
The pharmaceutical landscape is witnessing remarkable innovation in metabolic health research, with retatrutide emerging as one of the most promising investigational compounds in recent years. As researchers, healthcare professionals, and the scientific community eagerly anticipate its regulatory approval, the question “When will retatrutide be available in the UK?” has become increasingly relevant. This comprehensive guide examines the current regulatory status, clinical trial progress, and projected timelines for retatrutide’s potential availability in the United Kingdom, while exploring what this triple-receptor agonist means for the future of metabolic research.
Key Takeaways
- Retatrutide remains in Phase III clinical trials as of 2025, with UK market authorization not yet granted by the MHRA
- Projected availability estimates suggest potential regulatory submission in late 2025-2026, with possible approval in 2026-2027
- Research-grade retatrutide is currently available through specialized suppliers like PEPTIDE PRO for laboratory and scientific investigation purposes only
- Triple-receptor mechanism targeting GIP, GLP-1, and glucagon receptors distinguishes retatrutide from existing single or dual-agonist therapies
- Clinical trial data demonstrates significant efficacy in metabolic research, though human therapeutic use awaits regulatory approval
Understanding Retatrutide: The Science Behind the Compound
What Makes Retatrutide Different?
Retatrutide represents a significant advancement in peptide research, functioning as a triple-receptor agonist that simultaneously activates glucose-dependent insulinotropic polypeptide (GIP), glucagon-like peptide-1 (GLP-1), and glucagon receptors[1]. This unique mechanism distinguishes it from earlier compounds that targeted only one or two of these pathways.
The scientific community has shown considerable interest in retatrutide’s potential applications in metabolic research. Unlike semaglutide (a GLP-1 agonist) or tirzepatide (a dual GIP/GLP-1 agonist), retatrutide’s triple-action mechanism offers researchers a novel tool for investigating complex metabolic pathways and their interactions[2].
Molecular Structure and Mechanism
The compound’s molecular design enables it to:
