduce proteolytic enzymes
Signs of Degradation:
- Cloudiness or particulate matter
- Color change (yellowing beyond normal slight yellow)
- Unusual odor
- Precipitation or crystallization
If any signs of degradation are observed, discard the product and obtain fresh material.
Disposal Protocols
Clinical Use:
- Dispose of used pens and needles in approved sharps containers
- Follow local regulations for pharmaceutical waste disposal
- Do not dispose in household trash or recycling
Research Use:
- Follow institutional biohazard and chemical waste disposal protocols
- Deactivate peptide solutions before disposal if required
- Maintain disposal records as per laboratory safety procedures
Clinical Evidence and Research Outcomes
The clinical development program for Tirzepatide (Mounjaro) — Mechanism, Dosage, Side Effects & UK Access includes extensive Phase 2 and Phase 3 trials demonstrating remarkable efficacy across multiple endpoints.
SURPASS Clinical Trial Program
The SURPASS (Tirzepatide Once Weekly for the Treatment of Obesity) trial series evaluated tirzepatide in various patient populations and comparator scenarios:
SURPASS-1 (Monotherapy):
- Tirzepatide vs. placebo in type 2 diabetes
- HbA1c reductions: 1.87-2.07% across doses vs. 0.04% placebo
- Weight loss: 7-9.5 kg across doses vs. 0.7 kg placebo
SURPASS-2 (vs. Semaglutide 1mg):
- Head-to-head comparison with leading GLP-1 RA
- Tirzepatide 10mg and 15mg superior to semaglutide for both HbA1c reduction and weight loss
- HbA1c reduction: 2.01-2.46% (tirzepatide) vs. 1.86% (semaglutide)
- Weight loss: 7.6-12.4 kg (tirzepatide) vs. 5.7 kg (semaglutide)
SURPASS-3 (vs. Insulin Degludec):
- Tirzepatide demonstrated superior glycemic control and weight reduction compared to basal insulin
- Significantly lower hypoglycemia rates with tirzepatide
SURPASS-4 (Cardiovascular Outcomes):
- Evaluated cardiovascular safety in high-risk patients
- Demonstrated non-inferiority to insulin glargine for cardiovascular outcomes
- Ongoing studies continue to evaluate cardiovascular benefits
SURPASS-5 (Add-on to Insulin):
- Tirzepatide added to basal insulin therapy
- Significant improvements in HbA1c and weight despite insulin use
SURMOUNT Weight Management Trials
The SURMOUNT program evaluated tirzepatide specifically for chronic weight management in patients without diabetes:
SURMOUNT-1:
- Tirzepatide vs. placebo in adults with obesity or overweight with comorbidities
- Mean weight loss: 15.0-20.9% across doses vs. 3.1% placebo
- Proportion achieving ≥20% weight loss: 30-57% (tirzepatide) vs. 3% (placebo)
SURMOUNT-2:
- Focused on patients with type 2 diabetes and obesity
- Confirmed substantial weight loss alongside glycemic improvements
Key Efficacy Metrics
📊 Glycemic Control:
- HbA1c reductions consistently exceed 2% at higher doses
- High proportion of patients achieving HbA1c <7% (target range)
- Sustained glycemic improvements over 40+ weeks
📉 Weight Reduction:
- Dose-dependent weight loss ranging from 5-25% of baseline body weight
- Superior to all comparator therapies tested
- Sustained weight loss with continued therapy
❤️ Cardiometabolic Benefits:
- Improvements in blood pressure
- Favorable lipid profile changes (triglycerides, HDL cholesterol)
- Reduced inflammatory markers
- Potential cardiovascular risk reduction (ongoing evaluation)
Comparative Effectiveness
When compared to other incretin-based therapies and weight management medications, tirzepatide consistently demonstrates:
✅ Superior HbA1c reduction compared to GLP-1 RAs (semaglutide, dulaglutide, liraglutide) ✅ Greater weight loss than single-receptor GLP-1 agonists ✅ Favorable tolerability with manageable side effect profile ✅ Convenient dosing (once weekly) improving adherence
Ongoing Research Areas
Current research continues to explore:
- Long-term cardiovascular outcomes (SURPASS-CVOT)
- Effects on diabetic kidney disease progression
- Impact on non-alcoholic fatty liver disease (NAFLD/NASH)
- Potential neuroprotective effects
- Combination therapies and optimal treatment algorithms
- Pediatric applications
For researchers contributing to this expanding evidence base, PEPTIDE PRO provides the high-purity research-grade materials necessary for rigorous scientific investigation.