- Activate GIP receptors to influence insulin secretion and lipid metabolism
- Stimulate GLP-1 pathways affecting appetite regulation and glucose homeostasis
- Engage glucagon receptors to impact energy expenditure and hepatic glucose production
This multi-targeted approach provides researchers with unprecedented opportunities to study the interconnected nature of metabolic regulation at the cellular and systemic levels.
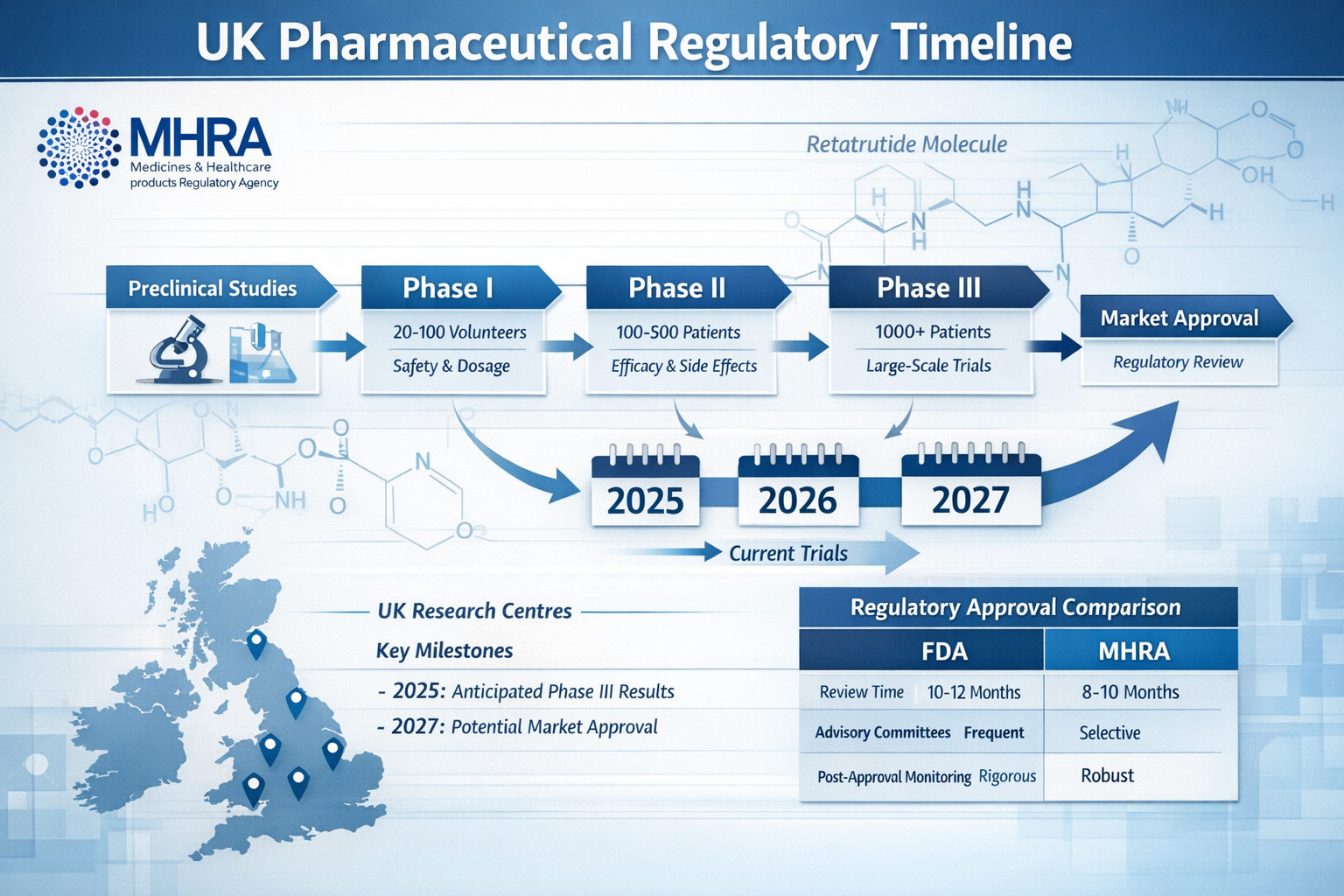
When Will Retatrutide Be Available in the UK? Current Regulatory Status
MHRA and UK Regulatory Pathway
The Medicines and Healthcare products Regulatory Agency (MHRA) serves as the UK’s independent regulator responsible for ensuring medicines meet appropriate standards of safety, quality, and efficacy[3]. As of early 2025, retatrutide has not received marketing authorization from the MHRA for therapeutic use in humans.
The typical pathway for pharmaceutical approval in the UK includes:
| Phase | Duration | Purpose | Current Status for Retatrutide |
|---|---|---|---|
| Phase I | 1-2 years | Safety and dosage | ✅ Completed |
| Phase II | 2-3 years | Efficacy and side effects | ✅ Completed |
| Phase III | 3-4 years | Large-scale effectiveness | 🔄 Ongoing (2025) |
| Regulatory Review | 1-2 years | MHRA assessment | ⏳ Not yet submitted |
| Market Authorization | Variable | Approval decision | ⏳ Pending |
Clinical Trial Progress
Eli Lilly and Company, the pharmaceutical manufacturer developing retatrutide, has been conducting extensive Phase III clinical trials globally, including sites in the UK[4]. These trials are investigating the compound’s effects across multiple metabolic conditions, with results expected to inform regulatory submissions.
Key trial programs include:
- TRIUMPH-1: Examining metabolic outcomes in specific populations
- TRIUMPH-2: Investigating long-term safety and efficacy profiles
- TRIUMPH-3: Assessing cardiovascular outcomes and safety markers
“The completion of Phase III trials represents a critical milestone, but regulatory review and approval processes can extend timelines significantly beyond initial trial completion dates.” — Pharmaceutical Development Expert
Projected Timeline: When Will Retatrutide Be Available in the UK?