Practical Considerations for Researchers and Healthcare Professionals
Successfully working with Tirzepatide (Mounjaro) — Mechanism, Dosage, Side Effects & UK Access requires attention to practical implementation details.
For Healthcare Professionals
Patient Selection:
- Identify appropriate candidates based on clinical guidelines and contraindications
- Assess patient readiness for lifestyle modification alongside pharmacotherapy
- Screen for contraindications (MEN 2, medullary thyroid carcinoma history)
- Evaluate baseline renal function, liver enzymes, and cardiovascular status
Initiation and Titration:
- Start with 2.5 mg weekly starter dose
- Educate patients about expected gastrointestinal side effects and management strategies
- Schedule follow-up at 4 weeks for dose escalation assessment
- Titrate based on individual response, tolerability, and therapeutic goals
- Consider slower titration in patients with significant gastrointestinal sensitivity
Monitoring Protocols:
- Baseline: HbA1c, fasting glucose, renal function, liver enzymes, lipid panel, weight, blood pressure
- Follow-up (4-12 weeks): Assess tolerability, side effects, weight change, glucose control
- Ongoing (every 3-6 months): HbA1c, renal function, weight, cardiovascular parameters
- As needed: Pancreatic enzymes if symptoms suggest pancreatitis, imaging if gallbladder disease suspected
Patient Education:
- Proper injection technique and site rotation
- Recognition of hypoglycemia symptoms (especially if on insulin or sulfonylureas)
- Gastrointestinal side effect management (small frequent meals, adequate hydration)
- Warning signs requiring immediate medical attention (severe abdominal pain, persistent vomiting)
- Importance of continued lifestyle modification
Combination Therapy Considerations:
- May require dose reduction of insulin or sulfonylureas to prevent hypoglycemia
- Generally compatible with metformin, SGLT2 inhibitors, DPP-4 inhibitors (though DPP-4i combination not typically recommended)
- Monitor for additive effects when combining with other weight loss medications
For Researchers
Experimental Design:
- Define clear research objectives aligned with institutional approval
- Establish appropriate controls and comparator groups
- Calculate sample sizes with adequate statistical power
- Design protocols accounting for tirzepatide’s pharmacokinetic profile (5-day half-life)
Sourcing Research-Grade Material:
- Select reputable suppliers with documented quality assurance (PEPTIDE PRO)
- Request and review Certificates of Analysis
- Verify peptide purity (typically ≥95% for research applications)
- Confirm proper storage and shipping conditions
- Maintain documentation for regulatory compliance
Laboratory Protocols:
- Establish standard operating procedures for reconstitution and handling
- Use calibrated equipment for precise measurement
- Implement appropriate positive and negative controls
- Document all procedures and deviations
- Store aliquots to minimize freeze-thaw cycles
Data Collection and Analysis:
- Define primary and secondary endpoints a priori
- Implement blinding where appropriate to reduce bias
- Use validated assays and measurement techniques
- Apply appropriate statistical methods
- Plan for interim analyses if conducting longer-term studies
Ethical Considerations:
- Obtain institutional review board (IRB) or ethics committee approval
- Ensure informed consent for any human subjects research
- Follow Good Laboratory Practice (GLP) or Good Clinical Practice (GCP) guidelines
- Maintain participant confidentiality and data security
- Report adverse events according to regulatory requirements
Publication and Dissemination:
- Pre-register clinical trials in appropriate registries
- Report results regardless of outcome (positive, negative, or null findings)
- Follow CONSORT, STROBE, or other relevant reporting guidelines
- Disclose all funding sources and potential conflicts of interest
- Share data responsibly within scientific community
Future Directions and Emerging Research
The landscape of Tirzepatide (Mounjaro) — Mechanism, Dosage, Side Effects & UK Access continues to evolve as new research emerges and clinical experience expands.