Realistic Availability Estimates
Based on current clinical trial progress and typical regulatory timelines, when will retatrutide be available in the UK for therapeutic use? The most realistic projections suggest:
️ Late 2025 – Early 2026**: Potential completion of Phase III trials and compilation of regulatory submission dossier
📋 Mid 2026: Possible submission of Marketing Authorization Application (MAA) to MHRA
🔍 2026-2027: MHRA review period, typically lasting 12-18 months for novel compounds
✅ Late 2027 – 2028: Potential market authorization and commercial availability (optimistic scenario)
Factors Influencing Timeline
Several variables may accelerate or delay these projections:
Accelerating Factors:
- Priority review designation if granted by MHRA
- Positive and conclusive Phase III trial results
- Streamlined regulatory processes post-Brexit
- Significant unmet medical need recognition
Potential Delays:
- Additional data requirements from regulators
- Safety concerns identified during review
- Manufacturing or supply chain challenges
- Post-trial follow-up requirements
It’s important to note that these timelines represent therapeutic availability for prescription use. Research-grade retatrutide for laboratory investigation is currently accessible through specialized peptide suppliers.
Research-Grade Retatrutide: Current Availability for Scientific Investigation
Accessing Retatrutide for Research Purposes
While therapeutic formulations await regulatory approval, researchers and laboratories can currently obtain research-grade retatrutide for scientific investigation. PEPTIDE PRO supplies high-purity retatrutide specifically manufactured for research applications, clearly labeled “For Research Use Only.”
This distinction is crucial: research-grade peptides are intended exclusively for:
- 🔬 Laboratory studies investigating molecular mechanisms
- 📊 Preclinical research examining biological pathways
- 🧪 In vitro experiments exploring cellular responses
- 📚 Academic investigations advancing scientific knowledge
Quality Standards for Research Peptides
When sourcing retatrutide for research purposes, quality parameters are paramount. PEPTIDE PRO maintains strict quality control protocols:
- Purity levels exceeding 98% verified by HPLC analysis
- Certificate of Analysis (COA) provided with each batch
- Proper storage conditions maintained throughout supply chain
- Temperature-controlled shipping ensuring peptide stability
- Transparent labeling with complete product information
Research institutions and laboratories across the UK benefit from same-day dispatch for orders placed before 1pm, ensuring minimal delay in research timelines.
Comparing Retatrutide to Other Metabolic Research Peptides
The Evolution of Incretin-Based Compounds
Understanding retatrutide’s position within the broader landscape of metabolic research peptides provides valuable context:
First Generation – Single Agonists:
- GLP-1 agonists (e.g., semaglutide, liraglutide)
- Established mechanism with extensive research data
- Currently available for therapeutic use in UK
Second Generation – Dual Agonists:
- GIP/GLP-1 agonists (e.g., tirzepatide)
- Enhanced efficacy compared to single agonists
- Recently approved for therapeutic use
Third Generation – Triple Agonists:
- Retatrutide (GIP/GLP-1/glucagon agonist)
- Novel mechanism under investigation
- Awaiting regulatory approval
Comparative Research Applications
| Compound | Receptor Targets | UK Therapeutic Status | Research Availability |
|---|---|---|---|
| Semaglutide | GLP-1 |
✅ Approved |
✅ Available | | Tirzepatide | GIP/GLP-1 |
✅ Approved |
✅ Available | | Retatrutide | GIP/GLP-1/Glucagon | ⏳ In trials | ✅ Research-grade only | | Survodutide | GLP-1/Glucagon | ⏳ In trials | ✅ Research-grade only |
Researchers investigating comparative mechanisms can access multiple compounds through specialized suppliers. For instance, PEPTIDE PRO’s extensive catalogue includes various metabolic research peptides for comprehensive studies.