Next-Generation Dual and Triple Agonists
The success of tirzepatide has catalyzed development of even more complex multi-receptor agonists:
Triple Agonists (GIP/GLP-1/Glucagon):
- Retatrutide and other investigational compounds adding glucagon receptor agonism
- Potential for even greater weight loss and metabolic improvements
- Early trial data showing 20-25%+ weight reductions
- Research-grade formulations becoming available for mechanistic studies
Optimized Dual Agonists:
- Refined receptor selectivity ratios
- Extended half-lives for less frequent dosing
- Oral formulations under development
- Tissue-selective agonists targeting specific receptor populations
Expanded Therapeutic Indications
Research is exploring tirzepatide applications beyond diabetes and obesity:
🧠 Neurodegenerative Diseases:
- GLP-1 receptors in brain implicated in neuroprotection
- Potential applications in Alzheimer’s disease, Parkinson’s disease
- Anti-inflammatory and anti-apoptotic effects in neural tissue
🫀 Cardiovascular Disease:
- Direct cardioprotective mechanisms beyond risk factor modification
- Potential reduction in heart failure hospitalization
- Anti-atherosclerotic effects
🫁 Non-Alcoholic Fatty Liver Disease (NAFLD/NASH):
- Improvements in hepatic steatosis and inflammation
- Potential disease-modifying effects in NASH
- Ongoing trials evaluating liver-specific outcomes
🦴 Metabolic Bone Disease:
- GIP receptor effects on bone metabolism
- Potential applications in osteoporosis prevention
- Balance between weight loss (potentially negative for bone) and direct bone effects
Personalized Medicine Approaches
Emerging research focuses on identifying which patients respond optimally to tirzepatide:
Pharmacogenomic Markers:
- Genetic variants affecting GIP/GLP-1 receptor expression or function
- Polymorphisms influencing drug metabolism or clearance
- Predictors of side effect susceptibility
Biomarker-Guided Therapy:
- Baseline incretin levels predicting response
- Inflammatory markers correlating with efficacy
- Gut microbiome profiles influencing outcomes
Phenotypic Stratification:
- Identifying obesity subtypes most responsive to incretin therapy
- Diabetes endotypes benefiting most from dual agonism
- Cardiovascular risk profiles guiding treatment selection
Combination Strategies
Research is evaluating tirzepatide in combination with:
- SGLT2 Inhibitors: Complementary mechanisms for glycemic control and weight loss
- Bariatric Surgery: Pharmacotherapy enhancing or maintaining surgical outcomes
- Lifestyle Interventions: Optimizing behavioral support alongside medication
- Other Peptides: Synergistic combinations with amylin analogs, glucagon analogs, etc.
Formulation Innovations
Ongoing development aims to improve delivery and patient experience:
- Oral Formulations: Overcoming peptide degradation in GI tract
- Longer-Acting Versions: Monthly or quarterly administration
- Microneedle Patches: Painless transdermal delivery
- Implantable Devices: Continuous sustained release systems
Access and Equity Considerations
Important research and policy questions include:
- Cost-Effectiveness: Long-term economic analyses informing coverage decisions
- Health Equity: Ensuring access across socioeconomic and demographic groups
- Global Availability: Expanding access in low- and middle-income countries
- Biosimilar Development: Future generic versions potentially reducing costs
For researchers investigating these frontier areas, access to high-quality research materials from suppliers like PEPTIDE PRO remains essential for advancing scientific understanding.
Frequently Asked Questions
What makes tirzepatide different from semaglutide?
Tirzepatide is a dual GIP and GLP-1 receptor agonist, while semaglutide is a selective GLP-1 receptor agonist. This dual mechanism gives tirzepatide broader metabolic activity, generally resulting in greater HbA1c reductions and weight loss in head-to-head trials. The addition of GIP receptor activation provides complementary effects on insulin secretion, adipocyte metabolism, and potentially other tissues.
How long does it take to see results with tirzepatide?
Glycemic improvements can be observed within the first few weeks of treatment, with progressive improvements over 12-24 weeks. Weight loss typically begins within the first month and continues progressively, with maximum effects generally seen after 6-12 months of treatment at maintenance doses. Individual response varies based on dose, adherence, lifestyle factors, and baseline characteristics.
Can tirzepatide be used for weight loss in people without diabetes?
Yes, tirzepatide is approved for chronic weight management in adults with obesity (BMI ≥30 kg/m²) or overweight (BMI ≥27 kg/m²) with at least one weight-related comorbidity, regardless of diabetes status. The SURMOUNT clinical trial program demonstrated substantial weight loss in patients without diabetes. However, availability through NHS for weight management indications may be more restricted than for diabetes treatment.
What should I do if I experience severe nausea or vomiting?
Mild to moderate nausea is common and often improves with continued use. Management strategies include eating smaller, more frequent meals, avoiding high-fat foods, and staying well-hydrated. If nausea is severe or accompanied by persistent vomiting, contact your healthcare provider, as this may lead to dehydration and require temporary dose reduction or additional supportive care. Severe, persistent abdominal pain could indicate pancreatitis and requires immediate medical evaluation.
Is tirzepatide safe during pregnancy?
Tirzepatide has limited safety data in pregnancy. Animal studies showed adverse developmental effects at exposures below recommended human doses. Tirzepatide should be discontinued at least 2 months before planned pregnancy due to its long half-life. Pregnant women or those planning pregnancy should discuss alternative diabetes management strategies with their healthcare provider. The benefits of glycemic control during pregnancy must be balanced against potential fetal risks.
Where can researchers obtain high-quality tirzepatide for laboratory studies?
Research-grade tirzepatide is available from specialized peptide suppliers like PEPTIDE PRO, which provides high-purity formulations with full documentation including Certificates of Analysis. These products are clearly labeled “For Research Use Only” and are intended for approved laboratory applications, not for human consumption. Researchers should ensure their protocols have appropriate institutional ethics approval and follow all regulatory requirements.
How does tirzepatide affect other medications?
Tirzepatide’s delayed gastric emptying may affect absorption of oral medications. Of particular concern are medications requiring rapid absorption or those with narrow therapeutic windows. Tirzepatide may increase the risk of hypoglycemia when combined with insulin or sulfonylureas, often requiring dose adjustments of these medications. Oral contraceptives should be taken at least 1 hour before or 4 hours after tirzepatide injection, or patients should use non-oral contraceptive methods. Always inform healthcare providers of all medications when starting tirzepatide.
What is the cost of tirzepatide in the UK?
Costs vary significantly depending on access pathway:
- NHS Prescription: Standard prescription charges (currently £9.90 per item in England, free in Scotland, Wales, Northern Ireland) for eligible patients
- Private Prescription: Typically £150-300+ per month depending on dose and provider
- Research-Grade: Varies by supplier, quantity, and purity specifications
Supply constraints have occasionally affected availability and pricing. For current pricing on research-grade materials, contact PEPTIDE PRO directly.
Conclusion: The Transformative Potential of Tirzepatide
Tirzepatide (Mounjaro) — Mechanism, Dosage, Side Effects & UK Access represents a paradigm shift in metabolic therapeutics, offering unprecedented efficacy through its innovative dual GIP/GLP-1 receptor agonism. The comprehensive evidence base demonstrates remarkable improvements in glycemic control and weight management that exceed existing therapies, positioning tirzepatide as a cornerstone treatment for type 2 diabetes and obesity.