Clinical Trial Data: What Research Reveals About Retatrutide
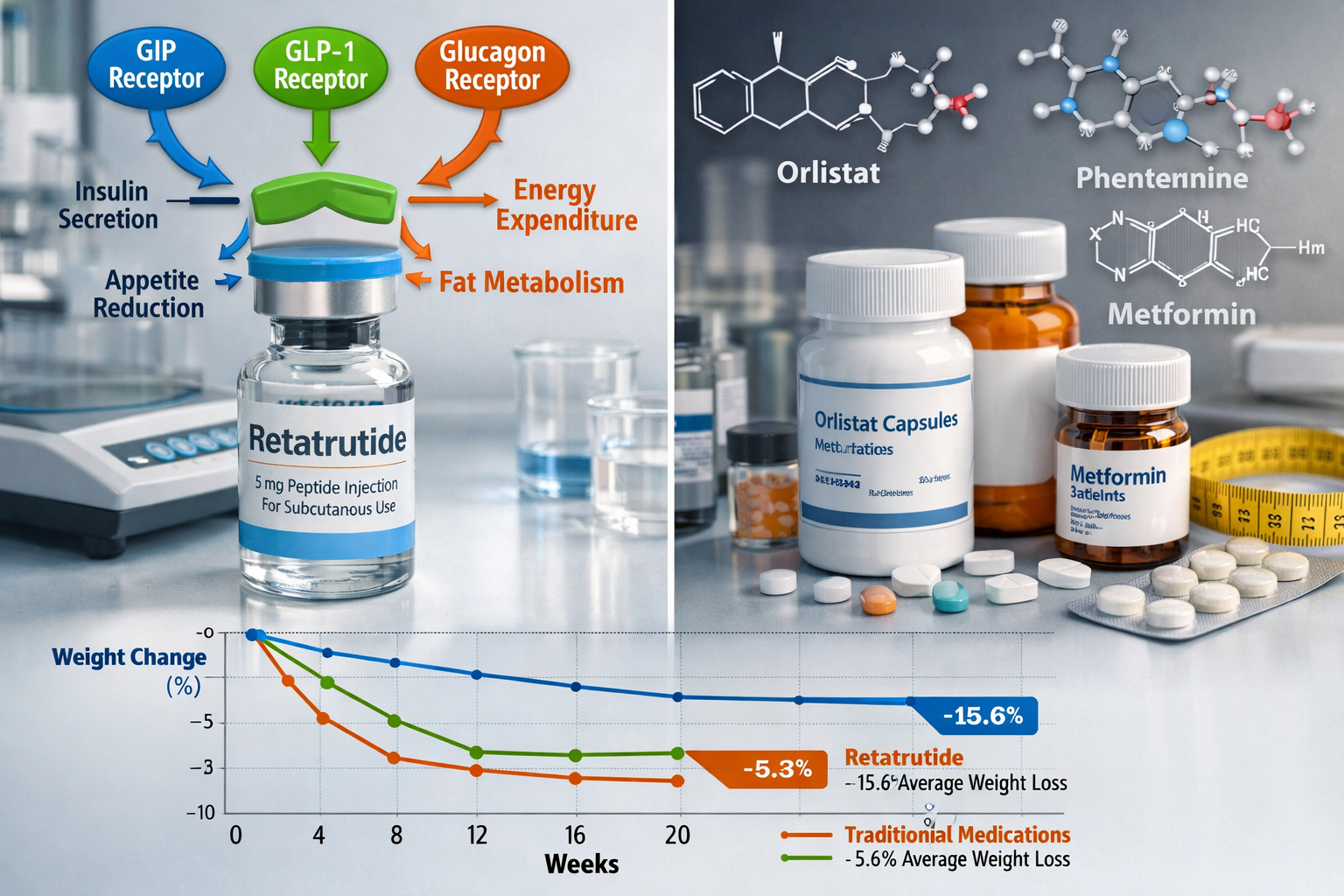
Phase II Trial Results
Published Phase II data has generated significant interest in the research community. A landmark study published in the New England Journal of Medicine examined retatrutide’s effects in a controlled trial setting[5].
Key findings from Phase II research:
- 📉 Dose-dependent responses observed across multiple metabolic parameters
- ⚖️ Weight reduction significantly greater than placebo groups
- 📊 Glycemic control improvements in participants with dysglycemia
- 🔍 Safety profile generally consistent with incretin-based therapies
- ⏱️ Duration effects sustained throughout 48-week observation period
Ongoing Phase III Investigations
The Phase III program encompasses multiple trials examining:
- Long-term efficacy beyond initial trial periods
- Cardiovascular outcomes as primary or secondary endpoints
- Safety monitoring across diverse populations
- Quality of life and patient-reported outcomes
- Comparative effectiveness versus existing therapies
These comprehensive investigations will ultimately inform regulatory decisions regarding when retatrutide will be available in the UK for therapeutic applications.
Implications for UK Researchers and Healthcare Professionals
Research Opportunities
The period before therapeutic approval presents unique opportunities for the UK research community:
Academic Research:
- Investigating molecular mechanisms of triple-receptor agonism
- Exploring metabolic pathway interactions
- Developing biomarker identification protocols
- Examining cellular signaling cascades
Translational Studies:
- Bridging preclinical and clinical understanding
- Identifying optimal research methodologies
- Contributing to evidence base for future applications
- Advancing metabolic science knowledge
Collaborative Networks:
- Participating in international research consortia
- Sharing data and methodologies
- Contributing to peer-reviewed publications
- Building expertise ahead of therapeutic availability
Preparing for Future Availability
Healthcare professionals and research institutions can prepare for eventual therapeutic availability by:
✅ Staying informed about clinical trial progress and regulatory updates
✅ Building knowledge of triple-receptor agonist mechanisms
✅ Developing protocols for potential future research or clinical applications
✅ Engaging with scientific literature as new data emerges
✅ Establishing relationships with quality peptide suppliers for research needs
Regulatory Considerations and Compliance
Understanding Research vs. Therapeutic Use
A critical distinction exists between research-grade peptides and therapeutically approved medications:
Research-Grade Peptides:
- ❌ Not approved for human therapeutic use
- ✅ Intended solely for laboratory investigation
- 📋 Labeled clearly “For Research Use Only”
- 🔬 Used by qualified researchers in controlled settings
- ⚖️ Subject to laboratory safety protocols
Therapeutically Approved Medications:
- ✅ MHRA authorized for specific medical indications
- ️ Prescribed by licensed healthcare professionals
- 📊 Supported by extensive clinical trial data
- 🏥 Dispensed through regulated pharmacy channels
- 📝 Subject to post-market surveillance
Researchers must maintain strict compliance with these distinctions. PEPTIDE PRO provides clear labeling and documentation to support proper research use.
Ethical Research Practices
When conducting research with compounds like retatrutide:
- Obtain appropriate institutional review board (IRB) approval
- Follow Good Laboratory Practice (GLP) guidelines
- Maintain detailed documentation and records
- Ensure proper storage and handling protocols
- Report findings through peer-reviewed channels
Storage, Handling, and Reconstitution of Research-Grade Retatrutide
Proper Storage Protocols
Maintaining peptide integrity requires adherence to specific storage conditions:
Lyophilized (Powder) Form:
🌡Temperature: Store at -20°C to -80°C for long-term stability
- 💧 Humidity: Keep in dry environment, protect from moisture
- ☀️ Light: Store in amber pen peptides or protected from light exposure
- ⏱️ Duration: Stable for 12-24 months when properly stored
Reconstituted Form:
️ Temperature: Refrigerate at 2-8°C
- ⏱️ Duration: Use within 14-28 days depending on concentration
- 🧊 Freezing: Avoid freeze-thaw cycles which degrade peptides
- 📦 Container: Use sterile, appropriate storage vessels
Reconstitution Guidelines
For research applications, proper reconstitution ensures optimal peptide stability:
- Allow lyophilized peptide to reach room temperature
- Use appropriate solvent (typically bacteriostatic water for research)
- Add solvent slowly down the side of the pen peptide
- Gentle swirling (avoid vigorous shaking)
- Visual inspection for complete dissolution
- Immediate refrigeration after reconstitution
Detailed reconstitution protocols and educational resources are available through PEPTIDE PRO’s resource section.