Key Points to Remember
✅ Dual mechanism targeting both GIP and GLP-1 receptors provides synergistic metabolic benefits ✅ Structured dosing protocol starting at 2.5 mg weekly with gradual escalation optimizes efficacy and tolerability ✅ Gastrointestinal side effects are common but generally manageable and often diminish with continued use ✅ UK access pathways include NHS prescriptions for eligible patients, private healthcare options, and research-grade formulations for laboratory applications ✅ Ongoing research continues to expand our understanding of tirzepatide’s full therapeutic potential across multiple disease states
Actionable Next Steps
For Healthcare Professionals:
- Review current clinical guidelines and local formulary policies for tirzepatide prescribing
- Identify appropriate patient candidates who may benefit from dual agonist therapy
- Develop patient education materials addressing administration, side effects, and expectations
- Establish monitoring protocols aligned with best practice recommendations
For Researchers:
- Explore research questions addressing knowledge gaps in tirzepatide pharmacology, mechanism, or clinical applications
- Source high-quality research-grade materials from reputable suppliers like PEPTIDE PRO
- Design rigorous protocols with appropriate controls and ethical oversight
- Contribute to the expanding evidence base through publication and data sharing
For Patients and Individuals:
- Discuss with your healthcare provider whether tirzepatide may be appropriate for your specific situation
- Understand the commitment required for weekly injections and ongoing lifestyle modification
- Set realistic expectations for outcomes and timeline
- Report side effects or concerns promptly to your medical team
The Broader Impact
Beyond individual patient outcomes, tirzepatide represents a proof-of-concept for multi-receptor agonist approaches that may transform treatment of metabolic diseases. The success of this dual agonist has catalyzed development of even more sophisticated triple agonists and tissue-selective compounds that promise further therapeutic advances.
As research continues and clinical experience expands, our understanding of optimal patient selection, dosing strategies, combination therapies, and long-term outcomes will continue to evolve. The scientific community’s ongoing investigation—supported by access to high-quality research materials from suppliers like PEPTIDE PRO—will be essential for realizing the full potential of this transformative therapeutic class.
Final Thoughts
The journey from basic incretin physiology research to the development of tirzepatide exemplifies the power of translational science. By understanding fundamental biological mechanisms and engineering molecules that harness these pathways, researchers and pharmaceutical developers have created a therapy that meaningfully improves the lives of millions affected by diabetes and obesity.
Whether you’re a healthcare professional optimizing patient care, a researcher advancing scientific knowledge, or an individual seeking information about treatment options, understanding Tirzepatide (Mounjaro) — Mechanism, Dosage, Side Effects & UK Access provides the foundation for informed decision-making and continued progress in metabolic health.
For additional information, technical support, or to explore research-grade peptide options, visit PEPTIDE PRO or contact their team directly.
References
[1] Frias JP, et al. Tirzepatide versus Semaglutide Once Weekly in Patients with Type 2 Diabetes. N Engl J Med. 2021;385(6):503-515.
[2] Rosenstock J, et al. Efficacy and safety of a novel dual GIP and GLP-1 receptor agonist tirzepatide in patients with type 2 diabetes (SURPASS-1): a double-blind, randomised, phase 3 trial. Lancet. 2021;398(10295):143-155.
[3] Jastreboff AM, et al. Tirzepatide Once Weekly for the Treatment of Obesity. N Engl J Med. 2022;387(3):205-216.
[4] Medicines and Healthcare products Regulatory Agency (MHRA). Public Assessment Report: Mounjaro (tirzepatide). 2022.
[5] National Institute for Health and Care Excellence (NICE). Tirzepatide for treating type 2 diabetes. Technology appraisal guidance. 2023.
[6] Thomas MK, et al. Dual GIP and GLP-1 Receptor Agonist Tirzepatide Improves Beta-cell Function and Insulin Sensitivity in Type 2 Diabetes. J Clin Endocrinol Metab. 2021;106(2):388-396.
[7] Heerspink HJL, et al. Effects of tirzepatide versus insulin glargine on kidney outcomes in type 2 diabetes in the SURPASS-4 trial: post-hoc analysis of an open-label, randomised, phase 3 trial. Lancet Diabetes Endocrinol. 2022;10(11):774-785.
[8] Sattar N, et al. Cardiovascular, mortality, and kidney outcomes with GLP-1 receptor agonists in patients with type 2 diabetes: a systematic review and meta-analysis of randomised trials. Lancet Diabetes Endocrinol. 2021;9(10):653-662.
[9] Electronic Medicines Compendium (EMC). Mounjaro 2.5 mg solution for injection in pre-filled pen – Summary of Product Characteristics. Accessed 2025.
[10] Coskun T, et al. LY3298176, a novel dual GIP and GLP-1 receptor agonist for the treatment of type 2 diabetes mellitus: From discovery to clinical proof of concept. Mol Metab. 2018;18:3-14.