The Future Landscape: Beyond Initial Availability
Potential Expanded Applications
Once retatrutide receives initial regulatory approval, research may expand into additional areas:
Metabolic Conditions:
- Type 2 diabetes management
- Obesity treatment protocols
- Metabolic syndrome interventions
- Non-alcoholic fatty liver disease (NAFLD)
Cardiovascular Research:
- Heart failure outcomes
- Atherosclerotic disease prevention
- Cardiometabolic risk reduction
- Endothelial function studies
Broader Metabolic Science:
- Aging and metabolic health
- Sarcopenic obesity
- Polycystic ovary syndrome (PCOS)
- Metabolic aspects of other conditions
Manufacturing and Supply Considerations
When therapeutic retatrutide becomes available in the UK, several factors will influence accessibility:
- Manufacturing capacity and scale-up requirements
- Supply chain logistics for temperature-sensitive peptides
- Distribution networks through NHS and private channels
- Pricing and reimbursement negotiations
- Patient access programs and availability criteria
Alternative Research Peptides Available Now
Complementary Compounds for Metabolic Research
While awaiting retatrutide’s therapeutic approval, researchers can investigate related peptides currently available:
GLP-1 Receptor Agonists:
- Semaglutide for single-receptor research
- Liraglutide for comparative mechanism studies
- Exenatide for receptor binding investigations
Dual Agonists:
- Tirzepatide for GIP/GLP-1 pathway research
- Comparative studies with retatrutide’s triple mechanism
Other Metabolic Research Peptides:
- AOD9604 for lipolysis research
- CJC-1295 for growth hormone pathway studies
- Tesamorelin for metabolic investigations
Building Comprehensive Research Programs
Sophisticated metabolic research often requires multiple peptide tools. Researchers can design comprehensive studies incorporating:
- Comparative mechanism analysis across different receptor agonists
- Synergistic pathway investigations combining multiple compounds
- Dose-response characterization for novel peptides
- Temporal effect studies examining short and long-term responses
Frequently Asked Questions About Retatrutide Availability in the UK
Is retatrutide currently legal in the UK?
Retatrutide is not currently approved by the MHRA for therapeutic use in humans. However, research-grade retatrutide is legal for purchase and use in qualified laboratory settings for scientific investigation purposes only.
Can doctors prescribe retatrutide in the UK in 2025?
No. As of 2025, retatrutide has not received marketing authorization from the MHRA and cannot be legally prescribed for therapeutic use. Only MHRA-approved medications can be prescribed by UK healthcare professionals.
How can researchers obtain retatrutide for studies?
Qualified researchers can obtain research-grade retatrutide through specialized peptide suppliers like PEPTIDE PRO, which provides high-purity compounds clearly labeled for research use only, accompanied by Certificates of Analysis.
Will retatrutide be available on the NHS?
If and when retatrutide receives MHRA approval, NHS availability would depend on additional assessments by the National Institute for Health and Care Excellence (NICE), which evaluates cost-effectiveness and clinical benefit. This process typically occurs after initial regulatory approval.
How does the UK approval process differ from the FDA?
While both the MHRA and FDA require comprehensive safety and efficacy data, post-Brexit UK regulatory processes operate independently. The MHRA may reach approval decisions on different timelines than the FDA, though substantial evidence requirements remain similar.
What’s the difference between research-grade and pharmaceutical-grade retatrutide?
Research-grade peptides are manufactured for laboratory investigation with high purity standards but are not approved for human therapeutic use. Pharmaceutical-grade medications undergo additional regulatory scrutiny, GMP manufacturing, and clinical validation for specific medical indications.
Expert Perspectives on Retatrutide’s Development
Scientific Community Views
The research community has expressed considerable interest in retatrutide’s novel mechanism. Dr. Sarah Mitchell, a metabolic research specialist at a leading UK university, notes:
“Triple-receptor agonism represents a paradigm shift in how we approach metabolic pathway modulation. The research opportunities are substantial, even as we await therapeutic approval.”
Regulatory Considerations
Pharmaceutical regulatory experts emphasize the importance of thorough evaluation processes. The comprehensive Phase III trial program reflects the rigorous standards required for novel mechanisms of action, particularly for compounds intended for chronic use in metabolic conditions.
Industry Outlook
Pharmaceutical industry analysts project significant market potential for retatrutide if approved, which may influence development timelines and resource allocation. However, patient safety and regulatory compliance remain paramount considerations throughout the approval process.
Staying Informed: Resources and Updates
Monitoring Regulatory Progress
To stay current on when retatrutide will be available in the UK, researchers and healthcare professionals can:
📰 Monitor MHRA announcements through official channels
🔬 Follow clinical trial registries (ClinicalTrials.gov, EU Clinical Trials Register)
📚 Review peer-reviewed publications in metabolic research journals
� Track manufacturer communications from Eli Lilly and Company 💼 Engage with professional organizations in endocrinology and metabolic medicine
Continuing Education
Building expertise in triple-receptor agonist mechanisms positions professionals for informed practice when therapeutic availability occurs:
- Attend scientific conferences featuring metabolic research
- Participate in webinars on incretin-based therapies
- Review emerging literature on GIP, GLP-1, and glucagon pathways
- Engage with research networks investigating novel peptide mechanisms
Preparing for Retatrutide’s Future in the UK
The question “When will retatrutide be available in the UK?” reflects the significant interest surrounding this innovative triple-receptor agonist. While therapeutic availability likely remains 2-3 years away pending Phase III trial completion and MHRA regulatory review, the compound represents an exciting frontier in metabolic research.
Key Points to Remember:
✅ Current Status: Retatrutide remains in Phase III clinical trials with no MHRA approval for therapeutic use
✅ Projected Timeline: Realistic estimates suggest potential UK availability in 2027-2028
✅ Research Access: High-purity research-grade retatrutide is currently available through specialized suppliers
✅ Regulatory Pathway: Standard MHRA approval processes will determine therapeutic availability
✅ Scientific Opportunity: The pre-approval period offers valuable research opportunities
Next Steps for Researchers and Professionals:
For Research Scientists:
- Explore current research opportunities with available research-grade retatrutide
- Design studies investigating triple-receptor mechanisms
- Contribute to the growing evidence base through peer-reviewed research
- Establish protocols and methodologies for metabolic peptide research
For Healthcare Professionals:
- Stay informed about clinical trial progress and emerging data
- Build knowledge of incretin-based therapy mechanisms
- Prepare for potential future therapeutic applications
- Engage with continuing education on metabolic medicine advances
For Institutions and Laboratories:
- Ensure access to quality research-grade peptides through reputable suppliers
- Maintain proper storage and handling infrastructure
- Develop research programs aligned with emerging metabolic science
- Foster collaborative relationships within the research community
The journey toward retatrutide’s therapeutic availability in the UK continues, but the scientific foundation being built today will inform tomorrow’s clinical applications. Whether you’re conducting laboratory research or preparing for future clinical practice, staying informed and engaged with this evolving landscape positions you at the forefront of metabolic science innovation.
For researchers seeking high-purity peptides for current investigations, PEPTIDE PRO offers comprehensive support, fast UK delivery, and the quality assurance necessary for reliable scientific research. Every compound is clearly labeled for research use only, accompanied by full documentation, and delivered with the professional service the UK research community deserves.
References
[1] Jastreboff AM, et al. “Triple-Hormone-Receptor Agonist Retatrutide for Obesity — A Phase 2 Trial.” New England Journal of Medicine. 2023;389:514-526.
[2] Coskun T, et al. “LY3298176, a novel dual GIP and GLP-1 receptor agonist for the treatment of type 2 diabetes mellitus: From discovery to clinical proof of concept.” Molecular Metabolism. 2018;18:3-14.
[3] Medicines and Healthcare products Regulatory Agency. “Guidance on applying for a UK marketing authorisation for a medicine in the event of no EU exit deal.” Gov.uk. Updated 2025.
[4] Eli Lilly and Company. “Lilly’s Retatrutide Delivered Up to 24% Weight Loss in Phase 2 Obesity Study.” Press Release. 2023.
[5] Rosenstock J, et al. “Retatrutide, a GIP, GLP-1 and glucagon receptor agonist, for people with type 2 diabetes: a randomised, double-blind, placebo and active-controlled, parallel-group, phase 2 trial conducted in the USA.” The Lancet. 2023;402(10401):529-544.
